Abstract
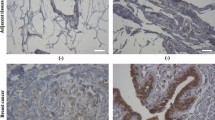
Breast cancer is one of the most common cancers in women worldwide and metastasis is the major cause of breast cancer death. Development of new therapeutic agents for inhibiting breast cancer metastasis is therefore an urgent need. We previously demonstrated that recombinant DNA-derived viral capsid protein VP1 (rVP1) of foot-and-mouth disease virus-induced apoptosis of MCF-7 breast cancer cells in vitro. Here, we investigated whether rVP1 exhibits any inhibitory effects on migration/metastasis and human epidermal growth factor receptor 2 (HER-2), a well-known biomarker for poor prognosis of breast cancer. The effects of rVP1 on cancer cell migration/invasion and metastasis were evaluated using Transwell migration assay and animal cancer models of metastasis. Western blotting, RT-PCR, flow cytometry, immunohistochemistry, and immunofluorescence staining techniques were used to investigate the effects of rVP1 on HER-2 and signal transduction mediators. Non-cytotoxic concentrations of rVP1-induced mesenchymal-epithelial transition and significantly suppressed AP-2α and HER-2 expression as well as the migration and invasion of a variety of breast cancer cell lines in a β1-integrin-dependent manner in vitro. Gross and histopathologic examinations showed that rVP1 also suppressed metastasis of several breast cancer cell lines, including HER-2-overexpressing SK-BR-3 and BT-474 cells to lung, liver, or peripheral lymph node in orthotopic allograft/xenograft murine models. In addition, rVP1 significantly prolonged survival in breast cancer-bearing mice. Notably, no apparent side effects of rVP1 were detected, as shown by normal complete blood count levels and serum biochemistry profiles, including AST, ALT, BUN, and creatine. This study demonstrates that rVP1 suppresses the migration, invasion, and metastasis of breast cancer cells via binding to β1 integrin receptor and down-regulation of AP-2α and HER-2 expression. The effectiveness of rVP1 on inhibiting migration/metastasis of breast cancer and HER-2 expression suggests that it may be suitable for serving as potential therapeutics for metastatic breast cancer particularly HER-2-overexpressing cancer.







Similar content being viewed by others
Abbreviations
- rVP1:
-
Recombinant viral capsid protein 1
- FMDV:
-
Foot-and-mouth disease virus
- MET:
-
Mesenchymal-to-epithelial transition
- HER-2:
-
Human epidermal growth factor receptor 2
- AP-2α:
-
Activator protein-2α
- PMMEC:
-
Primary mouse mammary epithelial cells
References
Jemal A, Siegel R, Xu J, Ward E (2010) Cancer statistics, 2010. CA Cancer J Clin 60:277–300. doi:10.3322/caac.20073
Jemal A, Siegel R, Ward E, Murray T, Xu J, Smigal C, Thun MJ (2006) Cancer statistics, 2006. CA Cancer J Clin 56:106–130. doi:56/2/106
Jemal A, Siegel R, Ward E, Murray T, Xu J, Thun MJ (2007) Cancer statistics, 2007. CA Cancer J Clin 57:43–66. doi:57/1/43
Giordano SH, Hortobagyi GN (2003) Inflammatory breast cancer: clinical progress and the main problems that must be addressed. Breast Cancer Res 5:284–288. doi:10.1186/bcr608
Tai W, Mahato R, Cheng K (2010) The role of HER2 in cancer therapy and targeted drug delivery. J Control Release 146:264–275. doi:10.1016/j.jconrel.2010.04.009
Powe DG, Akhtar G, Habashy HO, Abdel-Fatah T, Rakha EA, Green AR, Ellis IO (2009) Investigating AP-2 and YY1 protein expression as a cause of high HER2 gene transcription in breast cancers with discordant HER2 gene amplification. Breast Cancer Res 11:R90. doi:10.1186/bcr2461
Breuhahn K, Longerich T, Schirmacher P (2006) Dysregulation of growth factor signaling in human hepatocellular carcinoma. Oncogene 25:3787–3800. doi:10.1038/sj.onc.1209556
Ciardiello F, Tortora G (2003) Epidermal growth factor receptor (EGFR) as a target in cancer therapy: understanding the role of receptor expression and other molecular determinants that could influence the response to anti-EGFR drugs. Eur J Cancer 39:1348–1354. doi:S0959804903002351
Mendelsohn J, Baselga J (2003) Status of epidermal growth factor receptor antagonists in the biology and treatment of cancer. J Clin Oncol 21:2787–2799. doi:10.1200/JCO.2003.01.504
Nadella KS, Jones GN, Trimboli A, Stratakis CA, Leone G, Kirschner LS (2008) Targeted deletion of Prkar1a reveals a role for protein kinase A in mesenchymal-to-epithelial transition. Cancer Res 68:2671–2677. doi:10.1158/0008-5472.CAN-07-6002
Davidson B, Konstantinovsky S, Nielsen S, Dong HP, Berner A, Vyberg M, Reich R (2004) Altered expression of metastasis-associated and regulatory molecules in effusions from breast cancer patients: a novel model for tumor progression. Clin Cancer Res 10:7335–7346. doi:10.1158/1078-0432.CCR-04-0183
Stickeler E, Klar M, Watermann D, Geibel A, Foldi M, Hasenburg A, Gitsch G (2009) Pegylated liposomal doxorubicin and trastuzumab as 1st and 2nd line therapy in her2/neu positive metastatic breast cancer: a multicenter phase II trial. Breast Cancer Res Treat 117:591–598. doi:10.1007/s10549-008-0306-9
McGrogan BT, Gilmartin B, Carney DN, McCann A (2008) Taxanes, microtubules and chemoresistant breast cancer. Biochim Biophys Acta 1785:96–132. doi:10.1016/j.bbcan.2007.10.004
van Regenmortel MH, Mayo MA, Fauquet CM, Maniloff J (2000) Virus nomenclature: consensus versus chaos. Arch Virol 145:2227–2232
Belsham GJ (1993) Distinctive features of foot-and-mouth disease virus, a member of the picornavirus family; aspects of virus protein synthesis, protein processing and structure. Prog Biophys Mol Biol 60:241–260. doi:0079-6107(93)90016-D
Berinstein A, Roivainen M, Hovi T, Mason PW, Baxt B (1995) Antibodies to the vitronectin receptor (integrin alpha V beta 3) inhibit binding and infection of foot-and-mouth disease virus to cultured cells. J Virol 69:2664–2666
Mason PW, Rieder E, Baxt B (1994) RGD sequence of foot-and-mouth disease virus is essential for infecting cells via the natural receptor but can be bypassed by an antibody-dependent enhancement pathway. Proc Natl Acad Sci USA 91:1932–1936
Logan D, Abu-Ghazaleh R, Blakemore W, Curry S, Jackson T, King A, Lea S, Lewis R, Newman J, Parry N et al (1993) Structure of a major immunogenic site on foot-and-mouth disease virus. Nature 362:566–568. doi:10.1038/362566a0
Wang JH, Liang CM, Peng JM, Shieh JJ, Jong MH, Lin YL, Sieber M, Liang SM (2003) Induction of immunity in swine by purified recombinant VP1 of foot-and-mouth disease virus. Vaccine 21:3721–3729
Jackson T, Blakemore W, Newman JW, Knowles NJ, Mould AP, Humphries MJ, King AM (2000) Foot-and-mouth disease virus is a ligand for the high-affinity binding conformation of integrin alpha5beta1: influence of the leucine residue within the RGDL motif on selectivity of integrin binding. J Gen Virol 81:1383–1391
Jackson T, Sheppard D, Denyer M, Blakemore W, King AM (2000) The epithelial integrin alphavbeta6 is a receptor for foot-and-mouth disease virus. J Virol 74:4949–4956
Jackson T, Clark S, Berryman S, Burman A, Cambier S, Mu D, Nishimura S, King AM (2004) Integrin alphavbeta8 functions as a receptor for foot-and-mouth disease virus: role of the beta-chain cytodomain in integrin-mediated infection. J Virol 78:4533–4540
Neff S, Sa-Carvalho D, Rieder E, Mason PW, Blystone SD, Brown EJ, Baxt B (1998) Foot-and-mouth disease virus virulent for cattle utilizes the integrin alpha(v)beta3 as its receptor. J Virol 72:3587–3594
Jackson T, Mould AP, Sheppard D, King AM (2002) Integrin alphavbeta1 is a receptor for foot-and-mouth disease virus. J Virol 76:935–941
Peng JM, Liang SM, Liang CM (2004) VP1 of foot-and-mouth disease virus induces apoptosis via the Akt signaling pathway. J Biol Chem 279:52168–52174. doi:10.1074/jbc.M403686200
Peng JM, Chen YH, Hung SW, Chiu CF, Ho MY, Lee YJ, Lai TC, Hsiao M, Liang CM, Liang SM (2012) Recombinant viral protein promotes apoptosis and suppresses invasion of ovarian adenocarcinoma cells by targeting alpha5beta1 integrin to down-regulate Akt and MMP-2. Br J Pharmacol 165:479–493. doi:10.1111/j.1476-5381.2011.01581.x
Chen TA, Wang JL, Hung SW, Chu CL, Cheng YC, Liang SM (2011) Recombinant VP1, an Akt inhibitor, suppresses progression of hepatocellular carcinoma by inducing apoptosis and modulation of CCL2 production. PLoS ONE 6:e23317. doi:10.1371/journal.pone.0023317
Chiu CF, Peng JM, Hung SW, Liang CM, Liang SM (2012) Recombinant viral capsid protein VP1 suppresses migration and invasion of human cervical cancer by modulating phosphorylated prohibitin in lipid rafts. Cancer Lett 320:205–214. doi:10.1016/j.canlet.2012.02.027
Shieh JJ, Liang CM, Chen CY, Lee F, Jong MH, Lai SS, Liang SM (2001) Enhancement of the immunity to foot-and-mouth disease virus by DNA priming and protein boosting immunization. Vaccine 19:4002–4010. doi:S0264-410X(01)00114-1
Yang NS, Wang JH, Lin KF, Wang CY, Kim SA, Yang YL, Jong MH, Kuo TY, Lai SS, Cheng RH, Chan MT, Liang SM (2005) Comparative studies of the capsid precursor polypeptide P1 and the capsid protein VP1 cDNA vectors for DNA vaccination against foot-and-mouth disease virus. J Gene Med 7:708–717. doi:10.1002/jgm.723
Yang SP, Song ST, Tang ZM, Song HF (2003) Optimization of antisense drug design against conservative local motif in simulant secondary structures of HER-2 mRNA and QSAR analysis. Acta Pharmacol Sin 24:897–902
Huang CC, Lo CP, Chiu CY, Shyur LF (2010) Deoxyelephantopin, a novel multifunctional agent, suppresses mammary tumour growth and lung metastasis and doubles survival time in mice. Br J Pharmacol 159:856–871. doi:10.1111/j.1476-5381.2009.00581.x
Guccione M, Silbiger S, Lei J, Neugarten J (2002) Estradiol upregulates mesangial cell MMP-2 activity via the transcription factor AP-2. Am J Physiol Renal Physiol 282:F164–F169. doi:10.1152/ajprenal.00318.2000
Wei YY, Chen YJ, Hsiao YC, Huang YC, Lai TH, Tang CH (2008) Osteoblasts-derived TGF-beta1 enhance motility and integrin upregulation through Akt, ERK, and NF-kappaB-dependent pathway in human breast cancer cells. Mol Carcinog 47:526–537. doi:10.1002/mc.20411
Lehmann BD, Bauer JA, Chen X, Sanders ME, Chakravarthy AB, Shyr Y, Pietenpol JA (2011) Identification of human triple-negative breast cancer subtypes and preclinical models for selection of targeted therapies. J Clin Invest 121:2750–2767. doi:10.1172/JCI45014
Nanni P, Nicoletti G, De Giovanni C, Croci S, Astolfi A, Landuzzi L, Di Carlo E, Iezzi M, Musiani P, Lollini PL (2003) Development of rhabdomyosarcoma in HER-2/neu transgenic p53 mutant mice. Cancer Res 63:2728–2732
White DE, Kurpios NA, Zuo D, Hassell JA, Blaess S, Mueller U, Muller WJ (2004) Targeted disruption of beta1-integrin in a transgenic mouse model of human breast cancer reveals an essential role in mammary tumor induction. Cancer Cell 6:159–170. doi:10.1016/j.ccr.2004.06.025
Huveneers S, Truong H, Danen HJ (2007) Integrins: signaling, disease, and therapy. Int J Radiat Biol 83:743–751. doi:10.1080/09553000701481808
Ramirez NE, Zhang Z, Madamanchi A, Boyd KL, O’Rear LD, Nashabi A, Li Z, Dupont WD, Zijlstra A, Zutter MM (2011) The alpha(2)beta(1) integrin is a metastasis suppressor in mouse models and human cancer. J Clin Invest 121:226–237. doi:10.1172/JCI42328
Henderson IC, Berry DA, Demetri GD, Cirrincione CT, Goldstein LJ, Martino S, Ingle JN, Cooper MR, Hayes DF, Tkaczuk KH, Fleming G, Holland JF, Duggan DB, Carpenter JT, Frei E 3rd, Schilsky RL, Wood WC, Muss HB, Norton L (2003) Improved outcomes from adding sequential Paclitaxel but not from escalating Doxorubicin dose in an adjuvant chemotherapy regimen for patients with node-positive primary breast cancer. J Clin Oncol 21:976–983
Yang J, Richards J, Guzman R, Imagawa W, Nandi S (1980) Sustained growth in primary culture of normal mammary epithelial cells embedded in collagen gels. Proc Natl Acad Sci USA 77:2088–2092
Acknowledgments
We thank Dr. Michael Hsiao (Genomics Research Center, Academia Sinica, Taiwan) for kindly providing the H184B5F5, BT-474, and SK-BR-3 cell lines. We also thank the Taiwan Mouse Clinic which is funded by the National Research Program for Genomic Medicine (NRPGM) and the National Science Council (NSC) of Taiwan, and the National Laboratory Animal Center, Taipei, Taiwan for technical support with the complete blood count (CBC) and serum biochemistry assay; and Miss Miranda Loney, Institute of Molecular Biology and Agricultural Biotechnology Research Center English Editor’s Office, Academia Sinica, Taiwan, for English editorial assistance. This work was supported by the NSC, Taiwan (grant NSC 99-2313-B-001-004-MY3 to Shu-Mei Liang), and Academia Sinica (to Shu-Mei Liang and Chi-Ming Liang).
Conflict of interest
The authors declare that they have no conflict of interests.
Author information
Authors and Affiliations
Corresponding authors
Additional information
An erratum to this article is available at http://dx.doi.org/10.1007/s10549-016-3711-5.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Fig. 1
rVP1 at concentration below 0.2 μM does not decrease cell viability and induce apoptosis of breast cancer cells. Breast cancer cell lines of mouse (TS/A and 4T1) and human (MDA-MB-231 and MCF-7) as well as primary mouse mammary epithelial cells (PMMEC) were treated with various serial two-fold diluted concentrations of rVP1 (0.1 μM-1.6 μM) at 37 °C for 24 h in serum-free medium. Cell viability was determined using MTT assay. Results were reproduced in three independent experiments. Data represent mean ± SD (n = 4). (TIF 503 KB)
Rights and permissions
About this article
Cite this article
Hung, SW., Chiu, CF., Chen, TA. et al. Recombinant viral protein VP1 suppresses HER-2 expression and migration/metastasis of breast cancer. Breast Cancer Res Treat 136, 89–105 (2012). https://doi.org/10.1007/s10549-012-2238-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-012-2238-7