Abstract
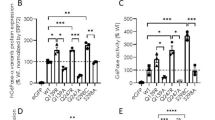
Deficiency of phosphomannomutase (PMM2, MIM#601785) is the most common congenital disorder of glycosylation. Herein we report the genetic analysis of 22 Spanish PMM2 deficient patients and the functional analysis of 14 nucleotide changes in a prokaryotic expression system in order to elucidate their molecular pathogenesis. PMM2 activity assay revealed the presence of six protein changes with no enzymatic activities (p.R123Q, p.R141H, p.F157S, p.P184T, p.F207S and p.D209G) and seven mild protein changes with residual activities ranging from 16 to 54% (p.L32R, p.V44A p.D65Y, p.P113L p.T118S, p.T237M and p.C241S) and also one variant change with normal activity (p.E197A). The results obtained from Western blot analysis, degradation time courses of 11 protein changes and structural analysis of the PMM2 protein, suggest that the loss-of-function of most mutant proteins is based on their increased susceptibility to degradation or aggregation compared to the wild type protein, considering PMM2 deficiency as a conformational disease. We have identified exclusively catalytic protein change (p.D209G), catalytic protein changes affecting protein stability (p.R123Q and p.R141H), two protein changes disrupting the dimer interface (p.P113L and p.T118S) and several misfolding changes (p.L32R, p.V44A, p.D65Y, p.F157S, p.P184T, p.F207S, p.T237M and p.C241S). Our current work opens a promising therapeutic option using pharmacological chaperones to revert the effect of the characterized misfolding mutations identified in a wide range of PMM2 deficient patients.




Similar content being viewed by others
References
Bernier V, Lagace M, Bichet DG, Bouvier M (2004) Pharmacological chaperones: potential treatment for conformational diseases. Trends Endocrinol Metab 15:222–228
Briones P, Vilaseca MA, Schollen E, Ferrer I, Maties M, Busquets C, Artuch R, Gort L, Marco M, van Schaftingen E, Matthijs G, Jaeken J, Chabas A (2002) Biochemical and molecular studies in 26 Spanish patients with congenital disorder of glycosylation type Ia. J Inherit Metab Dis 25:635–646
Bross P, Corydon TJ, Andresen BS, Jorgensen MM, Bolund L, Gregersen N (1999) Protein misfolding and degradation in genetic diseases. Hum Mutat 14:186–198
Cohen FE, Kelly JW (2003) Therapeutic approaches to protein-misfolding diseases. Nature 426:905–909
Dipple KM, McCabe ER (2000) Phenotypes of patients with "simple" Mendelian disorders are complex traits: thresholds, modifiers, and systems dynamics. Am J Hum Genet 66:1729–1735
Eklund EA, Merbouh N, Ichikawa M, Nishikawa A, Clima JM, Dorman JA, Norberg T, Freeze HH (2005) Hydrophobic Man-1-P derivatives correct abnormal glycosylation in Type I congenital disorder of glycosylation fibroblasts. Glycobiology 15:1084–1093
Freeze HH (2009) Towards a therapy for phosphomannomutase 2 deficiency, the defect in CDG-Ia patients. Biochim Biophys Acta 1792:835–840
Gersting SW, Kemter KF, Staudigl M, Messing DD, Danecka MK, Lagler FB, Sommerhoff CP, Roscher AA, Muntau AC (2008) Loss of function in phenylketonuria is caused by impaired molecular motions and conformational instability. Am J Hum Genet 83:5–17
Grunewald S, Schollen E, Van Schaftingen E, Jaeken J, Matthijs G (2001) High residual activity of PMM2 in patients' fibroblasts: possible pitfall in the diagnosis of CDG-Ia (phosphomannomutase deficiency). Am J Hum Genet 68:347–354
Haeuptle MA, Hennet T (2009) Congenital disorders of glycosylation: an update on defects affecting the biosynthesis of dolichol-linked oligosaccharides. Hum Mutat 30:1628–1641
Hardre R, Khaled A, Willemetz A, Dupre T, Moore S, Gravier-Pelletier C, Le Merrer Y (2007) Mono, di and tri-mannopyranosyl phosphates as mannose-1-phosphate prodrugs for potential CDG-Ia therapy. Bioorg Med Chem Lett 17:152–155
Kjaergaard S, Skovby F, Schwartz M (1998) Absence of homozygosity for predominant mutations in PMM2 in Danish patients with carbohydrate-deficient glycoprotein syndrome type 1. Eur J Hum Genet 6:331–336
Kjaergaard S, Skovby F, Schwartz M (1999) Carbohydrate-deficient glycoprotein syndrome type 1A: expression and characterisation of wild type and mutant PMM2 in E. coli. Eur J Hum Genet 7:884–888
Le Bizec C, Vuillaumier-Barrot S, Barnier A, Dupre T, Durand G, Seta N (2005) A new insight into PMM2 mutations in the French population. Hum Mutat 25:504–505
Matthijs G (2000) Congenital disorders of glycosylation. Trends Biochem Sci 25:428
Matthijs G, Schollen E, Van Schaftingen E, Cassiman JJ, Jaeken J (1998) Lack of homozygotes for the most frequent disease allele in carbohydrate-deficient glycoprotein syndrome type 1A. Am J Hum Genet 62:542–550
McKinney JA, Turel B, Winge I, Knappskog PM, Haavik J (2009) Functional properties of missense variants of human tryptophan hydroxylase 2. Hum Mutat 30:787–794
Pey AL, Desviat LR, Gamez A, Ugarte M, Perez B (2003) Phenylketonuria: Genotype-phenotype correlations based on expression analysis of structural and functional mutations in PAH. Hum Mutat 21:370–378
Pey AL, Stricher F, Serrano L, Martinez A (2007) Predicted effects of missense mutations on native-state stability account for phenotypic outcome in phenylketonuria, a paradigm of misfolding diseases. Am J Hum Genet 81:1006–1024
Pey AL, Ying M, Cremades N, Velazquez-Campoy A, Scherer T, Thony B, Sancho J, Martinez A (2008) Identification of pharmacological chaperones as potential therapeutic agents to treat phenylketonuria. J Clin Invest 118:2858–2867
Pirard M, Matthijs G, Heykants L, Schollen E, Grunewald S, Jaeken J, van Schaftingen E (1999) Effect of mutations found in carbohydrate-deficient glycoprotein syndrome type IA on the activity of phosphomannomutase 2. FEBS Lett 452:319–322
Silvaggi NR, Zhang C, Lu Z, Dai J, Dunaway-Mariano D, Allen KN (2006) The X-ray crystal structures of human alpha-phosphomannomutase 1 reveal the structural basis of congenital disorder of glycosylation type 1a. J Biol Chem 281:14918–14926
Vega AI, Perez-Cerda C, Desviat LR, Matthijs G, Ugarte M, Perez B (2009) Functional analysis of three splicing mutations identified in the PMM2 gene: toward a new therapy for congenital disorder of glycosylation type Ia. Hum Mutat 30:795–803
Vuillaumier-Barrot S, Hetet G, Barnier A, Dupre T, Cuer M, de Lonlay P, Cormier-Daire V, Durand G, Grandchamp B, Seta N (2000) Identification of four novel PMM2 mutations in congenital disorders of glycosylation (CDG) Ia French patients. J Med Genet 37:579–580
Westphal V, Peterson S, Patterson M, Tournay A, Blumenthal A, Treacy EP, Freeze HH (2001) Functional significance of PMM2 mutations in mildly affected patients with congenital disorders of glycosylation Ia. Genet Med 3:393–398
Acknowledgements
This work was supported by grants from Fondo de Investigaciones Sanitarias (PI070118 and PI10/00455) and from Fundación Ramón Areces. An institutional grant from the Fundación Ramón Areces to the Centro de Biología Molecular Severo Ochoa is also gratefully acknowledged. AG is supported by the National “Ramón y Cajal” program from MICINN.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by: Jaak Jaeken
Competing interest. None declared.
Rights and permissions
About this article
Cite this article
Vega, A.I., Pérez-Cerdá, C., Abia, D. et al. Expression analysis revealing destabilizing mutations in phosphomannomutase 2 deficiency (PMM2-CDG). J Inherit Metab Dis 34, 929–939 (2011). https://doi.org/10.1007/s10545-011-9328-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10545-011-9328-2