Summary
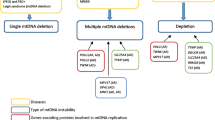
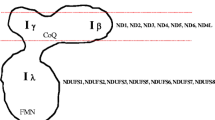
Mitochondria are the main energy-producing organelles of the cell. Five complexes embedded in the mitochondrial inner membrane, together constituting the oxidative phosphorylation (OXPHOS) system, comprise the final steps in cellular energy production. Many patients with a mitochondrial defect suffer from a so-called combined deficiency, meaning that the enzymatic activities of two or more complexes of the OXPHOS system are decreased. Numerous mutations have been described in nuclear genes that are involved in the functioning of a single complex of the OXPHOS system. However, little attention has been paid to patients with a deficiency of more than one complex of this particular system. In this study we have investigated four nuclear genes (OXA1L, MRS2L, YME1L and MIPEP) that might be involved in the pathology of combined enzymatic deficiencies of the OXPHOS system. Based on the results of yeast knockouts of these four proteins, we have sequenced the open reading frame of OXA1L in eight patients with an enzymatic deficiency of complexes I and IV. MRS2L, YME1L and MIPEP have been sequenced in three patients with a combined defect of complexes III and IV. No mutations were detected in these patients, showing that at least in these patients the OXPHOS system deficiency cannot be explained by a mutation in these four genes.
Similar content being viewed by others
References
Antonicka H, Mattman A, Carlson CG, et al (2003) Mutations in COX15 produce a defect in the mitochondrial heme biosynthetic pathway, causing early-onset fatal hypertrophic cardiomyopathy. Am J Hum Genet 72: 101–114.
Bentlage HA, Wendel U, Schagger H, et al (1996) Lethal infantile mitochondrial disease with isolated complex I deficiency in fibroblasts but with combined complexes I and IV deficiencies in muscle. Neurology 47: 243–248.
Bonnefoy N, Kermorgant M, Groudinsky O, et al (1994) Cloning of a human gene involved in cytochrome oxidase assembly by functional complementation of an oxa1-mutation in Saccharomyces cerevisiae. Proc Natl Acad Sci USA 91: 11978–11982.
Borisov VB (2002) Defects in mitochondrial respiratory complexes III and IV, and human pathologies. Mol Aspects Med 23: 385–412.
Bui DM, Gregan J, Jarosch E, et al (1999) The bacterial magnesium transporter CorA can functionally substitute for its putative homologue Mrs2p in the yeast inner mitochondrial membrane. J Biol Chem 274: 20438–20443.
Chew A, Buck EA, Peretz S, et al (1997) Cloning, expression, and chromosomal assignment of the human mitochondrial intermediate peptidase gene (MIPEP). Genomics 40: 493–496.
Coenen MJ, Antonicka H, Ugalde C, et al (2004) Mutant mitochondrial elongation factor G1 and combined oxidative phosphorylation deficiency. N Engl J Med 351: 2080–2086.
DiMauro S, Andreu AL (2000) Mutations in mtDNA: are we scraping the bottom of the barrel? Brain Pathol 10: 431–441.
He S, Fox TD (1997) Membrane translocation of mitochondrially coded Cox2p: distinct requirements for export of N and C termini and dependence on the conserved protein Oxa1 p. Mol Biol Cell 8: 1449–1460.
Hell K, Herrmann J, Pratje E, et al (1997) Oxa1p mediates the export of the N- and C-termini of pCoxII from the mitochondrial matrix to the intermembrane space. FEBS Lett 418: 367–370.
Isaya G, Kalousek F, Rosenberg LE (1992) Amino-terminal octapeptides function as recognition signals for the mitochondrial intermediate peptidase. J Biol Chem 267: 7904–7910.
Isaya G, Miklos D, Rollins RA (1994) MIP1, a new yeast gene homologous to the rat mitochondrial intermediate peptidase gene, is required for oxidative metabolism in Saccharomyces cerevisiae. Mol Cell Biol 14: 5603–5616.
Isaya G, Sakati WR, Rollins RA, et al (1995) Mammalian mitochondrial intermediate peptidase: structure/function analysis of a new homologue from Schizophyllum commune and relationship to thimet oligopeptidases. Genomics 28: 450–461.
Klanner C, Prokisch H, Langer T (2001) MAP-1 and IAP-1, two novel AAA proteases with catalytic sites on opposite membrane surfaces in mitochondrial inner membrane of Neurospora crassa. Mol Biol Cell 12: 2858–2869.
Kolisek M, Zsurka G, Samaj J, et al (2003) Mrs2p is an essential component of the major electrophoretic Mg2+ influx system in mitochondria. EMBO J 22: 1235–1244.
Leigh D (1951) Subacute necrotizing encephalomyelopathy in an infant. J Neurol Neurosurg Psychiatry 14: 216–221.
McEwen JE, Ko C, Kloeckner-Gruissem B, Poyton, RO (1986) Nuclear functions required for cytochrome c oxidase biogenesis in Saccharomyces cerevisiae. Characterization of mutants in 34 complementation groups. J Biol Chem 261: 11872–11879.
Mootha VK, Lepage P, Miller K, et al (2003) Identification of a gene causing human cytochrome c oxidase deficiency by integrative genomics. Proc Natl Acad Sci USA 100: 605–610.
Nakai T, Yasuhara T, Fujiki Y, Ohashi A (1995) Multiple genes, including a member of the AAA family, are essential for degradation of unassembled subunit 2 of cytochrome c oxidase in yeast mitochondria. Mol Cell Biol 15: 4441–4452.
Nargang FE, Preuss M, Neupert W, Herrmann JM (2002) The Oxa1 protein forms a homooligomeric complex and is an essential part of the mitochondrial export translocase in Neurospora crassa. J Biol Chem 277: 12846–12853.
Pearce DA, Sherman F (1995) Degradation of cytochrome oxidase subunits in mutants of yeast lacking cytochrome c and suppression of the degradation by mutation of yme1. J Biol Chem 270: 20879–20882.
Rahman S, Blok RB, Dahl HH, et al (1996) Leigh syndrome: clinical features and biochemical and DNA abnormalities. Ann Neurol 39: 343–351.
Rotig A, Parfait B, Heidet L, et al (1997) Sequence and structure of the human OXA1L gene and its upstream elements. Biochim Biophys Acta 1361: 6–10.
Seyda A, Newbold RF, Hudson TJ, et al (2001) A novel syndrome affecting multiple mitochondrial functions, located by microcell-mediated transfer to chromosome 2p14-2p13. Am J Hum Genet 68: 386–396.
Shah ZH, Hakkaart GA, Arku B, et al (2000) The human homologue of the yeast mitochondrial AAA metalloprotease Yme1p complements a yeast yme1 disruptant. FEBS Lett 478: 267–270.
Smith RL, Maguire ME (1998) Microbial magnesium transport: unusual transporters searching for identity. Mol Microbiol 28: 217–226.
Sue CM, Schon EA (2001) Mitochondrial respiratory chain diseases and mutations in nuclear DNA: a promising start? Brain Pathol 10: 442–450.
Weber ER, Hanekamp T, Thorsness PE (1996) Biochemical and functional analysis of the YME1 gene product, an ATP and zinc-dependent mitochondrial protease from S. cerevisiae. Mol Biol Cell 7: 307–317.
Yano T (2002) The energy-transducing NADH:quinone oxidoreductase, complex I. Mol Aspects Med 23: 345–368.
Zhu Z, Yao J, Johns T, et al (1998) SURF1, encoding a factor involved in the biogenesis of cytochrome c oxidase, is mutated in Leigh syndrome. Nature Genetics 20: 337–343.
Zsurka G, Gregan J, Schweyen RJ (2001) The human mitochondrial mrs2 protein functionally substitutes for its yeast homologue, a candidate magnesium transporter. Genomics 72: 158–168.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Coenen, M.J.H., Smeitink, J.A.M., Smeets, R. et al. Mutation detection in four candidate genes (OXA1L, MRS2L, YME1L and MIPEP) for combined deficiencies in the oxidative phosphorylation system. J Inherit Metab Dis 28, 1091–1097 (2005). https://doi.org/10.1007/s10545-005-4483-y
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s10545-005-4483-y