Abstract
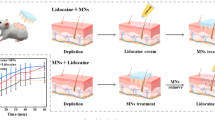
This study showed that drug-coated PLLA (Poly (L-lactide)) microneedle arrays can induce rapid and painless local anesthesia. Microneedle arrays were fabricated using a micro-molding technique, and the needle tips were coated with 290.6 ± 45.9 μg of lidocaine, the most widely used local anesthetic worldwide. A dip-coating device was newly designed for the coating step using an optimized coating formulation. Lidocaine coated on the arrays was released rapidly into PBS within 2 min, and its stability in storage lasted 3 weeks at 4, 25, and 37°C. Furthermore, the microneedle arrays showed consistent in vitro skin penetration and delivered 200.8 ± 43.9, 224.2 ± 39.3, and 244.1 ± 19.6 μg of lidocaine into the skin 1, 2, and 5 min after application with a high delivery efficiency of 69, 77, and 84%. Compared to a commercially available topical anesthetic EMLA® cream, a 22.0, 13.6, and 14.0-fold higher amount of lidocaine was delivered into the skin. Note, in vitro skin permeation of Lidocaine was also notably enhanced by a 2-min-application of the lidocaine-coated microneedle arrays. Altogether, these results suggest that the biocompatible lidocaine-coated PLLA microneedle arrays could provide significantly rapid local anesthesia in a painless manner without any of the issues from topical applications or hypodermic injections of local anesthetics.





Similar content being viewed by others
References
T. Alster, Review of lidocaine/tetracaine cream as a topical anesthetic for dermatologic laser procedures. Pain and therapy 2(1), 11–19 (2013)
S. Berkman, J. MacGregor, T. Alster, Adverse effects of topical anesthetics for dermatologic procedures. Expert Opin. Drug Saf. 11(3), 415–423 (2012)
X. Chen et al., Site-selectively coated, densely-packed Microprojection Array patches for targeted delivery of vaccines to skin. Adv. Funct. Mater. 21(3), 464–473 (2011)
J. Cooper et al., Evaluation of a needle-free injection system for local anaesthesia prior to venous cannulation. Anaesthesia 55(3), 247–250 (2000)
A. Dhawan et al., Complications of bioabsorbable suture anchors in the shoulder. Am. J. Sports Med. 40(6), 1424–1430 (2012)
M.G. Gebauer, A.F. McClure, T.L. Vlahakis, Stability indicating HPLC method for the estimation of oxycodone and lidocaine in rectal gel. Int. J. Pharm. 223(1), 49–54 (2001)
H.S. Gill, M.R. Prausnitz, Coated microneedles for transdermal delivery. J. Control. Release 117(2), 227–237 (2007a)
H.S. Gill, M.R. Prausnitz, Coating formulations for microneedles. Pharm. Res. 24(7), 1369–1380 (2007b)
J. Gupta et al., Rapid local anesthesia in human subjects using minimally invasive microneedles. Clin. J. Pain 28(2), 129 (2012)
C.S. Houck, N.F. Sethna, Transdermal analgesia with local anesthetics in children: review, update and future directions. Expert. Rev. Neurother. 5(5), 625–634 (2005)
Y. Ito et al., Feasibility of microneedles for percutaneous absorption of insulin. Eur. J. Pharm. Sci. 29(1), 82–88 (2006)
Y. Ito et al., Dissolving microneedles to obtain rapid local anesthetic effect of lidocaine at skin tissue. J. Drug Target. 21(8), 770–775 (2013)
U. Jacobi et al., Porcine ear skin: an in vitro model for human skin. Skin Res. Technol. 13(1), 19–24 (2007)
N.P. Katz et al., Rapid onset of cutaneous anesthesia with EMLA cream after pretreatment with a new ultrasound-emitting device. Anesth. Analg. 98(2), 371–376 (2004)
T.-Y. Kim et al., Anesthetic effects of lidocaine hydrochloride gel using low frequency ultrasound of 0.5 MHz. J Pharm Pharm Sci 10(1), 1–8 (2007)
Y.-C. Kim, J.-H. Park, M.R. Prausnitz, Microneedles for drug and vaccine delivery. Adv. Drug Deliv. Rev. 64(14), 1547–1568 (2012)
D.J. Kouba et al., Guidelines for the use of local anesthesia in office-based dermatologic surgery. J. Am. Acad. Dermatol. 74(6), 1201–1219 (2016)
J.W. Lee, J.-H. Park, M.R. Prausnitz, Dissolving microneedles for transdermal drug delivery. Biomaterials 29(13), 2113–2124 (2008)
X. Li et al., Microneedle pretreatment improves efficacy of cutaneous topical anesthesia. Am. J. Emerg. Med. 28(2), 130–134 (2010)
C. Liossi, Management of paediatric procedure-related cancer pain. Pain Reviews 6(4), 279–302 (1999)
C. Lysakowski et al., A needle-free jet-injection system with lidocaine for peripheral intravenous cannula insertion: a randomized controlled trial with cost-effectiveness analysis. Anesth. Analg. 96(1), 215–219 (2003)
Y. Ma, H.S. Gill, Coating solid dispersions on microneedles via a molten dip-coating method: development and in vitro evaluation for transdermal delivery of a water-insoluble drug. J. Pharm. Sci. 103(11), 3621–3630 (2014)
Malamed, S.F., Handbook of local anesthesia. (Elsevier Health Sciences, 2014)
L. Misery, Skin, immunity and the nervous system. Br. J. Dermatol. 137(6), 843–850 (1997)
S.N. Murthy, S.M. Sammeta, C. Bowers, Magnetophoresis for enhancing transdermal drug delivery: mechanistic studies and patch design. J. Control. Release 148(2), 197–203 (2010)
S. Mutalik, How to make local anesthesia less painful. Journal of cutaneous and aesthetic surgery 1(1), 37 (2008)
K. Na et al., Biodegradable thermo-sensitive nanoparticles from poly (L-lactic acid)/poly (ethylene glycol) alternating multi-block copolymer for potential anti-cancer drug carrier. Eur. J. Pharm. Sci. 27(2), 115–122 (2006)
A. Nayak et al., Lidocaine carboxymethylcellulose with gelatine co-polymer hydrogel delivery by combined microneedle and ultrasound. Drug delivery 23(2), 668–679 (2016)
J.-H. Park, M.G. Allen, M.R. Prausnitz, Biodegradable polymer microneedles: fabrication, mechanics and transdermal drug delivery. J. Control. Release 104(1), 51–66 (2005)
J.-H. Park, M.G. Allen, M.R. Prausnitz, Polymer microneedles for controlled-release drug delivery. Pharm. Res. 23(5), 1008–1019 (2006)
M.R. Prausnitz, Microneedles for transdermal drug delivery. Adv. Drug Deliv. Rev. 56(5), 581–587 (2004)
S. Saleem et al., Improvement of hand sensibility resulting from application of anaesthetic cream on the forearm: importance of dose and time. Hand Therapy 20(4), 109–114 (2015)
R.J. Scarfone, M. Jasani, E.J. Gracely, Pain of local anesthetics: rate of administration and buffering. Ann. Emerg. Med. 31(1), 36–40 (1998)
D.W. Smith, M.R. Peterson, S.C. DeBerard, Local anesthesia: topical application, local infiltration, and field block. Postgrad. Med. 106(2), 58–66 (1999)
S.P. Sullivan, N. Murthy, M.R. Prausnitz, Minimally invasive protein delivery with rapidly dissolving polymer microneedles. Adv. Mater. 20(5), 933–938 (2008)
C.-F. Wahlgren, H. Quiding, Depth of cutaneous analgesia after application of a eutectic mixture of the local anesthetics lidocaine and prilocaine (EMLA cream). J. Am. Acad. Dermatol. 42(4), 584–588 (2000)
M.S. Wallace et al., Topical delivery of lidocaine in healthy volunteers by electroporation, electroincorporation, or iontophoresis: an evaluation of skin anesthesia. Reg. Anesth. Pain Med. 26(3), 229–238 (2001)
Y. Wang et al., Preparation and evaluation of lidocaine hydrochloride-loaded TAT-conjugated polymeric liposomes for transdermal delivery. Int. J. Pharm. 441(1), 748–756 (2013)
D. Wu et al., Improvement of transdermal delivery of sumatriptan succinate using a novel self-dissolving microneedle array fabricated from sodium hyaluronate in rats. Biol. Pharm. Bull. 38(3), 365–373 (2015)
W.T. Zempsky et al., Evaluation of a low-dose lidocaine lontophoresis system for topical anesthesia in adults and children: a randomized, controlled trial. Clin. Ther. 26(7), 1110–1119 (2004)
W.T. Zempsky, B. Robbins, K. McKay, Reduction of topical anesthetic onset time using ultrasound: a randomized controlled trial prior to venipuncture in young children. Pain Med. 9(7), 795–802 (2008)
Y. Zhang et al., Development of lidocaine-coated microneedle product for rapid, safe, and prolonged local analgesic action. Pharm. Res. 29(1), 170–177 (2012a)
Y. Zhang et al., Adjuvants to prolong the local anesthetic effects of coated microneedle products. Int. J. Pharm. 439(1), 187–192 (2012b)
Z. Zhu et al., Rapidly dissolvable microneedle patches for transdermal delivery of exenatide. Pharm. Res. 31(12), 3348–3360 (2014)
Acknowledgments
This work was supported financially by Ministry of Science, ICT, and Future Planning (Project No. NRF-2014M3A9E4064580) and grants of the Advanced Biomass R & D Center (ABC) of the Global Frontier Project funded by the Ministry of Science, ICT, and Future Planning (ABC-2011-0031350).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of this article.
Electronic supplementary material
Supplementary Figure 1
Schematic illustration of dip-coating device for 2D PLLA microneedle array. Pristine PLLA microneedle array attached on the plate which the array was hold on. As watched by microscope, the plate with the array was moved down and up to the reservoir in which coating solution was contained for dip-coating with lidocaine on the array. (DOCX 161 kb)
Supplementary Figure 2
The images of PLLA microneeldle array before dip-coating (a) and after dip-coating (b) by newly developed dip-coating device. The array was fictured and observed by USB microscopy (DOCX 794 kb)
Rights and permissions
About this article
Cite this article
Baek, SH., Shin, JH. & Kim, YC. Drug-coated microneedles for rapid and painless local anesthesia. Biomed Microdevices 19, 2 (2017). https://doi.org/10.1007/s10544-016-0144-1
Published:
DOI: https://doi.org/10.1007/s10544-016-0144-1