Abstract
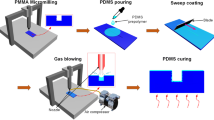
Current methods for formation of microvascular channel scaffolds are limited with non-circular channel cross-sections, complicated fabrication, and less flexibility in microchannel network design. To address current limitations in the creation of engineered microvascular channels with complex three-dimensional (3-D) geometries in the shape of microvessels, we have developed a reproducible, cost-effective, and flexible micromanufacturing process combined with photolithographic reflowable photoresist and soft lithography techniques to fabricate cylindrical microchannel and networks. A positive reflowable photoresist AZ P4620 was used to fabricate a master microchannel mold with semi-circular cross-sections. By the alignment and bonding of two polydimethylsiloxane (PDMS) microchannels replicated from the master mold together, a cylindrical microchannel or microchannel network was created. Further examination of the channel dimensions and surface profiles at different branching levels showed that the shape of the microfluidic channel was well approximated by a semi-circular surface, and a multi-level, multi-depth channel network was created. In addition, a computational fluidic dynamics (CFD) model was used to simulate shear flows and corresponding pressure distributions inside of the microchannel and channel network based on the dimensions of the fabricated channels. The fabricated multi-depth cylindrical microchannel network can provide platforms for the investigation of microvascular cells growing inside of cylindrical channels under shear flows and lumen pressures, and work as scaffolds for the investigation of morphogenesis and tubulogenesis.









Similar content being viewed by others
References
M. Abdelgawad, C. Wu, W.-Y. Chien, W.R. Geddie, M.A.S. Jewett, Y. Sun, Lab Chip 11, 545 (2011)
P. Abgrall, A.M. Gue, J. Micromech. Microeng. R15, 17 (2007)
A. Agarwal, N. Ranganathan, W.L. Ong, K.C. Tang, L. Yobas, Sens. Actuators A 142, 80 (2008)
Application notes from MicroChemicals: Reflow of Photoresist, http://www.microchemicals.eu/technical_information
H. Becker, C. Gartner, Anal. Bioanal. Chem. 390, 89 (2008)
L.M. Bellan, S.P. Singh, P.W. Henderson, T.J. Porri, H.G. Craighead, J.A. Spector, Soft Matter 5, 1354 (2009)
J.T. Borenstein, M.M. Tupper, P.J. Mack, E.J. Weinberg, A.S. Khalil, J. Hsiao, G. García-Cardeña, Biomed. Microdevices 12, 71 (2010)
J.P. Camp, T. Stokol, M.L. Shuler, Biomed. Microdevices 10, 179 (2008)
Y.-C. Chen, G.-Y. Chen, Y.-C. Lin, G.-J. Wang, Microfluid. Nanofluid. 9, 585 (2010)
J.A. Chen, Y. Zheng, Q. Tan, Y.L. Zhang, J. Li, W.R. Geddie, M.A.S. Jewett, Y. Sun, Biomicrofluidics 5, 014113 (2011)
K.M. Chrobak, D.R. Potter, J. Tien, Microvasc. Res. 71, 185–196 (2006)
O.C. Colgan, G. Ferguson, N.T. Collins, R.P. Murphy, G. Meade, P.A. Cahill, P.M. Cummins, Am. J. Physiol. Heart Circ. Physiol. 292, H3190 (2007)
C. Couzon, A. Duperray, C. Verdier, Eur. Biophys. J. 38, 1035 (2009)
A. Crespi, Y. Gu, B. Ngamson, H.J.W.M. Hoekstra, C. Dongre, M. Pollnau, R. Ramponi, H.H. van den Vlekkert, P. Watts, G. Cerullo, R. Osellame, Lab Chip 10, 1167 (2010)
D. Daly, R.F. Stevens, M.C. Hutley, N. Davies, Meas. Sci. Technol. 1, 759 (1990)
P.F. Davies, Physiol. Rev. 75, 519–560 (1995)
M.J. de Boer, R.W. Tjerkstra, J.W. Berenschot, H.V. Jansen, G.J. Burger, J.G.E. Gardeniers, M. Elwenspoek, A. van den Berg, J. Microelectromech. Syst. 9, 94 (2000)
de Gennes, Rev. Mod. Phys. 57, 827–863 (1985)
D.C. Duffy, J.C. McDonald, O.J.A. Schueller, G.M. Whitesides, Anal. Chem. 70, 4974 (1998)
H. G. Elias, VCH Publishers, New York (1997)
D.R. Emerson, K. Cieslicki, X. Gu, R.W. Barber, Lab Chip 6, 447 (2006)
L.K. Fiddes, N. Raz, S. Srigunapalan, Biomaterials 31, 3459 (2010)
A.B. Fisher, S. Chien, A.I. Barakat, R.M. Nerem, Am. J. Physiol. Lung Cell. Mol. Physiol. 281(3), L529 (2001)
Y. C. Fung, New York, NY: Springer; (1997)
V.V. Gafiychuk, I.A. Lubashevsky, J. Theor. Biol. 212, 1 (2001)
A. Gnasso, C. Carallo, C. Irace, V. Spagnuolo, G. De Novara, P.L. Mattioli, A. Pujia, Circulation 94, 3257–3262 (1996)
T.R. Jay, M.B. Stern, Opt. Eng. 33, 3552–3555 (1994)
T. Kadohama, N. Akasaka, K. Nishimura, Y. Hoshino, T. Sasajima, B.E. Sumpio, Endothelium 13, 43 (2006)
S. Kaihara, J. Borenstein, R. Koka, S. Lalan, E.R. Ochoa, M. Ravens, H. Pien, B. Cunningham, J.P. Vacanti, Tissue Eng. 6, 105 (2000)
A. Kamiya, R. Bukhari, T. Togawa, Bull. Math. Biol. 46, 127–137 (1984)
J. Koskela, Master's thesis, Tampereen teknillinen yliopisto. (2010)
M. LaBarbera, Science 249, 992–1000 (1990)
T.G. Leong, A.M. Zarafshar, D.H. Gracias, Small 6, 792 (2010)
D. Lim, Y. Kamotani, B. Cho, J. Mazumder, S. Takayama, Lab Chip 3, 318 (2003)
R.H. Liu, M.A. Stremler, K.V. Sharp, M.G. Olsen, J.G. Santiago, R.J. Adrian, H. Aref, D.J. Beebe, J. Microelectromech. Syst. 9, 190 (2000)
H. Lu, L.Y. Koo, W.M. Wang, D.A. Lauffenburger, L.G. Griffith, K.F. Jensen, Anal. Chem. 76, 5257 (2004)
A.M. Malek, S.L. Alper, S. Izumo, JAMA 282(21), 2035–2042 (1999)
V. Maselli, R. Osellame, G. Cerullo, R. Ramponi, P. Laporta, L. Magagnin, P.L. Cavallotti, Appl. Phys. Lett. 88, 191107 (2006)
J.A. McCann, S.D. Peterson, M.W. Plesniak, T.J. Webster, K.M. Haberstroh, Ann. Biomed. Eng. 33, 328 (2005)
A. Meeson, M. Palmer, M. Calfon, R. Lang, Development 122, 3929 (1996)
C.D. Murray, Proc Natl Acad Sci USA 12, 207 (1926a)
C.D. Murray, J. Gen. Physiol. 9, 835 (1926b)
R.M. Nerem, R.W. Alexander, D.C. Chappell, R.M. Medford, S.E. Varner, W.R. Taylor, Am. J. Med. Sci. 316(3), 169 (1998)
F.T. O’Neill, J.T. Sheridan, Optik 113, 391 (2002)
C.M. Potter, M.H. Lundberg, L.S. Harrington, C.M. Warboys, T.D. Warner, R.E. Berson, A.V. Moshkov, J. Gorelik, P.D. Weinberg, J.A. Mitchell, Arterioscler. Thromb. Vasc. Biol. 31, 384 (2011)
G.M. Riha, P.H. Lin, A.B. Lumsden, Q. Yao, C. Chen, Ann. Biomed. Eng. 33, 772 (2005)
I. Rodriguez, P. Spicar-Mihalic, C.L. Kuyper, G.S. Fiorini, D.T. Chiu, Anal. Chim. Acta 496, 205 (2003)
W. Schaper, Circulation 104, 1994 (2001)
A. Schilling, R. Merz, C. Ossmann, H.P. Herzig, Opt. Eng. 39, 2171–2176 (2000)
K. Sekimoto, R. Oguma, K. Kawasaki, Ann. Phys. 176, 359–392 (1987)
C.T. Seo, C.H. Bae, D.S. Eun, J.K. Shin, J.H. Lee, Jpn. J. Appl. Phys. 43, 7773 (2004)
J. Shao, L. Wu, J. Wu, Y. Zheng, H. Zhao, Q. Jin, J. Zhao, Lab Chip 9, 3118 (2009)
T.F. Sherman, J. Gen. Physiol. 78, a 431 (1981)
S.S. Shevkoplyas, S.C. Gifford, T. Yoshida, M.W. Bitensky, Microvasc. Res. 65, 132 (2003)
S.H. Song, C.K. Lee, T.J. Kim, I.C. Shin, S.C. Jun, H.I. Jung, Microfluid. Nanofluid. 9, 533 (2010)
A.F. Stalder, Z. Liu, J. Hennig, J.G. Korvink, K.C. Li, and M. Markl, Part 1, 27-38, Springer Science (2011)
J. Surapisitchat, R.J. Hoefen, X. Pi, M. Yoshizumi, C. Yan, B.C. Berk, Proc. Natl. Acad. Sci. U. S. A. 98, 6476 (2001)
J.M. Tarbell, Cardiovasc. Res. 87, 320 (2010)
N. Van Royen, J.J. Piek, W. Schaper, C. Bode, I. Buschmann, J. Nucl. Cardiol. 8, 687 (2001)
O.V. Voinov, Jour. Appl. Mech. Tech. Phys. 40, 86–92 (1999)
G.-J. Wang, K.-H. Ho, S.-H. Hsu, K.-P. Wang, Biomed. Microdevices 9, 657 (2007)
E. Warabi, Y. Wada, H. Kajiwara, M. Kobayashi, N. Koshiba, T. Hisada, M. Shibata, J. Ando, M. Tsuchiya, T. Kodama, N. Noguchi, Free Radic. Biol. Med. 37, 682 (2004)
G.M. Whitesides, E. Ostuni, S. Takayama, X. Jiang, D.E. Ingber, Annu. Rev. Biomed. Eng. 3, 335 (2001)
M.E. Wilson, N. Kota, Y.T. Kim, Y. Wang, D.B. Stolz, P.R. LeDuc, O.B. Ozdoganlar, Lab Chip 11, 1550 (2011)
C.J. World, G. Garin, B. Berk, Curr. Atheroscler. Rep. 8, 240 (2006)
Y. Xia, G.M. Whiteside, Annu. Rev. Mater. Sci. 28, 153 (1998)
B. Young, J. W. Heath, Wheater’s functional histology: A Text and Colour Atlas, 4th edn. (Churchill livingstone, 2000)
M. Zamir, J.A. Medeiros, J. Gen. Physiol. 79, 353 (1982)
Y. Zeng, T.-S. Lee, P. Yu, P. Roy, H.-T. Low, J. Biomech. Eng. 128, 185 (2006)
Acknowledgments
Xiang Li and Zhouchun Huang are co-first authors. We thank Mr. Michael Martin for proofreading and editing the paper. This research work was supported by WVU EPSCoR program funded by the National Science Foundation (EPS-1003907). Partial support for this work was provided by the National Science Foundation's ADVANCE IT Program under Award HRD-1007978. Any opinions, findings, and conclusions or recommendations expressed in this material are those of the author(s) and do not necessarily reflect the views of the National Science Foundation. The microfabrication work was done in WVU Shared Research Facilities (Cleanroom facilities) and Microfluidic Integrative Cellular Research on Chip Laboratory (MICRoChip Lab) at West Virginia University.
Author information
Authors and Affiliations
Corresponding author
Additional information
Zhouchun Huang and Xiang Li contributed equally.
Rights and permissions
About this article
Cite this article
Huang, Z., Li, X., Martins-Green, M. et al. Microfabrication of cylindrical microfluidic channel networks for microvascular research. Biomed Microdevices 14, 873–883 (2012). https://doi.org/10.1007/s10544-012-9667-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10544-012-9667-2