Abstract
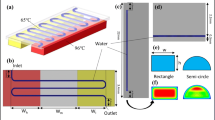
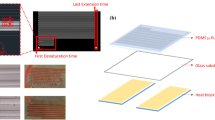
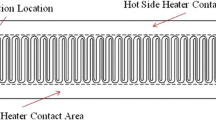
A new micromachined circulating polymerase chain reaction (PCR) chip is reported in this study. A novel liquid transportation mechanism utilizing a suction-type membrane and three microvalves were used to create a new microfluidic control module to rapidly transport the DNA samples and PCR reagents around three bio-reactors operating at three different temperatures. When operating at a membrane actuation frequency of 14.29 Hz and a pressure of 5 psi, the sample flow rate in the microfluidic control module can be as high as 18 μL/s. In addition, an array-type microheater was adopted to improve the temperature uniformity in the reaction chambers. Open-type reaction chambers were designed to facilitate temperature calibration. Experimental data from infrared images showed that the percentage of area inside the reaction chamber with a thermal variation of less than 1°C was over 90% for a denaturing temperature of 94°C. Three array-type heaters and temperature sensors were integrated into this new circulating PCR chip to modulate three specific operating temperatures for the denaturing, annealing, and extension steps of a PCR process. With this approach, the cycle numbers and reaction times of the three separate reaction steps can be individually adjusted. To verify the performance of this circulating PCR chip, a PCR process to amplify a detection gene (150 base pairs) associated with the hepatitis C virus was performed. Experimental results showed that DNA samples with concentrations ranging from 105 to 102copies/μL can be successfully amplified. Therefore, this new circulating PCR chip may provide a useful platform for genetic identification and molecular diagnosis.






Similar content being viewed by others
Abbreviations
- 2D:
-
Two-dimensional
- Au:
-
Gold Bio-MEMS Bio-micro-electro-mechanical-systems
- bp:
-
Base pair
- HCV:
-
Hepatitis C virus
- DNA:
-
Deoxyribonucleic acid
- EMV:
-
Electromagnetic valve
- IR:
-
Infrared
- PCR:
-
Polymerase chain reaction
- PDMS:
-
Polydimethylsiloxane
- Pt:
-
Platinum
- SEM:
-
Scanning electron microscope
- Ti:
-
Titanium
- UV:
-
Ultra-violet
References
R.C. Anderson, X. Su, G.J. Bogdan, J. Fenton, Nucleic Acids Res. 28(12) (2000). doi:10.1093/nar/28.12.e60
P. Belgrader, C.J. Elkin, S.B. Brown, S.N. Nasarabadi, R.G. Langlois, F.P. Milanovich et al., Anal. Chem. 75(14), 3446–3450 (2003). doi:10.1021/ac034062u
Z. Chen, S. Qian, W.R. Abrams, D. Malamud, H.H. Bau, Anal. Chem. 76(13), 3707–3715 (2004). doi:10.1021/ac049914k
J.H. Chien, D.S. Lee, Y.T. Cheng, S.H. Yeh, W.P. Chou, P.H. Chen, Opt. Commun. 266, 744–750 (2006). doi:10.1016/j.optcom.2006.05.043
T. Fukuba, T. Naganuma, T. Fuiji, Proceedings of the 2002 International Symposium, 101–105 (2004)
A. Gulliksen, L. Solli, F. Karlsen, H. Rogne, E. Hovig, T. Nordstrøm, R. Sirevåg, Anal. Chem. 76, 9–14 (2004). doi:10.1021/ac034779h
M. Hashimoto, P.C. Chen, M.W. Mitchell, D.E. Nikitopoulos, S.A. Soper, M.C. Murphy, Lab Chip 4(6), 638–345 (2004). doi:10.1039/b406860b
T.M. Hsieh, C.H. Luo, F.C. Huang, J.H. Wang, L.J. Chien, G.B. Lee, Sensors Actuators B. 130, 848–856 (2008)
F.C. Huang, C.S. Liao, G.B. Lee, Electrophoresis 27(16), 3297–3305 (2006). doi:10.1002/elps.200600458
C.W. Huang, S.B. Huang, G.B. Lee, J. Micromech. Microeng. 16, 2265–2272 (2006). doi:10.1088/0960-1317/16/11/003
M. Koch, N. Harris, A. Evans, N. White, A. Brunnschweiler, Sens. Actuators A. 70, 98–103 (1998). doi:10.1016/S0924-4247(98)00120-4
M.U. Kopp, A.J. de Mello, A. Manz, Science 280(5366), 1046–1048 (1998). doi:10.1126/science.280.5366.1046
M. Krishnan, V.M. Ugaz, M.A. Burn, Science 298(5594), 793 (2002). doi:10.1126/science.298.5594.793
D.J. Laser, J.G. Santiago, J. Micromech. Microeng. 14, R35–R64 (2004)
C.S. Liao, G.B. Lee, J.J. Wu, C.C. Chang, T.M. Hsieh, C.H. Luo, Biosens. Bioelectron. 20, 1341–1348 (2004). doi:10.1016/j.bios.2004.05.006
A. Manz, D.J. Harrison, E.M.J. Verpoorte, J.C. Fettinger, A. Paulus, H. Lüdi, H.M. Widmer, J. Chromatography. 593(1–2), 253–258 (1992)
K.B. Mullis, Sci Am. 262(4), 56–65 (1990)
K.B. Mullis, F. Faloona, S. Scharf, R. Saiki, G. Horn, H. Erlich, Cold Spring Harbor Symp. Quant. Biol. 51, 263–273 (1986)
H. Nakano, K. Matsuda, M. Yohda, T. Nagamune, I. Endo, T. Yamane, Biosci. Biotechnol. Biochem. 58(2), 349–352 (1994)
M.A. Northrup, M.T. Ching, R.M. White, R.T. Wltson, Transducer, Chicago, USA, 924–926 (1993)
M.A. Northrup, C. Gonzalez, D. Hadley, R.F. Hills, P. Landre, S. Lehew, R. Saiki, J.J. Shinsky, R. Watson, R. Watson Jr., Proceeding Transducers, 764–767 (1995)
S. Poser, T. Schulz, U. Dillner, V. Baier, J.M. Kőhler, D. Schimkat, Sens. Actuators A. 62, 672–675 (1997)
R.K. Saiki, S. Scharf, F. Faloona, K.B. Mullis, G.T. Horn, H.A. Erlich, N. Arnheim, Science. 230(4732), 1350–1354 (1985)
K. Sun, A. Yamaguchi, Y. Ishida, S. Matsuo, H. Misawa, Sens. Actuators B. 84, 283–289 (2002)
M.A. Unger, H.P. Chou, T. Thorsen, A. Scherer, R.Q. Stephen, Science 288(5463), 113–116 (2000). doi:10.1126/science.288.5463.113
C.H. Wang, Y.Y. Chen, C.S. Liao, T.M. Hsieh, C.H. Luo, J.J. Wu, H.H. Lee, G.B. Lee, J. Micromech. Microeng. 17(2), 367–375 (2007)
A.T. Woolley, D. Hadley, P. Landre, A.J. de Mello, R.A. Mathies, M.A. Northrup, Anal. Chem. 68(23), 4081–4086 (1996)
Acknowledgements
The authors gratefully acknowledge the financial supports provided by the National Science Council of Taiwan NSC 96-2622-E-002-001 and NSC 96-2120-M-006-008 for this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chien, LJ., Wang, JH., Hsieh, TM. et al. A micro circulating PCR chip using a suction-type membrane for fluidic transport. Biomed Microdevices 11, 359–367 (2009). https://doi.org/10.1007/s10544-008-9242-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10544-008-9242-z