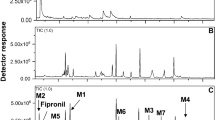
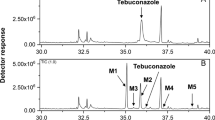
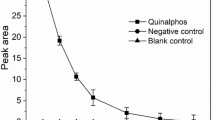
Abstract
Bisphenol A and its halogenated analogues are commonly used industrial chemicals with strong toxicological effects over many organisms. In this study, metabolic fate of bisphenol A and its halogenated analogues were evaluated with Cunninghamella elegans ATCC36112. Bisphenol A and related analogues were rapidly transformed into several metabolites by C. elegans within 2–4 days. Detailed analysis of metabolites reveals that both phase I and II metabolism occurred in C. elegans. Cytochrome P450-dependent hydroxylation was observed in BPA. However, major reaction with bisphenol A and analogues with 1-2 halogen atoms were the formation of glucose-conjugate, not being inhibited by cytochrome P450 inhibitor. Overall metabolic rates decreased with increasing number of substitution at 2- and 6-position of BPA structures, which may be consequences of limited bioavailability or steric hindrance to conjugate-forming reaction. Information from the current study will provide detailed insights over the fungal metabolism of BPA and analogues.







Similar content being viewed by others
References
Alexander HC, Dill DC, Smith LW, Guiney PD, Dorn P (1988) Bisphenol A: acute aquatic toxicity. Environ Toxicol Chem 7:19–26
Calafat AM, Kuklenyik Z, Reidy JA, Caudill SP, Ekong J, Needham LL (2005) Urinary concentration of bisphenol A and 4-nonylphenol in a human reference population. Envion Health Perspect 113:391–395
Chai W, Sakamaki H, Kitanaka S, Saito M, Horiuchi CA (2003) Biodegradation of bisphenol A by cultured cells of Caragana chamlagu. Biosci Biotechnol Biochem 67:218–220
Chai W, Handa Y, Suzuki M, Saito M, Kato N, Horiuchi CA (2005) Biodegradation of bisphenol A by fungi. Appl Biochem Biotechnol 120:175–182
Chen P-J, Linden KG, Hinton DE, Kashiwada S, Rosenfeldt EJ, Kullman SW (2006) Biological assessment of bisphenol A degradation in water following direct photolysis and UV advanced oxidation. Chemosphere 65:1094–1102
Domoradzki JY, Thornton CM, Pottenger LH, Hansen SC, Card TL, Markham DA, Dryzga MD, Shiotsuka RN, Waechter JM Jr (2004) Age and dose dependency of the pharmacokinetics and metabolism of bisphenol A in neonatal Sprague-Dawley rats following oral administration. Toxicol Sci 77:230–242
Hirano T, Honda Y, Watanabe T, Kuwahara M (2000) Degradation of bisphenol A by the lignin-degrading enzyme, manganese peroxidase, produced by the white-rot Basidiomycete, Pleurotus ostreatus. Biosci Biotechnol Biochem 64:1958–1962
Holzapfel CW, Williams DBG (1995) A facile route to 3a,8a-dihydrofuro[2,3-b]benzofuran. Tetrahedron 51:8555–8564
Huang H, Leung LK (2009) Bisphenol A downregulates CYP19 transcription in JEG-3 cells. Toxicol Lett 189:248–252
Ike M, Chen M-Y, Jin C-S, Masanori F (2002) Acute toxicity, mutagenicity, and estrogenicity of biodegradation products of bisphenol A. Environ Toxicol 17:457–461
Inoue H, Yokota H, Makino T, Yuasa A, Kato S (2001) Bisphenol A glucuronide, a major metabolite in rat bile after liver perfusion. Drug Metab Dispos 29:1084–1087
Iwashita K (2002) Recent studies of protein secretion by filamentous fungi. J Biosci Bioeng 94:530–535
Kang J-H, Kondo F, Katayama Y (2006a) Human exposure to bisphenol A. Toxicology 226:79–89
Kang J-H, Katayama Y, Kondo F (2006b) Biodegradation or metabolism of bisphenol A: from microorganisms to mammals. Toxicology 217:81–90
Lindholst C, Wayne PM, Marriott P, Pederson SN, Bjerregaard P (2003) Metabolism of bisphenol A in zebrafish (Danio rerio) and rainbow trout (Oncorhynchus mykiss) in relation to estrogenic response. Comp Biochem Physiol C 135:169–177
Lobos JH, Leib TK, Su TM (1992) Biodegradation of bisphenol A and other bisphenols by a Gram-negative aerobic bacterium. Appl Environ Microbiol 58:1823–1831
Morohoshi K, Shiraishi F, Oshima Y, Koda T, Nakajima N, Edmonds JS, Morita M (2003) Synthesis and estrogenic activity of bisphenol A mono- and di-β-D-glucopyranosides, plant metabolites of bisphenol A. Environ Toxicol Chem 22:2275–2279
Nakagawa Y, Suzuki T (2001) Metabolism of bisphenol A in isolated rat hepatocytes and oestrogenic activity of a hydroxylated metabolite in MCF-7 human breast cancer cells. Xenobiotica 31:113–123
Nakajima N, Ohshima Y, Serizawa S, Kouda T, Edmonds JS, Shiraishi F, Aono M, Kubo A, Tamaoki M, Saji H, Morita M (2002) Processing of bisphenol A by plant tissues: glucosylation by cultured BY-2 cells and glucosylation/translocation by plants of Nicotiana tabacum. Plant Cell Physiol 43:1036–1042
Newbold RR, Jefferson WN, Padilla-Banks E (2007) Long-term adverse effects of neonatal exposure to bisphenol A on the murine female reproductive tract. Reprod Toxicol 24:253–258
Niwa T, Fujimoto M, Kishimoto K, Yabusaki Y, Ishibashi F, Katagiri M (2001) Metabolism and interaction of bisphenol A in human hepatic cytochrome P450 and steroidogenic CYP17. Biol Pharm Bull 24:1064–1067
Noureddin MI, Furumoto T, Ishida Y, Fukui H (2004) Absroption and metabolism of bisphenol A, a possible endocrine disruptor, in the aquatic edible plant, water convolvulus (Ipomoea aquatica). Biosci Biotechnol Biochem 68:1398–1402
Pottenger LH, Domoradzki JY, Markham DA, Hansen SC, Cagen SZ, Waechter JM Jr (2000) The relative bioavailability and metabolism of bisphenol A in rats is dependent upon the route of administration. Toxicol Sci 54:3–18
Ramakrishnan S, Wayne NL (2008) Impact of bisphenol A on early embryonic development and reproductive maturation. Reprod Toxicol 25:420–425
Sajiki J, Yonekubo J (2004) Inhibition of seawater on bisphenol A (BPA) degradation by Fenton reagents. Environ Int 30:145–150
Smith MH, Gold MH (1979) Phanerochaete chrysosporium β-glucosidases: Induction, cellular localization, and physical characterization. Appl Environ Microbiol 37:938–942
Takeuchi T, Tsutsumi O, Ikezuki Y, Kamei Y, Osuga Y, Fujiwara T, Takai Y, Momoeda M, Yano T, Taketani Y (2006) Elevated serum bisphenol A levels under hyperandrogenic conditions may be caused by decreased UDP-glucuronosyltransferase activity. Endocr J 53:485–491
Toyama Y, Suzuki-Toyota F, Maekawa M, Ito C, Toshimori K (2004) Adverse effects of bisphenol A to spermiogenesis in mice and rats. Arch Histol Cytol 67:373–381
Vandenberg LN, Maffini MV, Wadia PR, Sonnenschein C, Rubin BS, Soto AM (2007) Exposure to environmentally relevant doses of the xenoestrogen bisphenol-A alters development of the fetal mouse mammary gland. Endocrinology 148:116–127
Venkatachalam N, Vinu A, Anandan S, Arabindoo B, Murugesan V (2006) Visible light active photocatalytic degradation of bisphenol-A using nitrogen doped TiO2. J Nanosci Nanotechnol 6:2499–2507
Volkel W, Colnot T, Csanady GA, Filser JG, Dekant W (2002) Metabolism and kinetics of bisphenol A in humans at low doses following oral administration. Chem Res Toxicol 15:1281–1287
Voordeckers JW, Fennell DE, Jones K, Haggblom MM (2002) Anaerobic biotransformation of tetrabromobisphenol A, tetrachlorobisphenol A, and bisphenol A in estuarine sediments. Environ Sci Technol 36:696–701
Yim S-H, Kim HJ, Lee IS (2003) Microbial metabolism of the environmental estrogen bisphenol A. Arch Pharm Res 26:805–808
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Keum, Y.S., Lee, H.R., Park, H.W. et al. Biodegradation of bisphenol A and its halogenated analogues by Cunninghamella elegans ATCC36112. Biodegradation 21, 989–997 (2010). https://doi.org/10.1007/s10532-010-9358-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10532-010-9358-8