Abstract
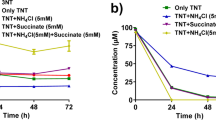
Anaerobic bacteria have been used to produce 2,4-dihydroxylamino-nitrotoluene (2,4DHANT), a reductive metabolite of 2,4,6-trinitrotoluene (TNT). Here, an aerobic TNT biodegrader Pseudomonas sp. strain TM15 produced 2,4DHANT as evidenced by the molecular ion with m/z of 199 identified from LC-TOFMS analyses. TNT biodegradation with a high cell concentration (109 cells/ml) led to a significant accumulation of 2,4DHANT in the culture medium, as well as hydroxylamino-dinitrotoluenes (HADNTs), although these products were not accumulated when a low cell concentration was used; also, the accumulation of diamino-nitrotoluene and of an unidentified metabolite were observed in the culture medium with the high cell concentration (1010 cells/ml). 2,4DHANT overproduction was a function of the aeration speed since cultures with low aeration speeds (30 rpm) had a 19-fold higher DHANT productivity than those aerated with high speeds (180 rpm); this indicates that molecular oxygen was related to the formation of 2,4DHANT. The quantification of dissolved oxygen (DO) in the media demonstrated that the productivity of 2,4DHANT was increased at low DO values. Moreover, supplying oxygen to the culture media produced a remarkable decrease of 2,4DHANT accumulation; these results clearly indicate that high 2,4DHANT production was a consequence of the oxygen deficit in the culture medium. This finding is useful for understanding the TNT biodegradation (bioremediation technology) in an anoxic environment.







Similar content being viewed by others
References
Ahlborg G Jr, Einisto P, Sorsa M (1988) Mutagenic activity and metabolites in the urine of workers exposed to trinitrotoluene (TNT). Br J Ind Med 45:353–358
Ahmad F, Hughes JB (2002) Reactivity of partially reduced arylhydroxylamine and nitrosoarene metabolites of 2,4,6-trinitrotoluene (TNT) toward biomass and humic acids. Environ Sci Technol 36:4370–4381
Banerjee H, Hawkins Z, Dutta S, Smoot D (2003) Effects of 2-amino-4,6-dinitrotoluene on p53 tumor suppressor gene expression. Mol Cell Biochem 252:387–389
Barrows SE, Cramer CJ, Truhlar DG, Elovitz MS, Weber EJ (1996) Factors controlling regioselectivity in the reduction of polynitroaromatics in aqueous solution. Environ Sci Technol 30:3028–3038
Berthe-Corti L, Jacobi H, Kleihauer S, Witte I (1998) Cytotoxicity and mutagenicity of a 2,4,6-trinitrotoluene (TNT) and hexogen contaminated soil in S. typhimurium and mammalian cells. Chemosphere 37:209–218
Borch T, Inskeep WP, Harwood JA, Gerlach R (2005) Impact of ferrihydrite and anthraquinone-2,6-disulfonate on the reductive transformation of 2,4,6-trinitrotoluene by a gram-positive fermenting bacterium. Environ Sci Technol 39:7126–7133
Chen WS, Juan CN, Wei KM (2005) Mineralization of dinitrotoluenes and trinitrotoluene of spent acid in toluene nitration process by Fenton oxidation. Chemosphere 60:1072–1079
Esteve-Nunez A, Caballero A, Ramos JL (2001) Biological degradation of 2,4,6-trinitrotoluene. Microbiol Mol Biol Rev 65:335–352, table of contents
Fernando T, Bumpus JA, Aust SD (1990) Biodegradation of TNT (2,4,6-trinitrotoluene) by Phanerochaete chrysosporium. Appl Environ Microbiol 56:1666–1671
Fiorella PD, Spain JC (1997) Transformation of 2,4,6-trinitrotoluene by Pseudomonas pseudoalcaligenes JS52. Appl Environ Microbiol 63:2007–2015
Fleischmann TJ, Walker KC, Spain JC, Hughes JB, Morrie Craig A (2004) Anaerobic transformation of 2,4,6-TNT by bovine ruminal microbes. Biochem Biophys Res Commun 314:957–963
Haidour A, Ramos JL (1996) Identification of products resulting from the biological reduction of 2,4,6-trinitrotoluene, 2,4-dinitrotoluene, and 2,6-dinitrotoluene by Pseudomonas sp. Environ Sci Technol 30:2365–2370
Hannink N, Rosser SJ, French CE, Basran A, Murray JA, Nicklin S, Bruce NC (2001) Phytodetoxification of TNT by transgenic plants expressing a bacterial nitroreductase. Nat Biotechnol 19:1168–1172
Harter DR (1985) The use and importance of nitroaromatic compounds in the chemical industry. In: Rickert DE (ed). Hemisphere publishing, Washington, DC
Hawari J, Beaudet S, Halasz A, Thiboutot S, Ampleman G (2000) Microbial degradation of explosives: biotransformation versus mineralization. Appl Microbiol Biotechnol 54:605–618
Heiss G, Knackmuss HJ (2002) Bioelimination of trinitroaromatic compounds: immobilization versus mineralization. Curr Opin Microbiol 5:282–287
Homma-Takeda S, Hiraku Y, Ohkuma Y, Oikawa S, Murata M, Ogawa K, Iwamuro T, Li S, Sun GF, Kumagai Y, Shimojo N, Kawanishi S (2002) 2,4,6-trinitrotoluene-induced reproductive toxicity via oxidative DNA damage by its metabolite. Free Radic Res 36:555–566
Huang S, Lindahl PA, Wang C, Bennett GN, Rudolph FB, Hughes JB (2000) 2,4,6-trinitrotoluene reduction by carbon monoxide dehydrogenase from Clostridium thermoaceticum. Appl Environ Microbiol 66:1474–1478
Kroger M, Schumacher ME, Risse H, Fels G (2004) Biological reduction of TNT as part of a combined biological-chemical procedure for mineralization. Biodegradation 15:241–248
Letzel S, Goen T, Bader M, Angerer J, Kraus T (2003) Exposure to nitroaromatic explosives and health effects during disposal of military waste. Occup Environ Med 60:483–488
Leungsakul T, Keenan BG, Smets BF, Wood TK (2005) TNT and nitroaromatic compounds are chemoattractants for Burkholderia cepacia R34 and Burkholderia sp. strain DNT. Appl Microbiol Biotechnol 69:321–325
Lewis TA, Goszczynski S, Crawford RL, Korus RA, Admassu W (1996) Products of anaerobic 2,4,6-trinitrotoluene (TNT) transformation by Clostridium bifermentans. Appl Environ Microbiol 62:4669–4674
Lewis TA, Newcombe DA, Crawford RL (2004) Bioremediation of soils contaminated with explosives. J Environ Manage 70:291–307
Liou MJ, Lu MC, Chen JN (2004) Oxidation of TNT by photo-Fenton process. Chemosphere 57:1107–1114
Maeda T, Kadokami K, Ogawa HI (2006a) Characterization of 2,4,6-trinitrotoluene (TNT)-metabolizing bacteria isolated from TNT-polluted soils in the Yamada Green Zone, Kitakyushu, Japan. J Environ Biotechnol 6:33–39
Maeda T, Kubota A, Nagafuchi N, Kadokami K, Ogawa HI (2006b) Crucial problem in rapid spectrophotometric determination of 2,4,6-trinitrotoluene and its breakthrough method. J Microbiol Methods 66:568–571
Maeda T, Nagafuchi N, Kubota A, Kadokami K, Ogawa HI (2006c) One-step isolation and identification of hydroxylamino-dinitrotoluenes, unstable products from 2,4,6-trinitrotoluene metabolites, with thin-layer chromatography and laser time-of-flight mass spectrometry. J Chromatogr Sci 44:96–100
Maeda T, Nagafuchi N, Kubota A, Kadokami K, Ogawa HI (2007a) Identification of spontaneous conversion products of unstable 2,4,6-trinitrotoluene metabolites, hydroxylamino-dinitrotoluenes, by combination of thin-layer chromatography and laser time-of-flight mass spectrometry. J Chromatogr Sci 45:345–349
Maeda T, Nakamura R, Kadokami K, Ogawa HI (2007b) Relationship between mutagenicity and reactivity or biodegradability for nitroaromatic compounds. Environ Toxicol Chem 26:237–241
Newcombe DA, Crawford RL (2007) Transformation and fate of 2,4,6-trinitrotoluene (TNT) in anaerobic bioslurry reactors under various aeration schemes: implications for the decontamination of soils. Biodegradation 18:741–754
Nishino SF, Spain JC (1993) Degradation of nitrobenzene by a Pseudomonas pseudoalcaligenes. Appl Environ Microbiol 59:2520–2525
Padda RS, Wang C, Hughes JB, Kutty R, Bennett GN (2003) Mutagenicity of nitroaromatic degradation compounds. Environ Toxicol Chem 22:2293–2297
Rieger P-G, Knackmuss H-J (1995) Basic knowledge and perspectives on biodegradation of 2,4,6-trinitrotoluene and related nitroaromatic compounds in contaminated soil. In: Spain JC (ed). Plenum Press, New York, NY
Saka M (2004) Developmental toxicity of p,p′-dichlorodiphenyltrichloroethane, 2,4,6-trinitrotoluene, their metabolites, and benzo[a]pyrene in Xenopus laevis embryos. Environ Toxicol Chem 23:1065–1073
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular Cloning, A Laboratory Manual, 2nd edn. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY
Snellinx Z, Nepovim A, Taghavi S, Vangronsveld J, Vanek T, van der Lelie D (2002) Biological remediation of explosives and related nitroaromatic compounds. Environ Sci Pollut Res Int 9:48–61
Sun Y, Iemitsu M, Shimojo N, Miyauchi T, Amamiya M, Sumi D, Hayashi T, Sun G, Shimojo N, Kumagai Y (2005) 2,4,6-Trinitrotoluene inhibits endothelial nitric oxide synthase activity and elevates blood pressure in rats. Arch Toxicol 79:705–710
Tan EL, Ho CH, Griest WH, Tyndall RL (1992) Mutagenicity of trinitrotoluene and its metabolites formed during composting. J Toxicol Environ Health 36:165–175
Vorbeck C, Lenke H, Fischer P, Spain JC, Knackmuss HJ (1998) Initial reductive reactions in aerobic microbial metabolism of 2,4,6-trinitrotoluene. Appl Environ Microbiol 64:246–252
Won WD, DiSalvo LH, Ng J (1976) Toxicity and mutagenicity of 2,4,-6-trinitrotoluene and its microbial metabolites. Appl Environ Microbiol 31:576–580
Yin H, Wood TK, Smets BF (2005) Reductive transformation of TNT by Escherichia coli: pathway description. Appl Microbiol Biotechnol 67:397–404
Acknowledgments
We are grateful to Chugoku Kayaku Co. Ltd., Hiroshima, Japan, for their gift of 2,4,6-trinitrotoluene, and to Dr. R. Spanggord, Chemical Sciences and Technology Department, SRI International, Melno Park, CA, for his gift of 2-hydroxylamino-4,6-dinitrotoluene, 4-hydroxylamino-2,6-dinitrotolulene, and 2,4-diamino-nitrotoluene. We express much gratitude to Dr. Robin Gerlach, Montana State University, Bozeman, MT, for his help in identifying 2,4-dihydroxylamino-6-nitrotoluene, to Dr. Hideaki Uchida and Dr. Yoshizumi Takigawa, Agilent Technologies Japan, Ltd., for their support with LC/TOF–MS, and to Dr. Norikazu Nishino, Department of Biological Functions and Engineering, Graduate School of Life Science and Systems Engineering, Kyushu Institute of Technology, for his support with the LC/MS analyses. We are also grateful for the assistance of Dr. Thomas K. Wood of Texas A & M University, College Station, TX, for his kind helps with preparing this manuscript.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kubota, A., Maeda, T., Nagafuchi, N. et al. TNT biodegradation and production of dihydroxylamino-nitrotoluene by aerobic TNT degrader Pseudomonas sp. strain TM15 in an anoxic environment. Biodegradation 19, 795–805 (2008). https://doi.org/10.1007/s10532-008-9182-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10532-008-9182-6