Abstract
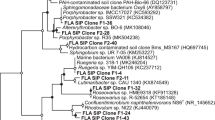
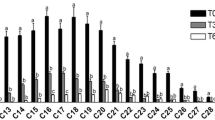
The aerobic polyaromatic hydrocarbon (PAH) degrading microbial communities of two petroleum-impacted Spartina-dominated salt marshes in the New York/New Jersey Harbor were examined using a combination of microbiological, molecular and chemical techniques. Microbial isolation studies resulted in the identification of 48 aromatic hydrocarbon-degrading bacterial strains from both vegetated and non-vegetated marsh sediments. The majority of the isolates were from the genera Paenibacillus and Pseudomonas. Radiotracer studies using 14C-phenanthrene and 14C-pyrene were used to measure the PAH-mineralization activity in salt marsh sediments. The results suggested a trend towards increased PAH mineralization in vegetated sediments relative to non-vegetated sediments. This trend was supported by the enumeration of PAH-degrading bacteria in non-vegetated and vegetated sediment using a Most Probable Numbers (MPN) technique, which demonstrated that PAH-degrading bacteria existed in non-vegetated and vegetated sediments at levels ranging from 102 to 105 cells/g sediment respectively. No difference between microbial communities present in vegetated versus non-vegetated sediments was found using terminal restriction fragment length polymorphism (of the 16S rRNA gene) or phospholipid fatty acid analysis. These studies provide information on the specific members and activity of the PAH-degrading aerobic bacterial communities present in Spartina-dominated salt marshes in the New York/New Jersey Harbor estuary.





Similar content being viewed by others
References
Aguilera M, Monteoliva-Sanchez M, Suarez A, Guerra V, Lizama C, Bennasar A, Ramos-Cormenzana A (2001) Paenibacillus jamilae sp. nov., an exopolysaccharide-producing bacterium able to grow in olive-mill wastewater. Int J Syst Evol Microbiol 51:1687–1692
Bent E, Breuil C, Enebak S, Chanway CP (2002) Surface colonization of lodgepole pine (Pinus contorta var. latifolia [Dougl. Engelm.]) roots by Pseudomonas fluorescens and Paenibacillus polymyxa under gnotobiotic conditions. Plant Soil 24:187–196
Berardesco G, Dyhrman S, Gallagher E, Shiaris MP (1998) Spatial and temporal variation of phenanthrene-degrading bacteria in intertidal sediments. Appl Environ Microbiol 64:2560–2565
Bergen A, Alderson C, Bergfors R, Aquila C, Matsil MA (2000) Restoration of a Spartina alterniflora salt marsh following a fuel oil spill, New York City, NY. Wetlands Ecol & Manag 8:185–195
Boldrin B, Tiehm A, Fritzsche C (1993) Degradation of phenanthrene, fluorene, fluoranthene, and pyrene by a Mycobacterium sp. Appl Environ Microbiol 59:1927–1930
Boonchan S, Britz ML, Stanley GA (2000) Degradation and mineralization of high-molecular-weight polycyclic aromatic hydrocarbons by defined fungal-bacterial cocultures. Appl Environ Microbiol 66:1007–1019
Burke DJ, Hamerlynck EP, Hahn D (2003) Interactions between the salt marsh grass Spartina patens, arbuscular mycorrhizal fungi and sediment bacteria during the growing season. Soil Biol Biochem 35:501–511
Cerniglia CE (1992) Biodegradation of polycyclic aromatic hydrocarbons. Biodegradation 3:351–368
Cigolini J (2000) Molecular analysis of polycyclic aromatic hydrocarbon degradation by Mycobacterium sp. strain PYO1. Ph.D. Thesis. Rutgers The State University of New Jersey, New Brunswick NJ
Daane LL, Harjono I, Zylstra GJ, Häggblom MM (2001) Isolation and characterization of PAH-degrading bacteria associated with the rhizosphere of salt marsh plants. Appl Environ Microbiol 67:2683–2651
Daane LL, Harjono I, Barns SM, Launen LA, Palleroni NJ, Häggblom MM (2002) PAH-degradation by Paenibacillus spp. and description of Paenibacillus naphthalenovorans sp. nov., a naphthalene-degrading bacterium from the rhizosphere of salt marsh plants. Int J Syst Evol Microbiol 52:131–139
DeMan JC (1975) The probability of most probable numbers. Eur J Appl Microbiol 1:67–78
Difco Laboratories (1998) Difco Manual 11th ed. Becton Dickinson and Company, Sparks, ME
Elo S, Suominen I, Kämpfer P, Juhanoja J, Salkinoja-Salonen M, Haahtela K (2001) Paenibacillus borealis sp. nov., a nitrogen-fixing species isolated from spruce forest humus in Finland. Int J Syst Evol Microbiol 51(2):535–545
Ensley BD, Ratzkin BJ, Osslund TD, Simon MJ, Wackett LP, Gibson DT (1983) Expression of naphthalene oxidation genes in Escherichia coli results in the biosynthesis of indigo. Science 222(4620):167–169
Garcia-Blanco S, Venosa A, Suidan M, Lee K, Cobanli S, Haines J (2007) Biostimulation for the treatment of an oil-contaminated coastal salt marsh. Biodegradation 18(1):1–15
Gigliotti CL, Brunciak PA, Dachs J, Glenn TR IV, Nelson ED, Totten LA, Eisenreich SJ (2002) Air–water exchange of polycyclic aromatic hydrocarbons in the New York–New Jersey, USA Harbor estuary. EnvironToxicol Chem 21:235–244
Heitkamp MA, Freeman JP, Miller DW, Cerniglia CE (1988) Pyrene degradation by a Mycobacterium sp.: identification of ring oxidation and ring fission products. Appl Environ Microbiol 54:2556–2565
Huntley SL, Bonnevie NL, Wenning RJ (1995) Polycyclic aromatic hydrocarbon and petroleum hydrocarbon contamination in sediment from the Newark Bay estuary, New Jersey. Arch Environ Contam Toxicol 28:93–107
Iida T, Nakamura K, Izumi A, Mukouzaka Y, Kudo T (2006) Isolation and characterization of a gene cluster for dibenzofuran degradation in a new dibenzofuran-utilizing bacterium, Paenibacillus sp. strain YK5. Arch Microbiol 184:305–315
Johnson JL (1994) Similarity analysis of rRNAs. In: Gerhardt P, Murray RGE, Wood WA, Krieg NR (eds) Methods for general and molecular bacteriology. American Society for Microbiology, Washington, DC, pp 683–700
Jones KW, Stern EA, Donato KR, Clescerini NL (1998) Decontamination of dredged material from the Port of New York and New Jersey. In: Proceedings of the 5th International Petroleum Environmental Conference, Albuquerque, NM, October 20–23, 1998, pp 225–239
Jackson WA, Pardue JH (1999) Potential for enhancement of biodegradation of crude oil in Louisiana salt marshes using nutrient amendments. Water Air Soil Pollut 109:343–355
Keck J, Sims RC, Coover M, Park K, Symons B (1989) Evidence for cooxidation of polynuclear aromatic hydrocarbons in soil. Water Res 23:1467–1476
Kelley I, Cerniglia CE (1995) Degradation of a mixture of high molecular-weight polycyclic aromatic hydrocarbons by a Mycobacterium strain PYR-1. J Soil Contam 4:77–91
Launen LA, Buggs VH, Eastep ME, Enriquez RC, Leonard JW, Blaylock MJ, Huang J-W, Häggblom MM (2002) Bioremediation of polyaromatic hydrocarbon-contaminated sediments in aerated bioslurry reactors. Bioremediation J 6(2):125–141
Litten S (2003) Contaminant assessment and reduction project water (CARP). New York State Department of environmental conservation. Bureau of Water Assessment and Management, Division of Water, Albany, NY pp 158
Lloyd-Jones G, Hunter DWF (1997) Characterization of fluoranthene- and pyrene-degrading Mycobacterium-like strains by RAPD and SSU sequencing. FEMS Microbiol Lett 153:51–56
Meyer S, Moser R, Neef A, Stahl U, Kämpfer P (1999) Differential detection of key enzymes of polyaromatic-hydrocarbon-degrading bacteria using PCR and gene probes. Microbiology 145:1731–1741
Mitsch WJ, Gosselink JG (2000) Wetlands. John Wiley and Sons, New York, NY
Pichinoty F, Waterbury JB, Mandel M, Asselineau J (1986) Bacillus gordonae sp. nov., une nouvelle espace appartenant au second groupe morphologique, dégrandant divers composés aromatiques. Ann Inst Pasteur/Microbiol (Paris) 137A:65–78
Sakai M, Ezaki S, Suzuki N, Kurane R (2005) Isolation and characterization of a novel polychlorinated biphenyl-degrading bacterium, Paenibacillus sp. KBC101. Appl Microbiol Biotech 68:111–116
Sakano Y, Kerkhof L (1998) Assessment of changes in microbial community structure during operation of an ammonia biofilter with molecular tools. Appl Environ Microbiol 64:4877–4882
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning—A laboratory manual. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY
Scala DJ, Kerkhof LJ (1998) Nitrous oxide reductase (nosZ) gene-specific PCR primers for detection of denitrifiers and three nosZ genes from marine sediments. FEMS Microbiol Lett 162:61–68
Siefert JL, Larios-Sanz M, Nakamura LK, Slepecky RA, Paul JH, Moore ERB, Fox GE, Jurtshuk P Jr (2000) Phylogeny of marine Bacillus isolates from the Gulf of Mexico. Curr Microbiol 41:84–88
Sokal RR, Rohlf FJ (1995) Biometry: the principles and practices of statistics in biological research, 3rd edn. W.H. Freeman and Company, New York, p 887
Stanier RY, Palleroni NJ, Doudoroff M (1966) The aerobic pseudomonads: a taxonomic study. J Gen Microbiol 43:159–271
Steinberg N, Suszkowski DJ, Clark L, Way J (2004) Health of the Harbor: the first comprehensive look at the state of the NY/NJ Harbor estuary, a report to the NY/NJ Harbor estuary program. Hudson River Foundation, New York, NY
US Environmental Protection Agency, USEPA-Region 2, USEPA-ORD (2003) Final report: sediment quality of the NY/NJ Harbor system: a 5-year revisit, 1993/4–1998. An investigation under the Regional Environmental Monitoring and Assessment Program (REMAP). Edison, NJ and Narragansett, RI
Van Metre PC, Mahler BJ (2005) Trends in hydrophobic organic contaminants in urban and reference lake sediments across the United States, 1970–2001. Environ Sci Technol 39:5567–5574
Wang R-F, Cao W-W, Cerniglia CE (1995) Phylogenetic analysis of polycyclic aromatic hydrocarbon degrading mycobacteria by 16S rRNA sequencing. FEMS Microbiol Lett 130:75–80
Wright AL, Weaver RW, Webb JW (1997) Oil bioremediation in salt marsh mesocosms as influenced by N and P fertilization, flooding, and season. Water Air Soil Pollut 95:179–191
Wright AL, Weaver RW (2004) Fertilization and bioaugmentation for oil biodegradation in salt marsh mesocosms Wat. Air Soil Pollut 156:229–240
Zylstra GJ, Kim E (1997) Aromatic hydrocarbon degradation by Sphingomonas yanoikuyae B1. J Ind Microbiol Biotech 19:408–414
Zhu X, Venosa AD, Makram TS, Lee K (2004) Guidelines for the bioremediation of oil-contaminated salt marshes. EPA/600/R-04/074. U.S. Environmental Protection Agency, Cincinnati, OH
Acknowledgements
Funding for this project was provided by the New Jersey Commission on Science and Technology, and the Northeast Hazardous Substance Research Center. Greg Flanagan and Al Habib provided expert technical assistance. We thank Dr. Lee Kerkhof (Institute of Marine and Coastal Sciences, Rutgers University) for assistance with the TRFLP analyses. We thank Carl Alderson for providing sampling access to the Gulfport Reach Site, and Drs. Michael Levandowsky and Evelyn Drake for sampling assistance at the Gulfport Reach and Refinery Sites. We thank Dr.’s Karen Cangialosi and Dick Jardine for assistance with the statistical analysis.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Launen, L.A., Dutta, J., Turpeinen, R. et al. Characterization of the indigenous PAH-degrading bacteria of Spartina dominated salt marshes in the New York/New Jersey Harbor. Biodegradation 19, 347–363 (2008). https://doi.org/10.1007/s10532-007-9141-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10532-007-9141-7