Abstract
Non-crop habitats play a key role in maintaining functional diversity and ecosystem services in farmland. However, the interplay between beneficial insects and landscape variables has rarely been investigated in Neotropical agroecosystems. We used flower flies as a model group to investigate the effects of landscape attributes on beneficial insects in agroecosystems across a gradient of landscape complexity. We specifically ask: (i) Do the abundance and species richness of flower flies in cereal crops increase with increasing landscape complexity? (ii) Do the effects of landscape variables on local flower fly communities differ between spatial scales? (iii) How do landscape complexity and local factors (crop size, altitude and insecticide applications) affect beta diversity? We sampled flower flies in 54 edges within 18 wheat crops in Paraná State, southern Brazil. The percentage of non-crop habitats, landscape diversity and edge density were the explanatory variables, which were calculated at multiple spatial scales for each landscape. We collected 8340 flower flies, distributed in 12 genera and 52 species. Species richness was positively associated with the percentage of non-crop habitats, but total abundance presented non-clear pattern. However, abundance without the dominant species was also positively associated with the percentage non-crop habitats. Similarly, beta diversity was related to non-crop habitats, suggesting that the reduction in non-crop habitats implies in species loss. We have provided the first insights into the importance of non-crop habitats on the conservation of beneficial insects within Neotropical farmlands. To guarantee high levels of biodiversity within agroecosystems we need to promote the conservation and restoration of non-crop habitats in the surrounding landscapes.

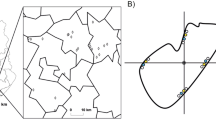
Source Metropolitan region of Londrina city 23°55′46″S and 51°19′11″W. Google Earth. September 04, 2013. The numbers from 1 to 5 represent the urban areas of the cities of Londrina (1), Cambé (2), Rolândia (3), Arapongas (4) and Ibiporã (5). Yellow circles represent the 2-km landscape sectors sampled in 2012, while black circles indicate the landscapes sampled in 2013. (Color figure online)




Similar content being viewed by others
References
Altieri MA (1999) The ecological role of biodiversity in agroecosystems. Agric Ecosyst Environ 74:19–31. https://doi.org/10.1016/b978-0-444-50019-9.50005-4
Amorim DS (2009) Neotropical Diptera diversity: richness, patterns, and perspectives. In: Pape T, Bickel D, Meier R (eds) Diptera diversity: status, challenges and tools. Koninklijke Brill, Leiden, pp 71–97. https://doi.org/10.1163/ej.9789004148970.i-459.17
Anderson MJ (2001) Permutational multivariate analysis of variance. Department of Statistics, University of Auckland, Auckland
Arcaya E, Pérez-Bañón C, Mengual X, Zubcoff-Vallejo JJ, Rojo S (2017) Life table and predation rates of the syrphid fly Allograpta exotica, a control agent of the cowpea aphid Aphis craccivora. Biol Control 115:74–84. https://doi.org/10.1016/j.biocontrol.2017.09.009
Aviron S, Burel F, Baudry J, Schermann N (2005) Carabid assemblages in agricultural landscapes: impacts of habitat features, landscape context at different spatial scales and farming intensity. Agric Ecosyst Environ 108:205–217
Baselga A (2010) Partitioning the turnover and nestedness components of beta diversity. Glob Ecol Biogeogr 19:134–143. https://doi.org/10.1111/j.1466-8238.2009.00490.x
Baselga A, Orme CDL (2012) Betapart: an R package for the study of beta diversity. Methods Ecol Evol 3:808–812. https://doi.org/10.1111/j.2041-210X.2012.00224.x
Batáry P, Báldi A, Kleijn D, Tscharntke T (2011) Landscape-moderated biodiversity effects of agri-environment management: a meta-analysis. Proc Soc Lond Ser B-Biol Sci 278:1894–1902. https://doi.org/10.1098/rspb.2010.1923
Bates D, Maechler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67:1–48. https://doi.org/10.18637/jss.v067.i01
Bianchi FJJA, Booij CJH, Tscharntke T (2006) Sustainable pest regulation in agricultural landscapes: a review on landscape composition, biodiversity and natural pest control. Proc Royal Soc B: Biol Sci 273(1595):1715–1727
Buhk C, Alt M, Steinbauer MJ, Beierkuhnlein C, Warren SD, Jentsc A (2017) Homogenizing and diversifying effects of intensive agricultural land-use on plant species beta diversity in Central Europe—A call to adapt our conservation measures. Sci Total Environ 576:225–233. https://doi.org/10.1016/j.scitotenv.2016.10.106
Billeter R, Liira J, Bailey D, Bugter R, Arens P, Augenstein I, Aviron S, Baudry J, Bukacek R, Burel F, Cerny M, De Blust G, De Cock R, Diekötter T, Dietz H, Dirksen J, Dormann C, Durka W, Frenzel M, Hamersky R, Hendrickx F, Herzog F, Klotz S, Koolstra B, Lausch A, Le Coeur D, Maelfait JP, Opdam P, Roubalova M, Schermann A, Schermann N, Schmid T, Schweiger O, Smulders MJM, Speelmans M, Simova P, Verboom J, van Wingerden WKRE, Zobel M (2008) Indicators for biodiversity in agricultural landscapes: a pan-European study. J Appl Ecol 45:141–150. https://doi.org/10.1111/j.1365-2664.2007.01393.x
Bolker BM (2010) bbmle: Tools for general maximum likelihood estimation. The Comprehensive R Archive Network (CRAN), Vienna, Austria
Borges ZM, Couri MS (2009) Revision of Toxomerus Macquart, 1855 (Diptera: Syrphidae) from Brazil with synonymic notes, identification key to the species and description of three new species. Zootaxa 2179:1–72
Borges ZM, Pamplona DM (2003) Revision of the Neotropical Xanthandrus Verral (Diptera, Syrphidae). Rev Bras Entomol 47:155–167. https://doi.org/10.1590/s0085-56262003000200002
Bortolotto OC, de Menezes AO Jr, Hoshino AT, Campos TA (2016) Distance from the edge of forest fragments influence the abundance of aphidophagous flower flies (Diptera: Syrphidae) in wheat fields. Acta Sci 38:157–164. https://doi.org/10.4025/actasciagron.v38i2.27711
Burgio G, Sommaggio D (2007) Syrphids as landscape indicators in Italian agroecosystems. Agric Ecosyst Environ 120:416–422. https://doi.org/10.1016/j.agee.2006.10.021
Burnham KP, Anderson DR (1998) Model Selection and Inference: a Practical Information-Theoretic Approach. Springer-Verlag, New York. https://doi.org/10.1007/978-1-4757-2917-7_3
Burnham KP, Anderson DR (2002) Model selection and multimodel inference: a practical information-theoretical approach, 2nd edn. Springer-Verlag, New York. https://doi.org/10.1007/b97636
Catano CP, Dickson TL, Myers JA (2017) Dispersal and neutral sampling mediate contingent effects of disturbance on plant beta-diversity: a meta-analysis. Ecol Lett 20(3):347–356. https://doi.org/10.1111/ele.12733
Clough Y, Holzschuh A, Gabriel D, Purtauf T, Kleijn D, Kruess A, Steffan-Dewenter I, Tscharntke T (2007) Alpha and beta diversity of arthropods and plants in organically and conventionally managed wheat fields. J Appl Ecol 44:804–812. https://doi.org/10.1111/j.1365-2664.2007.01294.x
de Sousa JMT, Marinoni RC, Marinoni L (2014) Open and disturbed habitats support higher diversity of Syrphidae (Diptera)? A case study during three yr of sampling in a fragment of Araucaria forest in Southern Brazil. J Insect Sci 14:1–8. https://doi.org/10.1093/jisesa/ieu098
Devoto M, Medan D, Roig-Alsina A, Montaldo NH (2009) Patterns of species turnover in plant-pollinator communities along a precipitation gradient in Patagonia (Argentina). Aust Ecol 34:848–857. https://doi.org/10.1111/j.1442-9993.2009.01987.x
ESRI (Environmental Systems Research Institute) (2005) ArcGIS Desktop: Release 8
Fahrig L (2003) Effects of habitat fragmentation on biodiversity. Annu Rev Ecol Evol Syst 34:487–515. https://doi.org/10.1146/annurev.ecolsys.34.011802.132419
Fahrig L (2017) Ecological responses to habitat fragmentation Per Se. Annu Rev Ecol Evol Syst 48(1):1–23
Fahrig L, Baudry J, Brotons L, Burel FG, Crist TO, Fuller RJ, Sirami C, Siriwardena GM, Martin JL (2011) Functional landscape heterogeneity and animal biodiversity in agricultural landscapes. Ecol Lett 14:101–112. https://doi.org/10.1111/j.1461-0248.2010.01559.x
FAO (1988) FAO/UNESCO Soil map of the world, revised legend with corrections and updates. World Soil Resources Report 60, FAO, Rome. Reprinted with updates as technical paper 20 (1997), ISRIC, Wageningen
Fontaine C, Dajoz I, Meriguet J, Loreau M (2006) Functional diversity of plant-pollinator Interaction webs enhances the persistence of plant communities. PLoS Biol 4:129–135. https://doi.org/10.1371/journal.pbio.0040001
Gassen DN (1986) Parasitos, patógenos e predadores de insetos associados à cultura do trigo. Embrapa – trigo, Brazil
Geiger F, Bengtsson JF, Berendse F, Weisser WW, Emmerson M, Morales MB, Ceryngier P, Liira J, Tscharntke T, Winqvist C, Eggers S, Bommarco R, Pärt T, Bretagnolle V, Plantegenest M, Clement LW, Dennis C, Palmer C, Oñate JJ, Guerrero I, Hawro V, Aavik T, Thies C, Flohre A, Hänke S, Fischer C, Goedhart PW, Inchausti P (2010) Persistent negative effects of pesticides on biodiversity and biological control potential on European farmland. Basic Appl Ecol 11:97–105. https://doi.org/10.1016/j.baae.2009.12.001
Gilbert F, Rotheray GE, Zafar R, Emerson P (1994) The evolution of feeding strategies. In: Eggleton P, Vane-Wright R (eds) Phylogenetics and Ecology. Academic Press, London, pp 324–343
González E, Salvo A, Valladares G (2015) Sharing enemies: evidence of forest contribution to natural enemy communities in crops, at different spatial scales. Insect Conserv Diver 8:359–366. https://doi.org/10.1111/icad.12117
González E, Salvo A, Defagó MT, Valladares G (2016a) A moveable feast: insects moving at the forest-crop interface are affected by crop phenology and the amount of forest in the landscape. PLoS ONE 11:e0158836. https://doi.org/10.1371/journal.pone.0158836
González E, Salvo A, Valladares G (2016b) Natural vegetation cover in the landscape and edge effects: differential responses of insect orders in a fragmented forest. Insect Sci 24:891–901. https://doi.org/10.1111/1744-7917.12377
González E, Salvo A, Valladares G (2017) Arthropod communities and biological control in soybean fields: forest cover at landscape scale is more influential than forest proximity. Agric Ecosyst Environ 239:359–367. https://doi.org/10.1016/j.agee.2017.02.002
Grez AA, Prado E (2000) Effect of plant patch shape and surrounding vegetation on the dynamics of predatory coccinellids and their prey Brevicoryne brassicae (Hemiptera: Aphididae). Environ Entomol 29:1244–1250. https://doi.org/10.1603/0046-225x-29.6.1244
Haenke S, Scheid B, Schaefer M, Tscharntke T, Thies C (2009) Increasing syrphid fly diversity and density in sown flower strips within simple vs. complex landscapes. J Appl Ecol 46:1106–1114. https://doi.org/10.1111/j.1365-2664.2009.01685.x
Haenke S, Kovács-Hostyánszki A, Fründ J, Batáry P, Jauker B, Tscharntke T, Holzschuh A (2014) Landscape configuration of crops and hedgerows drives local syrphid fly abundance. J Appl Ecol 51:505–513. https://doi.org/10.1111/1365-2664.12221
Hendrickx F, Maelfait JP, Wingerden WV, Schweiger O, Speelmans M, Aviron S, Augenstein I, Billeter R, Bailey D, Bukacek R, Burel F, Diekötter T, Dirksen J, Herzog F, Liira J, Roubalova M, Vandomme V, Rob Bugter R (2007) How landscape structure, land-use intensity and habitat diversity affect components of total arthropod diversity in agricultural landscapes. J Appl Ecol 44:340–351. https://doi.org/10.1111/j.1365-2664.2006.01270.x
Holland JM, Thomas SR (1996) Phacelia tanacetifolia flower strip: their effect on beneficial invertebrates and gamebird chick foo in an integrated farming system. Acta Jutl Nat Sci Ser 71:171–182
Holzschuh A, Steffan-Dewenter I, Tscharntke T (2010) How do landscape composition and configuration, organic farming and fallow strips affect the diversity of bees, wasps and their parasitoids? J Anim Ecol 79:491–500. https://doi.org/10.1111/j.1365-2656.2009.01642.x
Hurvich CM, Tsai CL (1989) Regression and time series model selection in small samples. Biometrika 76:297–307
IAPAR (2000) Cartas climáticas do estado do Paraná. http://www.iapar.br/modules/conteudo/conteudo.php?conteudo=856. Accessed 27 Nov 2016
Jauker F, Wolters V (2008) Flower flies are efficient pollinators of oilseed rape. Oecologia 156:819–823. https://doi.org/10.2307/2336663
Jauker F, Diekötter T, Schwarzbach F, Wolters V (2009) Pollinator dispersal in an agricultural matrix: opposing responses of wild bees and flower flies to landscape structure and distance from main habitat. Landsc Ecol 24:547–555. https://doi.org/10.1007/s10980-009-9331-2
Jönsson AM, Ekroos J, Dänhardt J, Andersson GKS, Olsson O, Smith HG (2015) Sown flower strips in southern Sweden increase abundances of wild bees and hoverflies in the wider landscape. Biol Conserv 184:51–58. https://doi.org/10.1016/j.biocon.2014.12.027
Kleijn D, van Langevelde F (2006) Interacting effects of landscape context and habitat quality on flower visiting insects in agricultural landscapes. Basic Appl Ecol 7:214–301. https://doi.org/10.1016/j.baae.2005.07.011
Krauss J, Gallenberger I, Steffan-Dewenter I (2011) Decreased functional diversity and biological pest control in conventional compared to organic crop fields. PLoS ONE 6:e19502. https://doi.org/10.1371/journal.pone.0019502
Landis DA, Wratten SD, Gurr GM (2000) Habitat management to conserve natural enemies of arthropod pests in agriculture. Annu Rev Entomol 45:175–201. https://doi.org/10.1146/annurev.ento.45.1.175
Larrivée M, Fahrig L, Drapeau P (2008) Edge effects created by wildfire and clear-cutting on boreal forest ground-dwelling spiders. For Ecol Manage 255:1434–1445. https://doi.org/10.1016/j.foreco.2007.10.062
Legendre P, Legendre L (eds) (2012) Numerical ecology, vol 24. Elsevier, Oxford
López O, Salto C, Luiselli S (2003) Foeniculum vulgare Miller como hospedera de pulgones y sus enemigos naturales en otoño. Rev FAVE Cienc. Agrar 2:55–65
Loreau M, Mouquet N, Gonzalez A (2003) Biodiversity as spatial insurance in heterogeneous landscapes. Proc Natl Acad Sci USA 100:12765–12770. https://doi.org/10.1073/pnas.2235465100
Lucas A, Bull JC, de Vere N, Neyland PJ, Forman DW (2017) Flower resource and land management drives hoverfly communities and bee abundance in seminatural and agricultural grasslands. Ecol Evol 7:8073–8086. https://doi.org/10.1002/ece3.3303
Magurran AE (2004) Measuring biological diversity. Blackwell, Maldan
Manfrino R, Salto C, Zumoffen L (2011) Estudio de las asociaciones áfidos entomófagos sobre Foeniculum vulgare (Umbelliferae) y Conyza bonariensis (Asteraceae) en la región central de Santa Fe, Argentina. Rev Soc Entomol Argent 70:99–109
Martensen AC, Pimentel RG, Metzger JP (2008) Relative effects of fragment size and connectivity on bird community in the Atlantic Rain Forest: implications for conservation. Biol Conserv 141:2184–2192. https://doi.org/10.1016/j.biocon.2008.06.008
Martensen AC, Ribeiro MC, Banks-Leite C, Prado PI, Metzger JP (2012) Associations of forest cover, fragment area, and connectivity with Neotropical understory bird species richness and abundance. Conserv Biol 26:1100–1111. https://doi.org/10.1111/j.1523-1739.2012.01940.x
McGarigal K (2015) Fragstats help. http://www.umass.edu/landeco/research/fragstats/documents/fragstats_documents.html. Accessed 28 March 2017
Medeiros HR, Hoshino AT, Ribeiro MC, Menezes Junior AO (2016) Landscape complexity affects cover and species richness of weeds in Brazilian agricultural environments. Basic Appl Ecol 17:731–740. https://doi.org/10.1016/j.baae.2016.10.001
Meehan TD, Werling BP, Landis DA, Gratton C (2011) Agricultural landscape simplification and insecticide use in the Midwestern United States. Proc Natl Acad Sci USA 108:11500–11505. https://doi.org/10.1073/pnas.1100751108
Mengual X (2011) Black-tie dress code: two new species of the genus Toxomerus (Diptera, Syrphidae). ZooKeys 140:1–26. https://doi.org/10.3897/zookeys.140.1930
Mengual X, Ståhls G, Rojo S (2008) First phylogeny of predatory flower flies (Diptera, Syrphidae, Syrphinae) using mitochondrial COI and nuclear 28S rRNA genes: conflict and congruence with the current tribal classification. Cladistics 24:543–562. https://doi.org/10.1111/j.1096-0031.2008.00200.x
Mengual X, Ruiz C, Rojo S, Ståhls G, Thompson FC (2009) A conspectus of the flower fly genus Allograpta (Diptera: Syrphidae) with description of a new subgenus and species. Zootaxa 2214:1–28
Meyer B, Jauker F, Steffan-Dewenter I (2009) Contrasting resource-dependent responses of flower fly richness and density to landscape structure. Basic Appl Ecol 10:178–186. https://doi.org/10.1016/j.baae.2008.01.001
Miranda GFG (2017) Revision of the Hybobathus arx and Pelecinobaccha summa species groups (Diptera: Syrphidae). Zootaxa 4338:1–43. https://doi.org/10.11646/zootaxa.4338.1.1
Miranda GFG, Marshall SA, Skevington JH (2014) Revision of the genus Pelecinobaccha Shannon, description of Relictanum gen. nov., and redescription of Atylobaccha flukiella (Curran, 1941) (Diptera: Syrphidae). Zootaxa 3819:1–154. https://doi.org/10.11646/zootaxa.3819.1.1
Gardiner MM, Landis DA, Gratton C, DiFonzo CD, O’Neal M, Chacon JM, Wayo MT, Schmidt NP, Mueller EE, Heimpel GE (2009) Landscape diversity enhances biological control of an introduced crop pest in the north-central USA. Ecol Appl 19:143–154. https://doi.org/10.1890/07-1265.1
Mueller AL, Dauber J (2016) Hoverflies (Diptera: Syrphidae) benefit from a cultivation of the bioenergy crop Silphium perfoliatum L. (Asteraceae) depending on larval feeding type, landscape composition and crop management. Agric For Entomol 18:419–431. https://doi.org/10.1111/afe.12175
Nunes-Silva P, Cordeiro GD, Obregon D, Lima-Neto JF, Thompson FC, Viana BF, Freitas BM, Kevan PG (2010) Pollenivory in larval and adult flower flies: pollen availability and visitation rate by Toxomerus politus Say (Diptera: Syrphidae) on sorghum Sorghum bicolor (L.) Moench (Poaceae). Studia Dipt 17:177–185
Oksanen J, Kindt R, Legendre P, O’Hara B, Stevens MHH (2007) The vegan package. Community ecology package. 10:631–637
Pérez-Bañón C, Rotheray G, Hancock G, Marcos-García MA, Zumbado MA (2003) Immature stages and breeding sites of some Neotropical saprophagous syrphids (Diptera: Syrphidae). Ann Entomol Soc Am 96:458–471. https://doi.org/10.1603/0013-8746(2003)096[0458:isabso]2.0.co;2
Pinheiro J, Bates D, DebRoy S, Sarkar D, R Core Team (2017) nlme: Linear and nonlinear mixed effects models. R package version 3.1-131, http://CRAN.R-project.org/package=nlme
Reemer M, Rotheray GE (2009) Pollen feeding larvae in the presumed predatory syrphine genus Toxomerus Macquart (Diptera, Syrphidae). J Nat Hist 43:939–949. https://doi.org/10.1080/00222930802610576
Resende A, Silva E, Silva V, Ribeiro R, Guerra J, Aguiar-Menezes E (2006) First record of Lipaphis pseudobrassicae Davis (Hemiptera: Aphididae) and its association with predator insects, parasitoids and ants in kale (Cruciferae) in Brazil. Neotrop Entomol 35:551–555. https://doi.org/10.1590/S1519-566X2006000400019
Ribeiro MC, Metzger JP, Martensen AC, Ponzoni FJ, Hirota MM (2009) The Brazilian Atlantic forest: how much is left, and how is the remaining forest distributed? Implications for conservation. Biol Conserv 142:1141–1153. https://doi.org/10.1016/j.biocon.2009.02.021
Ricarte A, Marcos-García MA, Moreno CE (2011) Assessing the effects of vegetation type on hoverfly (Diptera: Syrphidae) diversity in a Mediterranean landscape: implications for conservation. J Insect Conserv 15:865–877. https://doi.org/10.1007/s10841-011-9384-9
Rojo S, Gilbert F, Marcos-García MA, Nieto JM, Mier MP (2003) A world review of predatory flower flies (Diptera, Syrphidae: Syrphinae) and their prey. Cibio ediciones, Alicante
Rossetti MR, González E, Salvo A, Valladares G (2014) Not all in the same boat: trends and mechanisms in herbivory responses to forest fragmentation differ among insect guilds. Arthropod-Plant Interact 8:593–603. https://doi.org/10.1007/s11829-014-9342-z
Rotheray GE (1993) Colour guide to flower fly larvae (Diptera: Syrphidae). Whiteley, Sheffiled
Rotheray G, Gilbert F (2011) The natural history of flower flies. Forrest Text, Ceredigion
Schweiger O, Musche M, Bailey D, Billeter R, Diekötter T, Hendrickx F, Herzog F, Liira J, Maelfait JP, Speelmans M, Dziock F (2007) Functional richness of local hoverfly communities (Diptera, Syrphidae) in response to land use across temperate Europe. Oikos 116:461–472. https://doi.org/10.1111/j.2007.0030-1299.15372.x
Si X, Baselga A, Ding P (2015) Revealing beta-diversity patterns of breeding bird and lizard communities on inundated land-bridge islands by separating the turnover and nestedness components. PLoS ONE 10(5):e0127692. https://doi.org/10.1371/journal.pone.0127692
Simanonok MP, Burkle LA (2014) Partitioning interaction turnover among alpine pollination networks: spatial, temporal, and environmental patterns. Ecosphere 5:1–17. https://doi.org/10.1890/es14-00323.1
Smith H, Chaney W (2007) A survey of syrphid predators of Nasonovia ribisnigri in organic lettuce on the central coast of California. J Econ Entomol 100:39–48. https://doi.org/10.1093/jee/100.1.39
Sommaggio D (1999) Syrphidae: can they be used as environmental bioindicators? Agric Ecosyst Environ 74:343–356. https://doi.org/10.1016/s0167-8809(99)00042-0
Stapel JO, Cortesero AM, Lewis WJ (2000) Disruptive sublethal effects of insecticides on biological control: altered foraging ability and life span of a parasitoid after feeding on extrafloral nectar of cotton treated with systemic insecticides. Biol Control 17:243–249. https://doi.org/10.1006/bcon.1999.0795
Steffan-Dewenter I, Münzenberg U, Bürger C, Thies C, Tscharntke T (2002) Scale-dependent effects of landscape context on three pollinator guilds. Ecology 83:1421–1432. https://doi.org/10.1890/0012-9658(2002)083[1421:SDEOLC]2.0.CO;2
Sturza V, Dorfey C, Poncio S, Dequech S, Bolzan A (2011) First record of larvae of Allograpta exotica (Wiedemann) (Diptera, Syrphidae) preying on Aphis gossypii Glover (Hemiptera, Aphididae) in watermelon in Brazil. Rev Bras Entomol 55:272–274. https://doi.org/10.1590/S0085-56262011005000015
Thies C, Steffan-Dewenter I, Tscharntke T (2003) Effects of landscape context on herbivory and parasitism at different spatial scales. Oikos 101:18–25. https://doi.org/10.1034/j.1600-0706.2003.12567.x
Thompson FC (1999) A key to the genera the flower flies (Diptera: Syrphidae) of the Neotropical region including descriptions of new genera and species and a glossary of taxonomic terms. Contr Entomol Int 3:319–378
Thompson FC, Thompson BJ, Fairman JE (2000) Only in Costa Rica: new neotropical flower flies (Diptera: Syrphidae). Studia Dipt 7:33–43
Townes HA (1972) A light-weight Malaise trap. Entomol News 83:239–247
Tscharntke T, Klein AM, Kruess A, Steffan-Dewenter I, Thies C (2005) Landscape perspectives on agricultural intensification and biodiversity—ecosystem service management. Ecol Lett 8:857–874. https://doi.org/10.1111/j.1461-0248.2005.00782.x
Tscharntke T, Bommarco R, Clough Y, Crist TO, Kleijn D, Rand TA, Tylianakis JM, van Nouhuys S, Vidal S (2007) Conservation biological control and enemy diversity on a landscape scale. Biol Control 43:294–309. https://doi.org/10.1016/j.biocontrol.2007.08.006
Tscharntke T, Tylianakis JM, Rand TA, Didham RK, Fahrig L, Batáry P, Bengtsson J, Clough Y, Crist TO, Dormann CF, Ewers RW, Fründ J, Holt RD, Holzschuh A, Klein AM, Kleijn D, Kremen C, Landis DA, Laurance W, Lindenmayer D, Scherber C, Sodhi N, Steffan-Dewenter I, Thies C, Van Der Putten WH, Westphal C (2012) Landscape moderation of biodiversity patterns and processes—eight hypotheses. Biol Rev 87:661–685. https://doi.org/10.1111/j.1469-185X.2011.00216.x
Ulrich W, Almeida-Neto M, Gotelli NJ (2009) A consumer’s guide to nestedness analysis. Oikos 118:3–17. https://doi.org/10.1111/j.1600-0706.2008.17053.x
Whittaker RH (1965) Dominance and diversity in land plant communities. Science 147:250–260. https://doi.org/10.1126/science.147.3655.250
Yachi S, Loreau M (1999) Biodiversity and ecosystem functioning in a fluctuating environment: the insurance hypothesis. Proc Natl Acad Sci USA 96:1463–1468. https://doi.org/10.1073/pnas.96.4.1463
Zaviezo T, Grez AA, Estades CF, Perez A (2006) Effects of habitat loss, habitat fragmentation, and isolation on the density, species richness, and distribution of ladybeetles in manipulated alfalfa landscapes. Ecol Entomol 31:646–656. https://doi.org/10.1111/j.1365-2311.2006.00830.x
Zellweger F, Roth T, Bugmann H, Bollmann K (2017) Beta diversity of plants, birds and butterflies is closely associated with climate and habitat structure. Glob Ecol Biogeogr 26(8):898–906. https://doi.org/10.1111/geb.12598
Acknowledgements
We are grateful to the owners of private lands where the sampled wheat crops are located. We thank Luciane Marinoni for permission to study the Syrphidae collection of DZUP, Gil F. G. Miranda for helping with Ocyptamus sensu lato identifications and the two anonymous reviewers who helped us to substantially improve the manuscript. HRM and AHT received a research grant from the Brazilian Coordination for the Improvement of Higher Education Personnel (CAPES). MíNM was supported by CAPES (PNPD process 20131282). DWC was funded by the São Paulo Research Foundation (FAPESP—process 2014/01594-4). Brazilian Government Research Council (CNPq) provided a research Grant for MCR (312045/2013-1; 312292/2016-3), who also thanks the financial support by FAPESP (process 2013/50421-2). We thank Pavel Dodonov for the proofreading of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by P. Ponel.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Medeiros, H.R., Hoshino, A.T., Ribeiro, M.C. et al. Non-crop habitats modulate alpha and beta diversity of flower flies (Diptera, Syrphidae) in Brazilian agricultural landscapes. Biodivers Conserv 27, 1309–1326 (2018). https://doi.org/10.1007/s10531-017-1495-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-017-1495-5