Abstract
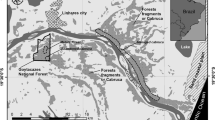
Cocoa (Theobroma cacao) is cultivated in the states of Bahia and Espírito Santo in eastern Brazil under the so-called ‘cabruca system’, where the understorey of native Atlantic forest is cleared and the canopy is thinned out to provide adequate shading for the cocoa trees. Apart from its economic and social role, the cabruca system is said to be important for the conservation of Atlantic forest biodiversity. In this paper we studied tree species richness and forest structure of cabrucas to examine the demographic health of these forests and discuss their long-term survival. Data were collected in 20 farms located alongside a 30 km track of the northern margin of the Rio Doce, in northern Espírito Santo. All trees ≥5 cm DBH were identified and their diameter was measured in 80 plots (600 m2), totalling 4.8 ha of sampled area. Recorded trees were also allocated to four different regeneration phases (pioneers, early secondary, late secondary and climax). The inventory resulted in 507 trees belonging to 105 species in 39 families. This species richness is much lower than in less disturbed forests located in the region. Pioneers and early secondary species dominate the cabruca forest in terms of number of species (56.2%), density (71.0%) and basal area (72.3%). The distribution of diameter frequency showed an imbalance in tree regeneration. Most trees in the range of 5–30 cm DBH were pioneers (40.7%), or early secondary species (32.6%), while late secondary and climax trees were less frequent (10.2 and 16.5% of the sampled trees, respectively). The dominance of species of early regeneration phases was also observed for trees >30 cm DBH (69.0% of pioneers or early secondary and 31.0% of late secondary or climax species). The results indicated that the cabruca forests are not only less diverse and less dense than secondary or primary forests of the region, but also, and more importantly, their natural succession and gap dynamics are being severely impaired. As a consequence, cabrucas present a structure where tree species of late successional phases are becoming increasingly rare while pioneers and early secondary species are becoming dominant. If current management practices of thinning and clearing of native trees are not improved, the long-term survival of these forests is questionable and their role in maintaining biodiversity in the long run is limited.
Similar content being viewed by others
References
Alger K. 1998. The Reproduction of the Cocoa Industry and Biodiversity in Southern Bahia, Brazil. Proc. 1st Int. Workshop on Sustainable Cocoa Growing, Panama, 3/30–4/2, 1998, Smithsonian Migratory Bird Center, Panama (http://www.nationalzoo.si.edu/smbc/Research/Cacao/cacao.asp).
Alves M.C. 1990. The role of cocoa plantations in the conservation of the Atlantic forest of Southern Bahia, Brazil. M.S. Thesis, University of Florida, Gainesville, Florida.
Alvim P.T. 1966. O problema do sombreamento do cacaueiro. Cacau Atualidades 3: 2–5.
Alvim R. and Nair P.K.R. 1986. Combination of cacao and eight other plantation crops: An agroforestry system in Southeast Bahia, Brazil. Agroforestry Systems 4: 3–15.
Alvim P.T. and Pereira C.P. 1965. Sombra e espaçamento nas plantações de cacau no estado da Bahia e Centro de Pesquisas do Cacau, Relatório Anual 1964, CEPLAC/CEPEC, Ilhéus, pp. 18–19.
Batista L.P. and Alvim R. 1981. Efeitos da intensidade luminosa e do fenótipo sobre o crescimento em altura do fuste do cacaueiro. Revista Theobroma 11: 61–76.
Bodmer R.E. 1991. Strategies of seed dispersal and seed predation in Amazonian ungulates. Biotropica 23: 255–261.
Budowski G. 1965. Distribution of tropical American rain forest species in the light of sucessional processes. Turrialba 15: 40–42.
Carey E.V., Brown S., Gillespie A.J.R. and Lugo A.E. 1994. Tree mortality in mature lowland tropical moist and tropical lower montane moist forests of Venezuela. Biotropica 26: 255–265.
Chiarello A.G. 1997. Mammalian community and vegetation structure of Atlantic forest fragments in south-eastern Brazil. Ph.D Thesis, University of Cambridge, Cambridge, UK.
Chiarello A.G. 1999. Effects of fragmentation of the Atlantic forest on mammal communities in south-eastern Brazil. Biological Conservation 89: 71–82.
Chiarello A.G. 2000a. Influência da caça ilegal sobre mamíferos e aves das matas de tabuleiro do norte do estado do Espírito Santo. Boletim do Museu de Biologia Mello Leitão (Nova Série) 11/12: 229–247.
Chiarello A.G. 2000b. Density and population size of mammals in remnants of Brazilian Atlantic forest. Conservation Biology 14: 1649–1657.
Cochran W.G. 1977. Sampling Techniques. John Wiley and Sons, New York.
Constanza R., D'Arge R., Groot R., Farber S., Grasso M., Hannon B. et al. 1997. The value of the world's ecosystem services and natural capital. Nature 387: 253–260.
Constanza R., D'Arge R., Groot R., Farber S., Grasso M., Hannon B. et al. 1998. The value of ecosystem services: putting the issues in perspective. Ecological Economics 25: 67–72.
Cullen Jr. L., Bodmer R.E. and Pádua C.V. 2000. Effects of hunting in habitat fragments of the Atlantic forests, Brazil. Biological Conservation 95: 49–56.
Cunningham R.K. and Burridge J.C. 1960. The growth of cacao (Theobroma cacao L.) with and without shade. Annals of Botany 24: 458–462.
Delabie J.H.C. 1988. Ocorrência de Wasmannia auropunctata (Roger, 1863) (Hymenoptera, Formicidae, Myrmicinae) em cacauais na Bahia, Brasil. Revista Theobroma 18: 29–37.
Duguma B., Gockowski J. and Bakala J. 1998. Smallholder cocoa (Theobroma cacao) cultivation in agroforestry systems of west and central Africa: challenges and opportunities. Proc. 1st Int. Workshop on Sustainable Cocoa Growing, Panama, 3/30–4/2, 1998, Smithsonian Migratory Bird Center, Panama (http://www.nationalzoo.si.edu//smbc//Research//Cacao//cacao.asp).
Ewel J.J. 1986. Designing agricultural ecosystems for the humid tropics. Annual Review of Ecology and Systematic 17: 245–271.
Fassbender H. 1993. Modelos edafológicos de sistemas agroforestales. Turrialba, CATIE, Costa Rica.
Fernandes E.N. and Vinha S.G. 1984. Recomposiçao florística do Parque Zoobotânico do Centro de Pesquisas do Cacau. Revista Theobroma 14: 1–25.
Gotelli N.J. and Colwell R.K. 2001. Quantifying biodiversity: procedures and pitfalls in the measurement and comparison of species richness. Ecology Letters 4: 379–391.
Gotelli N.J. and Entsminger G.L. 2003. EcoSim: Null models software for ecology. Version 7.0. Acquired Intelligence Inc. and Kesey-Bear, Burlington, Vermont (http://homepages.together.net/ ~ gentsmin/ecosim.htm).
Greenberg R. 1998. Biodiversity in the Cacao Agroecosystem: Shade Management and Landscape Considerations. Proc. 1st Int. Workshop on Sustainable Cocoa Growing, Panama, 3/30–4/2, 1998, Smithsonian Migratory Bird Center, Panama (http://www.nationalzoo.si.edu/smbc/Research/Cacao/cacao.asp).
Griffith D.M. 2000. Agroforestry: a refuge for tropical biodiversity after fire. Conservation Biology 14: 325–326.
Hummel M. 1995. Botanical analysis of the shade tree population in two cabruca cocoa plantations in southern Bahia, Brazil. Dissertation in Agricultural Biology, University of Stuttgart, Stuttgart, Germany.
Johns N.D. 1999. Conservation in Brazil's chocolate forest: the unlikely persistence of the traditional cocoa agroecosystem. Environmental Management 23: 31–47.
Julliot C. 1994. Frugivory and seed dispersal by red howler monkeys: evolutionary aspect. Revue d'Ecologie (Terre Vie) 49: 331–341.
Kinzey W.G. 1982. Distribution of primates and forest refuges. In: Prance G.T. (ed) Biological Diversification in the Tropics. Columbia University Press, New York, pp. 455–482.
Laurance W.F., Vasconcelos H.L. and Lovejoy T.E. 2000. Forest loss and fragmentation in the Amazon: implications for wildlife conservation. Oryx 34: 39–45.
Lieberman M., Lieberman D., Hartshorn G.S. and Peralta R. 1985. Small-scale altitudinal variation in lowland wet tropical forest vegetation. Journal of Ecology 73: 505–516.
Manokaram N. and Kochummen K.M. 1987. Recruitment, growth and mortality of tree species in a lowland dipterocarp forest in Peninsular Malaysia. Journal of Tropical Ecology 3: 315–330.
Montagnini F. 1992. Sistemas agroforestales: principios y aplicaciones en los trópicos. Organización para Estudios Tropicales (OET), San José, Costa Rica.
Mori S.A., Boom B.M., Carvalho A.M. and Santos T.S. 1983. Southern Bahian moist forests. The Botanical Review 49: 155–232.
Moura R.T. 1999. Análise Comparativa da Estrutura da Comunidade de Pequenos Mamíferos em Remanescentes da Mata Atlântica e Plantio de Cacau em Sistema de Cabruca no Sul da Bahia. M.Sc. Thesis, Universidade Federal de Minas Gerais, Belo Horizonte, Brazil.
Mueller-Dombois D. and Ellenberg H. 1974. Aims and Methods of Vegetation Ecology. John Wiley, New York.
Myers N. 1986. Forestland farming in western Amazonia: stable and sustainable. Forest Ecology and Management 15: 81–93.
Myers N. 1989. Deforestation Rates in Tropical Forests and their Climate Implications. Friends of the Earth, London.
Nair P.K.R. (ed) 1989. Agroforestry Systems in the Tropics. Kluwer Academic Publishers, Dordrecht, The Netherlands.
Pardini R. 2001. Pequenos mamíferos e a fragmentação da Mata Atlântica de Una, sul da Bahia-processos e conservação. Ph.D. Thesis, Universidade de São Paulo, São Paulo, Brazil.
Parrish J., Reitsma R. and Greenberg G. 1998. Cacao as crop and conservation tool. Lessons from the Talamanca region of Costa Rica. Proc. 1st International Workshop on Sustainable Cocoa Growing, Panama, 3/30–4/2, 1998, Smithsonian Migratory Bird Center, Panama (http://www.nationalzoo. si.edu/smbc/Research/Cacao/cacao.asp).
Peixoto A.L. and Gentry A. 1990. Diversidade e composição florística da Mata de Tabuleiro na reserva florestal de Linhares (Espírito Santo, Brasil). Revista Brasileira de Botânica 13: 19–25.
Peixoto A.L. and Silva I.M. 1997. Tabuleiro forests of northern Espírito Santo, southeastern Brazil. In: Davis S.D., Heywood V.H., Herrera-Macbryde O., Villa-Lobos J. and Hamilton A.C. (eds) Centres of Plant Diversity: A Guide and Strategy for their Conservation. The Americas, WWF and IUCN, London, pp. 369–372.
Peres C.A. 2000. Effects of subsistence hunting on vertebrate community structure in Amazonian forest. Conservation Biology 14: 240–253.
Phillips O.L. 1997. The changing ecology of tropical forests. Biodiversity and Conservation 6: 291–311.
Power A.G. and Flecker A.S. 1998. Agroecosystems and biodiversity. Proc. 1st Int. Workshop on Sustainable Cocoa Growing, Panama, 3/30–4/2, 1998, Smithsonian Migratory Bird Center, Panama (http://www.nationalzoo.si.edu/smbc/Research/Cacao/cacao.asp).
Prance G.T. 1987. Biogeography of neotropical plants. In: Whitmore T.C. and Prance G.T. (eds) Biogeography and Quaternary History in Tropical America. Clarendon Press, Oxford, UK, pp. 46–65.
Redford K.H. 1992. The empty forest. Bioscience 42: 412–422.
Reice S.R. 1994. Nonequilibrium determinants of biological community structure. American Scientist 82: 424–435.
Rizzini C.M., Aduan R.E., Jesus R.M. and Garay I. 1997. Floresta pluvial de tabuleiro, Linhares, ES, Brasil, sistemas primários e secundários. Leandra 12: 54–76.
Rolim S.G. and Nascimento H.E.M. 1997. Richness, diversity and species-abundance relationships of a tropical tree community in different samplings. Scientia Forestalis 52: 7–16.
Rolim S.G., Couto H.T.Z. and Jesus R.M. 2001. Fluctuaciones temporales en la composición florística del bosque tropical atlántico. Biotropica 33: 12–22.
Saatchi S., Agosti D., Alger K., Delabie J. and Musinski J. 2001. Examining fragmentation and loss of primary forest in the southern Bahian Atlantic forest of Brazil with radar imagery. Conservation Biology 15: 867–875.
Sambuichi R.H.R. 2002. Fitossociologia e diversidade de espécies arbó reas em cabruca (mata atlântica raleada sobre plantação de cacau) na região sul da Bahia, Brasil. Acta Botânica Brasilica 16: 89–101.
Sena Gomes A.R. 1992. Agrosilvicultural systems in southeastern Bahia. In: Anais do II Encontro Brasileiro de Economia e Planejamento Florestal, 30/09 a 04/10/1991. EMBRAPA/CNP Floresta. Curitiba, Paraná, pp. 109–122.
Silva J.M.C. and Tabarelli M. 2000. Tree species impoverishment and the future flora of the Atlantic forest northeast Brazil. Nature 404: 72–74.
Simberloff D. 1978. Use of rarefaction and related methods in ecology. In: Dickson K.L., Cairns Jr. J. and Livingston R.J. (eds) Biological Data in Water Pollution Assessment: Quantitative and Statistical Analyses, ASTM STP 652. American Society for Testing and Materials, Philadelphia, Pennsylvania, pp. 150–165.
Suguio K., Martin L. and Dominguez J.L.M. 1982. Evolução da planície costeira do Rio Doce (ES) durante o quaternário: Influencia das flutuaçoes do nível do mar. In: Suguio K., De Meis M.R.M. and Tessler M.G. (eds) Anais do IV Simpósio do Quaternário no Brasil. Rio de Janeiro, Brazil, pp. 93–116.
Swaine M.D., Hall J.B. and Alexander I.J. 1987. Tree population dynamics at Kade, Ghana (1968–1982). Journal of Tropical Ecology 3: 331–345.
Tabarelli M. and Mantovani W. 1996. Remoção de sementes de Bertholletia excelsa (Lecythidaceae) por animais em uma floresta de terra firme na Amazônia central, Brasil. Revista Brasileira de Biologia 56: 755–760.
Terborgh J. 1992. Maintenance of diversity in tropical forest. Biotropica 24: 283–292.
Thomas W.W. and Carvalho A.M.V. 1997. Atlantic moist forest of Southern Bahia, south-eastern Brazil. In: Davis S.D., Heywood V.H., Herrera-Macbryde O., Villa-Lobos J. and Hamilton A.C. (eds), Centres of Plant Diversity: A Guide and Strategy for their Conservation, The Americas. WWF and IUCN, London, pp. 364–368.
Thomas W.W., Carvalho A.M.V., Amorim A.M.A., Garrison J. and Arbeláez A.L. 1998. Plant endemism in two forests in southern Bahia, Brazil. Biodiversity and Conservation 7: 311–322.
Vinha S.G., Silva L.A.M., Carvalho A.M., Pereira R.C. and Reyes-Zumeta H. 1983. Plantas Herbáceas, Epífitas, Arbustivas e Trepadeiras Associadas à Cultura do Cacaueiro. Centro de Pesquisas do Cacau, Itabuna, Ilhéus, Bahia, Brazil.
Whitmore T.C. and Sayer J.A. 1992. Tropical Deforestation and Species Extinction. Chapman & Hall, London.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rolim, S.G., Chiarello, A.G. Slow death of Atlantic forest trees in cocoa agroforestry in southeastern Brazil. Biodiversity and Conservation 13, 2679–2694 (2004). https://doi.org/10.1007/s10531-004-2142-5
Issue Date:
DOI: https://doi.org/10.1007/s10531-004-2142-5