Abstract
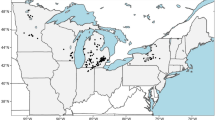
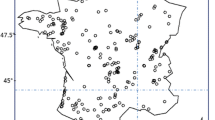
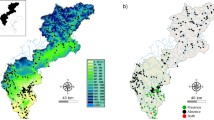
Bythotrephes longimanus is an invasive pelagic crustacean, which first arrived in North America from Europe in early 1980s and can now be found throughout the Great Lakes and in many inland lakes and waterways. Determining the suitability of lakes to Bythotrephes establishment is an important step in quantifying its potential habitat range and environmental risk. Lake environmental conditions, planktivorous fishes, sport fishes and Bythotrephes occurrence data from 179 south-central Ontario lakes were used in this study to model lake characteristics suitable for its establishment. The performance of principal component analysis and different predictive models was used to determine the habitats that are suitable for the survival of Bythotrephes and the factors that may regulate its spread. Four modeling approaches were employed: linear discriminant analysis; multiple logistic regression; random forests; and, artificial neural networks. Ensemble prediction based on the four modeling approaches was also used as an indicator for predicting Bythotrephes occurrence. Bythotrephes appears to establish more readily in larger, deeper lakes with lower elevation, that have more sport fishes. Bythotrephes occurrence can be best predicted by artificial neural networks when including the measures of fish data, in addition to lake environmental data. Lake elevation, surface area and sport fish occurrence were ranked as the most important predictors of Bythotrephes invasion. The inclusion of biotic variables (occurrence or diversity of sport or planktivorous fishes) enhanced cross-validated models relative to analyses based on environmental data alone.


Similar content being viewed by others
References
Barnhisel DR, Kerfoot WC (2004) Fitting into food webs: behavioral and functional response of young lake trout (Salvelinus namaycush) to an introduced prey, the spiny cladoceran (Bythotrephes cederstroemi). J Great Lakes Res 30:300–314
Branstrator DK, Brown ME, Shannon LJ, Thabes M, Heimgartner K (2006) Range expansion of Bythotrephes longimanus in North America: evaluating habitat characteristics in the spread of an exotic zooplankter. Biol Invasions 8:1367–1379
Buisson L, Thuiller W, Lek S, Lim P, Grenouillet G (2008) Climate change hastens the turnover of stream fish assemblages. Glob Chang Biol 14:2232–2248
Carlton JT (1985) Transoceanic and interoceanic dispersal of coastal marine organisms: the biology of ballast water. Oceanogr Mar Biol Annu Rev 23:313–371
Carlton JT, Geller JB (1993) Ecological roulette: the global transport of nonindigenous marine organisms. Science (Washington, D.C.) 261:78–82
Chatfield C (1995) Model uncertainty, data mining and statistical inference. J R Stat Soc Ser A Gen 158:419–466
Compton JA, Kerfoot WC (2004) Colonizing inland lakes: consequences of YOY fish ingesting the spiny cladoceran (Bythotrephes cederstroemi). J Great Lakes Res 30:315–326
De’ath G (2007) Boosted trees for ecological modeling and prediction. Ecology 88:243–251
Elith J, Graham CH, Anderson RP, Dudík M, Ferrier S, Guisan A, Hijmans RJ, Huettmann F, Leathwick JR, Lehmann A, Li J, Lohmann LG, Loiselle BA, Manion G, Moritz C, Nakamura M, Nakazawa Y, Overton JMcC, Peterson AT, Phillips SJ, Richardson K, Scachetti-Pereira R, Schapire RE, Soberón J, Williams S, Wisz MS, Zimmermann NE (2006) Novel methods improve prediction of species’ distributions from occurrence data. Ecography 29:129–151
Garson GD (1991) Interpreting neural-network connection weights. Artif Intell Expert 6:46–51
Goh ATC (1995) Back-propagation neural networks for modeling complex systems. Artif Intell Eng 9:143–151
Guisan A, Thuiller W (2005) Predicting species distribution: offering more than simple habitat models. Ecol Lett 8:993–1009
Guisan A, Zimmermann NE (2000) Predictive habitat distribution models in ecology. Ecol Model 135:147–186
Iverson LR, Prasad AM, Liaw A (2004) New machine learning tools for predictive vegetation mapping after climate change: bagging and random forest perform better than regression tree analysis. In: Smithers R (ed) Landscape ecology of trees and forests. Proceedings of the 12th Annual Conference of the IALE (UK). International Association for Landscape Ecology, UK, pp 317–320
Jackson DA (1993) Stopping rules in principal components analysis: a comparison of heuristical and statistical approaches. Ecology 74:2204–2214
Jackson DA (2002) Ecological impacts of Micropterus introductions: the dark side of black bass. In: Phillip D, Ridgway M (eds) Black bass: ecology, conservation and management. American Fisheries Society, Bethesda, pp 221–234
Jackson DA, Somers KM, Harvey HH (1989) Similarity coefficient: measures of co-occurrence and association or simply measures of occurrence? Am Nat 133:436–453
Knowler D, Barbier E (2005) Importing exotic plants and the risk of invasion: are market-based instruments adequate? Ecol Econ 52:341–354
Lodge DM (1993) Biological invasions: lessons for ecology. Trends Ecol Evol 8:133–137
MacIsaac HJ, Ketelaars HAM, Grigorovich I, Ramcharan C, Yan ND (2000) Modeling Bythotrephes longimanus invasions in the Great Lakes basin based on its European distribution. Arch Hydrobiol. 149:1–21
MacIsaac HJ, Borbely J, Muirhead J, Graniero P (2004) Backcasting and forecasting biological invasions of inland lakes. Ecol Appl 14:773–783
McCune B, Grace JB (2002) Analysis of ecological communities. MjM software design. Gleneden Beach, Oregon
Olden JD, Jackson DA (2000) Torturing the data for the sake of generality: how valid are our regression models? Ecoscience 7:501–510
Olden JD, Jackson DA (2002) Illuminating the “black box”: understanding variable contributions in artificial neural networks. Ecol Model 154:135–150
Olden JD, Jackson DA, Peres-Neto PR (2002) Predictive models of fish species distributions: a note on proper validation and chance predictions. Trans Am Fish Soc 131:329–336
Özesmi SL, Tan CO, Özesmi U (2006) Methodological issues in building, training, and testing artificial neural networks in ecological applications. Ecol Model 195:83–93
Pearce J, Ferrier S (2000) Evaluating the predictive performance of habitat models developed using logistic regression. Ecol Model 133:225–245
Phillips SJ, Anderson RP, Schapire RE (2006) Maximum entropy modeling of species geographic distributions. Ecol Model 190:231–259
Prasad AM, Iverson LR, Liaw A (2006) Newer classification and regression tree techniques: bagging and random forests for ecological prediction. Ecosystems 9:181–199
R Development Core Team (2010) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. http://www.R-project.org
Reichard SH, White P (2003) Invasion biology: an emerging field of study. Ann Mo Bot Gard 90:64–66
Ricciardi A, Rasmussen JB (1998) Predicting the identity and impact of future biological invaders: a priority for aquatic resource management. Can J Fish Aquat Sci 55:1759–1765
Rumelhart DE, Hinton GE, Williams RJ (1986) Learning representations by back-propagating errors. Nature 323:533–536
Sharma S, Jackson DA (2008) Predicting smallmouth bass (Micropterus dolomieu) occurrence across North America under climate change: a comparison of statistical approaches. Can J Fish Aquat Sci 65:471–481
Strecker AL, Arnott SE (2005) Impact of Bythotrephes invasion on zooplankton communities in acid-damaged and recovered lakes on the Boreal Shield. Can J Fish Aquat Sci 62:2450–2462
Strecker AL, Arnott SE (2008) Invasive predator, Bythotrephes, has varied effects on ecosystem function in freshwater lakes. Ecosystems 11:490–503
Strecker AL, Arnott SE, Yan ND, Girard R (2006) Variation in the response of crustacean zooplankton species richness and composition to the invasive predator Bythotrephes longimanus. Can J Fish Aquat Sci 63:2126–2136
Turpie JK, Heydenrych BJ, Lamberth SJ (2003) Economic value of terrestrial and marine biodiversity in the Cape Floristic Region: implications for defining effective and socially optimal conservation strategies. Biol Conserv 112:233–251
Vander Zanden MJ, Essington TE, Vadeboncoeur Y (2005) Is pelagic top-down control in lakes augmented by benthic energy pathways? Can J Fish Aquat Sci 62:1422–1431
Walker LR, Landau FH, Velazquez E, Shiels AB, Sparrow AD (2010) Early successional woody plants facilitate and ferns inhibit forest development on Puerto Rican landslides. J Ecol 98:625–635
Warrens MJ (2008) On association coefficients for 2 × 2 tables and properties that do not depend on the marginal distributions. Psychometrika 73:777–789
Weisz EJ, Yan ND (2010) Relative value of limnological, geographic, and human use variables as predictors of the presence of Bythotrephes longimanus in Canadian Shield lakes. Can J Fish Aquat Sci 67:462–472
Wilcove DS, Rothstein D, Dubow J, Phillips A, Losos E (1998) Quantifying threats to imperiled species in the United States: assessing the relative importance of habitat destruction, alien species, pollution, overexploitation, and disease. Bioscience 48:607–615
With KA (2002) The landscape ecology of invasive spread. Conserv Biol 16:1192–1203
Yan ND, Dunlop WI, Pawson TW, MacKay LE (1992) Bythotrephes cederstroemi (Schoedler) in Muskoka lakes: first records of the European invader in inland lakes in Canada. Can J Fish Aquat Sci 49:422–426
Yan ND, Blukacz A, Sprules WG, Kindy PK, Hackett D, Girard R, Clark BJ (2001) Changes in the zooplankton and the phenology of the spiny water flea, Bythotrephes, following its invasion of Harp Lake, Ontario, Canada. Can J Fish Aquat Sci 58:2341–2350
Young JD, Loew ER, Yan ND (2009) Examination of direct daytime predation by Coregonus artedi on Bythotrephes longimanus in Harp Lake, Ontario, Canada: no evidence for the refuge hypothesis. Can J Fish Aquat Sci 68:449–459
Acknowledgments
We would like to thank NSERC, CAISN and the various research funding partners for facilitating this research. We thank Norman Yan for leading the field sampling program, for providing the data, and for comments on earlier presentations of this work. We also thank the Ontario Ministry of Natural Resources for providing fish composition data.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, L., Jackson, D.A. Modeling the establishment of invasive species: habitat and biotic interactions influencing the establishment of Bythotrephes longimanus . Biol Invasions 13, 2499–2512 (2011). https://doi.org/10.1007/s10530-011-0071-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-011-0071-2