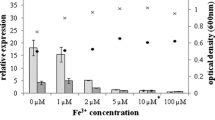
Abstract
Objective
To construct Pseudomonas aeruginosa PA14 derivatives that overproduce rhamnolipids (RL) by blocking the synthesis of the carbon-storage polymer polyhydroxyalkanoates (PHA) and by overexpressing the rhlAB-R operon that encodes for enzymes of RL synthesis and the RhlR transcriptional regulator.
Results
In contrast to previous results showing that overexpression of rhlAB-R genes in two P. aeruginosa strains (PAO1 and ATCC 9027) is sufficient to overproduce RL, we show that a PA14 derivative overexpressing the rhlAB-R operon did not increase the synthesis of these biosurfactants. In addition, PA14 mutants deficient in PHA production did not overproduce RL either. However, if the rhlAB-R genes were expressed in a mutant that is completely impaired in PHA synthesis, a significant increase in RL production was observed (59%). These results show that RL production in PA14 is limited both by the availability of fatty acid precursors and by the levels of the RhlA and RhlB enzymes that are involved in the synthesis of mono-RL.
Conclusions
The limitation of RL production by P. aeruginosa PA14 is multifactorial and diverse from the results obtained with other strains. Thus, the factors that limit RL production are particular to each P. aeruginosa strain, so strain-specific strategies should be developed to increase their production.


Similar content being viewed by others
References
Abdel-Mawgoud AM, Lépine F, Déziel E (2014) A stereospecific pathway diverts β-oxidation intermediates to the biosynthesis of rhamnolipids biosurfactants. Chem Biol 21:1–9
Banat I, Franzetti A, Bestetti G (2010) Microbial biosurfactants production, applications and future potential. Appl Microbiol Biotechnol 87:427–444
Berger E, Ramsay BA, Ramsay JA, Chavarie C (1989) PHB recovery by hypochlorite digestion of non-PHB biomass. Biotechnol Tech 3(4):227–232
Chandrasekaran EV, Bemiller JN (1980) Constituent analyses of glycosaminoglycans. Methods Carbohydr Chem 8:89–96
Croda-García G, Grosso-Becerra V, González A et al (2011) Transcriptional regulation of Pseudomonas aeruginosa rhlR: role of the Crp-ortholog Vfr (virulence factor regulator) and quorum-sensing regulators LasR and RhlR. Microbiology 157(9):2545–2555
Dobler L, Vilella LF, Almeida RC, Neves BC (2016) Rhamnolipids in perspective: gene regulation pathways, metabolic engineering, production and technological forecasting. New Biotechnol 33(1):123–133
Gellatly SL, Hancock REW (2013) Pseudomonas aeruginosa: new insights into pathogenesis and host defenses. Pathog Dis 67:159–173
Grosso-Becerra MV, Croda-García G, Merino E, Servín-González L et al (2014) Regulation of Pseudomonas aeruginosa virulence factors by two novel RNA thermometers. Proc Natl Acad Sci USA 111(43):15562–15567
Grosso-Becerra MV, González-Valdez A, Granados-Martínez MJ et al (2016) Pseudomonas aeruginosa ATCC 9027 is a non-virulent strain suitable for mono-rhamnolipids production. Appl Microbiol Biotech 100(23):9995–10004
Gust B, Challis GL, Fowler K et al (2003) PCR-targeted Streptomyces gene replacement identifies a protein domain needed for biosynthesis of the sesquiterpene soil odor geosmin. Proc Natl Acad Sci USA 100(4):1541–1546
Gutierrez M, Choi MH, Tian B et al (2013) Simultaneous inhibition of rhamnolipids and polyhydroxyalkanoic acid synthesis and biofilm formation by 2-bromoalkanoic acids: effect of inhibitor alkyl-chain length. PLoS ONE 8(9):e73986. https://doi.org/10.1371/journal.pone.0073986
Hoffmann N, Steinbüchel A, Rehm B (2000) The Pseudomonas aeruginosa phaG gene product is involved in the synthesis of polyhydroxyalkanoic acid consisting of medium-chain-length from non-related carbon sources. FEMS Microbiol Lett 184(2):253–259
Lee DG, Urbach JM, Wu G et al (2006) Genomic analysis reveals that Pseudomonas aeruginosa virulence is combinatorial. Genome Biol 7:R90
Lesic B, Rahme LG (2008) Use of the lambda Red recombinase system to rapidly generate mutants in Pseudomonas aeruginosa. BMC Biol Mol 9:20–28
Liberati NT, Urbach JM, Miyada S et al (2006) An ordered non-redundant library of Pseudomonas aeruginosa strain PA14 transposon insertion mutants. Proc Natl Acad Sci USA 103(8):2833–2838
Matsuyama T, Sogawa M, Yano I (1987) Direct colony thin-layer chromatography and rapid characterization of Serratia marscescens wetting agents. Appl Environ Microbiol 53:1186–1188
Müller MM, Hörmann B, Kugel M, Syldark C, Hausmann R (2011) Evaluation of rhamnolipid production capacity of Pseudomonas aeruginosa PAO1 in comparison to the rhamnolipids overproducing strains DSM2874. Appl Microbiol Biotechnol 89:585–592
Nitschke M, Costa SGVAO, Contiero J (2011) Rhamnolipids and PHAs: recent reports on Pseudomonas-derived molecules of increasing industrial interest. Process Biochem 46:621–630
Slepecky RA, Law JH (1960) A rapid spectophometric assay of alpha, beta-unsaturated acids and beta-hydroxy acids. Anal Chem 32:1697–1699
Soberón-Chávez G, Aguirre-Ramírez M, Sánchez R (2005a) The Pseudomonas aeruginosa RhlA enzyme is not only involved in rhamnolipid, but also in polyhydroxyalkanoate production. J Ind Microbiol Biotechnol 32:675–677
Soberón-Chávez G, Lépine F, Déziel E (2005b) Production of rhamnolipids by Pseudomonas aeruginosa. Appl Microbiol Biotechnol 68:718–725
West SEH, Schweizer HP, Dall C et al (1994) Construction of improved Escherichia-Pseudomonas shuttle vectors derived from pUC18/19 and sequence of the region required for their replication in Pseudomonas aeruginosa. Gene 148:81–86
Williams P, Cámara M (2009) Quorum sensing and environmental adaptation in Pseudomonas aeruginosa: a tale of regulatory networks and multifunctional signal molecules. Curr Opin Microbiol 12:182–191
Wittgens A, Tiso T, Arndt TT et al (2011) Growth independent rhamnolipid production from glucose using the non-pathogenic Pseudomonas putida KT2440. Microbial Cell Fact 10:80
Zhang Y, Miller RM (1992) Enhanced octadecane dispersion and biodegradation by a Pseudomonas rhamnolipid surfactant (biosurfactant). Appl Environ Microbiol 58:3276–3282
Acknowledgements
UGG and MPSA are doctoral student of Programa de Maestría y Doctorado en Ciencias Bioquímicas, Universidad Nacional Autónoma de México (UNAM), this study was performed in partial fulfillment of the requirements for UGG doctorate degree. UGG (CVU-422007) and MPSA (CVU-741217) received a fellowship from CONACYT. We acknowledge Abigail González-Valdez for technical assistance. This work was supported in part by grant IN200416 from Programa de Apoyo a Proyectos de Investigación e Innovación Tecnológica, (Dirección General de Asuntos del Personal Académico -UNAM) and grant 252269 from Consejo Nacional de Ciencia y Tecnología (CONACYT).
Supporting information
Supplementary Fig. 1—shows the standard curve use to determine PHA weight from absorbance at 235 nm.
Supplementary Fig. 2—presents the PA14 strain growth curve in PPGAS medium.
Supplementary Fig. 3—shows the PHA production of PA14 derivatives carrying mutations in genes involved in the synthesis of this fatty acid polymer, and the experiments showing that the phaC1 gene of the phaC2::MAR2xT7 mutant (ID 54085) is not functional since it is unable to complement the phaC1::MAR2xT7 mutant (ID 32531) for PHA synthesis.
Supplementary Table S1—shows the oligonucleotides used to construct the PA14 ΔphaG::aac(3)IV mutant and the pUCP20 derived plasmids expressing phaC1wt and phaC1m.
Supplementary Table S2—shows the results obtained from the analysis of the image of thin-layer chromatography presented in Fig. 2, using the ImageJ software.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gutiérrez-Gómez, U., Soto-Aceves, M.P., Servín-González, L. et al. Overproduction of rhamnolipids in Pseudomonas aeruginosa PA14 by redirection of the carbon flux from polyhydroxyalkanoate synthesis and overexpression of the rhlAB-R operon. Biotechnol Lett 40, 1561–1566 (2018). https://doi.org/10.1007/s10529-018-2610-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-018-2610-8