Abstract
Objective
To promote targeting specificity of anti-CD47 agents, we have constructed a novel bispecific antibody fusion protein against EGFR and CD47, which may minimize the “off-target” effects caused by CD47 expression on red blood cells.
Results
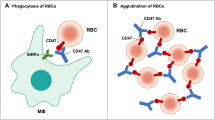
The novel bispecific antibody fusion protein, denoted as Bi-SP could simultaneously bind to EGFR and CD47 and exhibited potent phagocytosis-stimulation effects in vitro. Bi-SP treatment with a low dose more effectively inhibited tumor growth than either EGFR-targeting antibody, Pan or the SIRPα variant-Fc (SIRPαV-Fc) in the A431 xenograft tumor model. In addition, the treatment with Bi-SP produced less red blood cell (RBC) losses than the SIRPαV-Fc treatment, suggesting its potential use for minimizing RBC toxicity in therapy.
Conclusions
Bi-SP with improved therapeutic index has the potential to treat CD47+ and EGFR+ cancers in clinics.




Similar content being viewed by others
References
Chao MP et al (2010) Anti-CD47 antibody synergizes with rituximab to promote phagocytosis and eradicate non-Hodgkin lymphoma. Cell 142:699–713. https://doi.org/10.1016/j.cell.2010.07.044
Dheilly E et al (2017) Selective blockade of the ubiquitous checkpoint receptor CD47 is enabled by dual-targeting bispecific antibodies. Mol Ther 25:523–533. https://doi.org/10.1016/j.ymthe.2016.11.006
Fauvel B, Yasri A (2014) Antibodies directed against receptor tyrosine kinases: current and future strategies to fight cancer MAbs 6:838–851. https://doi.org/10.4161/mabs.29089
Huang Y, Ma Y, Gao P, Yao Z (2017) Targeting CD47: the achievements and concerns of current studies on cancer immunotherapy. J Thorac Dis 9:E168–E174. https://doi.org/10.21037/jtd.2017.02.30
Junutula JR et al (2010) Engineered thio-trastuzumab-DM1 conjugate with an improved therapeutic index to target human epidermal growth factor receptor 2-positive breast cancer. Clin Cancer Res 16:4769–4778. https://doi.org/10.1158/1078-0432.CCR-10-0987
Khandelwal S, van Rooijen N, Saxena RK (2007) Reduced expression of CD47 during murine red blood cell (RBC) senescence and its role in RBC clearance from the circulation. Transfusion 47:1725–1732. https://doi.org/10.1111/j.1537-2995.2007.01348.x
Kwong LS, Brown MH, Barclay AN, Hatherley D (2014) Signal-regulatory protein alpha from the NOD mouse binds human CD47 with an exceptionally high affinity– implications for engraftment of human cells. Immunology 143:61–67. https://doi.org/10.1111/imm.12290
Liu B et al. (2017) Elimination of tumor by CD47/PD-L1 dual-targeting fusion protein that engages innate and adaptive immune responses. MAbs 1–10. https://doi.org/10.1080/19420862.2017.1409319
Liu X et al (2015) CD47 blockade triggers T cell-mediated destruction of immunogenic tumors. Nat Med 21:1209–1215. https://doi.org/10.1038/nm.3931
Matozaki T, Murata Y, Okazawa H, Ohnishi H (2009) Functions and molecular mechanisms of the CD47-SIRPalpha signalling pathway. Trends Cell Biol 19:72–80. https://doi.org/10.1016/j.tcb.2008.12.001
Peipp M, Dechant M, Valerius T (2008) Effector mechanisms of therapeutic antibodies against ErbB receptors. Curr Opin Immunol 20:436–443. https://doi.org/10.1016/j.coi.2008.05.012
Piccione EC et al. (2015) A bispecific antibody targeting CD47 and CD20 selectively binds and eliminates dual antigen expressing lymphoma cells. MAbs 7:946–956. https://doi.org/10.1080/19420862.2015.1062192
Shatz W et al (2013) Knobs-into-holes antibody production in mammalian cell lines reveals that asymmetric afucosylation is sufficient for full antibody-dependent cellular cytotoxicity. MAbs 5:872–881. https://doi.org/10.4161/mabs.26307
Watanabe M, Wallace PK, Keler T, Deo YM, Akewanlop C, Hayes DF (1999) Antibody dependent cellular phagocytosis (ADCP) and antibody dependent cellular cytotoxicity (ADCC) of breast cancer cells mediated by bispecific antibody, MDX-210. Breast Cancer Res Treat 53:199–207
Weiskopf K, Weissman IL (2015) Macrophages are critical effectors of antibody therapies for cancer. MAbs 7:303–310. https://doi.org/10.1080/19420862.2015.1011450
Weiskopf K et al (2013) Engineered SIRPalpha variants as immunotherapeutic adjuvants to anticancer antibodies. Science 341:88–91. https://doi.org/10.1126/science.1238856
Wiersma VR et al (2014) A CD47-blocking TRAIL fusion protein with dual pro-phagocytic and pro-apoptotic anticancer activity. Br J Haematol 164:304–307. https://doi.org/10.1111/bjh.12617
Willingham SB et al (2012) The CD47-signal regulatory protein alpha (SIRPa) interaction is a therapeutic target for human solid tumors. Proc Natl Acad Sci USA 109:6662–6667. https://doi.org/10.1073/pnas.1121623109
Xiao Z et al (2015) Antibody mediated therapy targeting CD47 inhibits tumor progression of hepatocellular carcinoma. Cancer Lett 360:302–309. https://doi.org/10.1016/j.canlet.2015.02.036
Yang Y et al (2015) Generation and characterization of a target-selectively activated antibody against epidermal growth factor receptor with enhanced anti-tumor potency. MAbs 7:440–450. https://doi.org/10.1080/19420862.2015.1008352
Yang Y, Guo R, Tian X, Zhang Z, Zhang P, Li C, Feng Z (2017) Synergistic anti-tumor activity of Nimotuzumab in combination with Trastuzumab in HER2-positive breast cancer. Biochem Biophys Res Commun 489:523–527. https://doi.org/10.1016/j.bbrc.2017.06.001
Acknowledgements
We thank Binfeng Cheng, Huaizu Guo and Jin Xu for their expert technical assistance. This work was supported by grants from the Natural Science Foundation of China (81703054, 81403161, 21605074), National Training Program of Innovation for Undergraduates (201710472029), Key scientific research project of higher education of Henan Province, China (17A350012), Key Science and Technology Program of Henan Province, China (172102310614), Doctoral Foundation of Xinxiang Medical University (XYBSKYZZ201506).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yang, Y., Guo, R., Chen, Q. et al. A novel bispecific antibody fusion protein co-targeting EGFR and CD47 with enhanced therapeutic index. Biotechnol Lett 40, 789–795 (2018). https://doi.org/10.1007/s10529-018-2535-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-018-2535-2