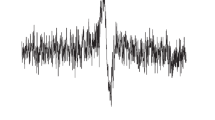
We studied the effect of dinitrosyl-iron complexes with N-acetyl-L-cysteine as a thiol-containing ligand (DNIC-Acc) after transdermal administration to rats. Electron paramagnetic resonance spectroscopy with a lipophilic NO spin trap (a complex of iron and diethyldithiocarbamate ions) showed that DNIC-Acc administration significantly increased the total level of NO in the lung and liver tissues of the animal, which was accompanied by a slight decrease in the mean BP (<10%).
Similar content being viewed by others
References
Timoshin AA, Drobotova DY, Tskitishvili OV, Serebryakova LI, Pisarenko OI, Ruuge EK, Vanin AF. Protective effect of dinitrosyl-iron complexes with glutathione in rat myocardial regional ischemia: a microdialysis assay study. Dokl. Biochem. Biophysics. 2010;432(1):106-109.
Timoshin AA, Lakomkin VL, Abramov AA, Ruuge EK, Vanin AF. The biological effect of dinitrosyl iron complexes with glutathione upon nitric oxide hyperproduction induced by endotoxin shock. Biophysics. 2019;64(1):89-94.
Timoshin AA, Lakomkin VL, Abramov AA, Ruuge EK, Vanin AF. Hypotensive Effect and Accumulation of Dinitrosyl Iron Complexes in Blood and Tissues after Intravenous and Subcutaneous Injection. Bull. Exp. Biol. Med. 2016;162(2):207-210. doi: https://doi.org/10.1007/s10517-016-3577-x.
Chen YJ, Ku WC, Feng LT, Tsai ML, Hsieh CH, Hsu WH, Liaw WF, Hung CH, Chen YJ. Nitric Oxide Physiological Responses and Delivery Mechanisms Probed by Water-Soluble Roussin’s Red Ester and {Fe(NO)2}10 DNIC. J. Am. Chem. Soc. 2008;130(33):10 929-10 938. doi: https://doi.org/10.1021/ja711494m
Ke CH, Chen CH, Tsai ML, Wang HC, Tsai FT, Chiang YW, Shih WC, Bohle DS, Liaw WF. {Fe(NO)(2)(9) Dinitrosyl Iron Complex Acting as a Vehicle for the NO Radical. J. Am. Chem. Soc. 2017;139(1):67-70. doi: https://doi.org/10.1021/jacs.6b11454
Rahmanto YS, Kalinowski DS, Lane DJR, Lok HC, Richardson V, Richardson DR. Nitrogen Monoxide (NO) Storage and Transport by Dinitrosyl-Dithiol-Iron Complexes: Long-Lived NO That Is Trafficked by Interacting Proteins. J. Biol. Chem. 2012;287(10):6960-6968. doi: https://doi.org/10.1074/jbc.R111.329847
Sen CK. Nutritional biochemistry of cellular glutathione. J. Nutr. Biochem. 1997;8(12):660-672.
Shumaev KB, Dudylina AL, Ivanova MV, Pugachenko IS, Ruuge EK. Dinitrosyl Iron Complexes: Formation and Antiradical Action in Heart Mitochondria. Biofactors. 2018;44(3):237-244. doi: https://doi.org/10.1002/biof.1418
Shumaev KB, Gubkin AA, Serezhenkov VA, Lobysheva II, Kosmachevskaya OV, Ruuge EK, Lankin VZ, Topunov AF, Vanin AF. Interaction of Reactive Oxygen and Nitrogen Species With Albumin- And Methemoglobin-Bound Dinitrosyl-Iron Complexes. Nitric Oxide. 2008;18(1):37-46. doi: https://doi.org/10.1016/j.niox.2007.09.085
Støttrup NB, Løfgren B, Birkler RD, Nielsen JM, Wang L, Caldarone CA, Kristiansen SB, Contractor H, Johannsen M, Bøtker HE, Nielsen TT. Inhibition of the Malate-Aspartate Shuttle by Pre-Ischaemic Aminooxyacetate Loading of the Heart Induces Cardioprotection. Cardiovasc. Res. 2010;88(2): 257-266. doi: https://doi.org/10.1093/cvr/cvq205
Timoshin AA, Vanin AF, Orlova TR, Sanina NA, Ruuge EK, Aldoshin SM, Chazov EI. Protein-bound Dinitrosyl-Iron Complexes Appearing in Blood of Rabbit Added With a Low-Molecular Dinitrosyl-Iron Complex: EPR Studies. Nitric Oxide. 2007;16(2):286-293. doi: https://doi.org/10.1016/j.niox.2006.09.005
Timoshin AА, Lakomkin VL, Аbramov AА, Ruuge EK, Kapel’ko VI, Chazov EI, Vanin AF. The Hypotensive Effect of the Nitric Monoxide Donor Oxacom at Different Routs of Its Administration to Experimental Animals. Eur. J. Pharmacol. 2015;765:525-532. doi: https://doi.org/10.1016/j.ejphar.2015.09.011
Vanin AF, Poltorakov AP, Mikoyan VD, Kubrina LN, Burbaev DS. Polynuclear Water-Soluble Dinitrosyl Iron Complexes With Cysteine or Glutathione Ligands: Electron Paramagnetic Resonance and Optical Studies. Nitric Oxide. 2010;23(2):136-149. doi: https://doi.org/10.1016/j.niox.2010.05.285
Vanin AF, Timoshin AA. Determination of in Vivo Nitric Oxide Levels in Animal Tissues Using a Novel Spin Trapping Technology. Methods Mol. Biol. 2011;704:135-149. doi: https://doi.org/10.1007/978-1-61737-964-2_11
Wu SC, Lu CY, Chen YL, Lo FC, Wang TY, Chen YJ, Yuan SS, Liaw WF, Wang YM. Water-Soluble Dinitrosyl Iron Complex (DNIC): A Nitric Oxide Vehicle Triggering Cancer Cell Death via Apoptosis. Inorg Chem. 2016;55(18):9383-9392. doi: https://doi.org/10.1021/acs.inorgchem.6b01562
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Byulleten’ Eksperimental’noi Biologii i Meditsiny, Vol. 170, No. 9, pp. 290-295, September, 2020
Rights and permissions
About this article
Cite this article
Timoshin, A.A., Shumaev, K.B., Lakomkin, V.L. et al. Study of Translocation of Stabilized NO Forms through Rat Skin using Electron Paramagnetic Resonance Method. Bull Exp Biol Med 170, 303–307 (2021). https://doi.org/10.1007/s10517-021-05056-z
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10517-021-05056-z