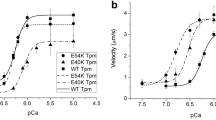
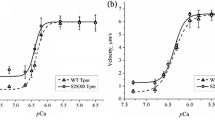
Tropomyosin (Tpm) is one of the main regulatory proteins in the myocardium. In some heart pathologies, interchain disulfide crosslinking in the Tpm molecule occurs. In the ventricle, this change in the structural properties of the Tpm molecule affects calcium regulation of the actin-myosin interaction. Using an in vitro motility assay, we found that Tpm crosslinking does not affect the actin-myosin interaction in the atria. We assume that the intramolecular crosslinking of Tpm in the atrium does not play such a crucial role in the pathogenesis of heart failure as it plays in the heart ventricles.
Similar content being viewed by others
References
Avner BS, Shioura KM, Scruggs SB, Grachoff M, Geenen DL, Helseth DL Jr, Farjah M, Goldspink PH, Solaro RJ. Myocardial infarction in mice alters sarcomeric function via post-translational protein modification. Mol. Cell. Biochem. 2012;363(1-3):203-215.
Canton M, Menazza S, Sheeran FL, Polverino de Laureto P, Di Lisa F, Pepe S. Oxidation of myofibrillar proteins in human heart failure. J. Am. Coll. Cardiol. 2011;57(3):300-309.
Canton M, Skyschally A, Menabò R, Boengler K, Gres P, Schulz R, Haude M, Erbel R, Di Lisa F, Heusch G. Oxidative modification of tropomyosin and myocardial dysfunction following coronary microembolization. Eur. Heart J. 2006;27(7):875-881.
Gordon AM, Homsher E, Regnier M. Regulation of contraction in striated muscle. Physiol. Rev. 2000;80(2):853-924.
Kremneva E, Boussouf S, Nikolaeva O, Maytum R, Geeves MA, Levitsky DI. Effects of two familial hypertrophic cardiomyopathy mutations in alpha-tropomyosin, Asp175Asn and Glu180Gly, on the thermal unfolding of actin-bound tropomyosin. Biophys. J. 2004;87(6):3922-3933.
Lehrer SS, Ly S, Fuchs F. Tropomyosin is in a reduced state in rat cardiac muscle. J. Muscle Res. Cell Motil. 2011;32(2):63-64.
Mashanov GI, Molloy JE. Automatic detection of single fluorophores in live cells. Biophys. J. 2007;92(6):2199-2211.
Matyushenko AM, Artemova NV, Shchepkin DV, Kopylova GV, Nabiev SR, Nikitina LV, Levitsky DI, Bershitsky SY. The interchain disulfide cross-linking of tropomyosin alters its regulatory properties and interaction with actin filament. Biochem. Biophys. Res. Commun. 2017;482(2):305-309.
Morano I. Tuning the human heart molecular motors by myosin light chains. J. Mol. Med. 1999;77(7):544-555.
Shchepkin DV, Kopylova GV, Nikitina LV. Study of reciprocal effects of cardiac myosin and tropomyosin isoforms on actin-myosin interaction with in vitro motility assay. Biochem. Biophys. Res. Commun. 2011;415(1):104-108
Shchepkin DV, Nikitina LV, Bershitsky SY, Kopylova GV. The isoforms of α-actin and myosin affect the Ca2+ regulation of the actin-myosin interaction in the heart. Biochem. Biophys. Res. Commun. 2017;490(2):324-329.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Byulleten’ Eksperimental’noi Biologii i Meditsiny, Vol. 167, No. 1, pp. 72-75, January, 2019
Rights and permissions
About this article
Cite this article
Shchepkin, D.V., Matyushenko, A.M., Bershitsky, S.Y. et al. Effect of Interchain Disulfide Crosslinking in the Tropomyosin Molecule on Actin-Myosin Interaction in the Atrial Myocardium. Bull Exp Biol Med 167, 65–68 (2019). https://doi.org/10.1007/s10517-019-04462-8
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10517-019-04462-8