Abstract
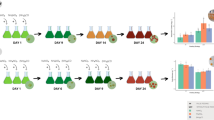
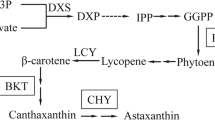
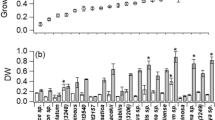
Haematococcus pluvialis is known as the richest natural source of carotenoid astaxanthin, whose synthesis is related to changes in cells according its complex life cycle and the availability of nutrients present in the culture media. This study’s objective was to evaluate the influence of different culture media under photoautotrophic and mixotrophic conditions on the growth, life cycle, biochemical composition, and astaxanthin production in H. pluvialis. Cultures were performed using the following media: KM1, MM2, Provasoli, modified Provasoli, and how to control the BBM, all kept at 24 ± 1 °C, with constant aeration, light/dark photoperiod 24:0 and irradiance of 60 μmol photons/m2/s. Growth and life cycle were evaluated by daily cell counts under an optical microscope; values of maximum cell density (MCD) and dry biomass were also assessed. Biochemical analyses were chlorophyll (a and b), astaxanthin, total proteins, and fatty acids. Cultivation under photoautotrophic conditions in BBM medium showed higher concentration of chlorophylls a (33 ± 4.0 μg/mL) and b (18 ± 1.6 μg/mL) and protein (62.7 ± 3.38%). Cultivation in modified Provasoli medium, under mixotrophic conditions, showed higher MCD (84.5 ± 11.7), biomass production (4.9 ± 0.0004 mg/mL), fatty acid methyl esters, and astaxanthin productivity (9.28 ± 0.4 mg/L/day). These results demonstrate that the culture media and the culture conditions to which H. pluvialis is submitted stimulate specific metabolic and biosynthetic pathways of the cells, which directly affect the biochemical composition of the microalgae. Modified Provasoli medium proved to be the most efficient in increasing astaxanthin productivity for this species, as it has shown better growth parameters and biomass production.





Similar content being viewed by others
References
Aflalo C, Meshulam Y, Zarka A, Boussiba S (2007) On the relative efficiency of two- vs. one-stage production of astaxanthin by the green alga Haematococcus pluvialis. Biotechnol Bioeng 98:300–305. https://doi.org/10.1002/bit.21391
An Z, Yang H, Liu X, Zhangv Y (2020) Effects of astaxanthin on the immune response and reproduction of Procambarus clarkii stressed with microcystin-leucine-arginine. Fish Sci 1:1–8. https://doi.org/10.1007/s12562-020-01434-0
Angell A, de Nys R, Mangott A, Vucko MJ (2018) The effects of concentration and supplementation time of natural and synthetic sources of astaxanthin on the colouration of the prawn Penaeus monodon. Algal Res 35:577–585. https://doi.org/10.1016/j.algal.2018.09.031
APHA/AWWA/WEF (2012) Standard Methods for the Examination of Water and Wastewater. Stand Methods 541. doi: ISBN 9780875532356
Barbarino E, Lourenço SO (2005) An evaluation of methods for extraction and quantification of protein from marine macro- and microalgae. J Appl Phycol 17:447–460. https://doi.org/10.1007/s10811-005-1641-4
Barbosa MJ, Morais R, Choubert G (1999) Effect of carotenoid source and dietary lipid content on blood astaxanthin concentration in rainbow trout (Oncorhynchus mykiss). Aquaculture 176:331–341. https://doi.org/10.1016/S0044-8486(99)00115-5
Berman-Frank I, Lundgren P, Chen YB et al (2001) Segregation of nitrogen fixation and oxygenic photosynthesis in the marine cyanobacterium Trichodesmium. Science (80- ) 294:1534–1537. https://doi.org/10.1126/science.1064082
Bligh EG, Dyer WJ (1959) A rapid method of total lipid extraction and purification. Can J Biochem Physiol 37:911–917. https://doi.org/10.1139/o59-099
Bold HC (1949) The morphology of Chlamydomonas chlamydogama sp. nov. Bull Torrey Bot Club 76:101–108
Borowitzka MA, Huisman JM, Osborn A (1991) Culture of the astaxanthin-producing green alga Haematococcus pluvialis 1. Effects of nutrients on growth and cell type. J Appl Phycol 3:295–304. https://doi.org/10.1007/BF02392882
Boyle NR, Morgan JA (2009) Flux balance analysis of primary metabolism in Chlamydomonas reinhardtii. BMC Syst Biol 3:1–14. https://doi.org/10.1186/1752-0509-3-4
Butler T, McDougall G, Campbell R, Stanley M, Day J (2017) Media screening for obtaining Haematococcus pluvialis red motile macrozooids rich in astaxanthin and fatty acids. Biology (Basel) 7:2. https://doi.org/10.3390/biology7010002
Cheng J, Li K, Yang Z, Zhou J, Cen K (2016) Enhancing the growth rate and astaxanthin yield of Haematococcus pluvialis by nuclear irradiation and high concentration of carbon dioxide stress. Bioresour Technol 204:49–54. https://doi.org/10.1016/j.biortech.2015.12.076
Cheng T, Xu X, Zhang W, Chen L, Liu T (2018) Protoplast preparation from enriched flagellates and resting cells of Haematococcus pluvialis. J Appl Microbiol 124:469–479. https://doi.org/10.1111/jam.13643
Chioccioli M, Hankamer B, Ross IL (2014) Flow cytometry pulse width data enables rapid and sensitive estimation of biomass dry weight in the microalgae Chlamydomonas reinhardtii and Chlorella vulgaris. PLoS One 9:1–12. https://doi.org/10.1371/journal.pone.0097269
Chiranjeevi P, Mohan SV (2016) Critical parametric influence on microalgae cultivation towards maximizing biomass growth with simultaneous lipid productivity. Renew Energy 98:64–71. https://doi.org/10.1016/j.renene.2016.03.063
Cui D, Hu C, Zou Z, Sun X, Shi J, Xu N (2020) Comparative transcriptome analysis unveils mechanisms underlying the promoting effect of potassium iodide on astaxanthin accumulation in Haematococcus pluvialis under high light stress. Aquaculture 525:735279. https://doi.org/10.1016/j.aquaculture.2020.735279
Cunha L, Besen K, Ha N et al (2020) Biofloc technology (BFT) improves skin pigmentation of goldfish (Carassius auratus). Aquaculture 522:735132. https://doi.org/10.1016/j.aquaculture.2020.735132
Dong S, Huang Y, Zhang R, Wang S, Liu Y (2014) Four different methods comparison for extraction of astaxanthin from green alga Haematococcus pluvialis. ScientificWorldJournal 2014:694305–694307. https://doi.org/10.1155/2014/694305
Doria E, Temporiti MEE, Damiani MC et al (2018) Influence of light stress on the accumulation of xanthophylls and lipids in Haematococcus pluvialis CCALA 1081 grown under autotrophic or mixotrophic conditions. J Mar Biol Aquac 4:36–40. https://doi.org/10.15436/2381-0750.18.1799
Faraone I, Sinisgalli C, Ostuni A, Armentano MF, Carmosino M, Milella L, Russo D, Labanca F, Khan H (2020) Astaxanthin anticancer effects are mediated through multiple molecular mechanisms: a systematic review. Pharmacol Res 155:104689. https://doi.org/10.1016/j.phrs.2020.104689
Giannelli L, Yamada H, Katsuda T, Yamaji H (2015) Effects of temperature on the astaxanthin productivity and light harvesting characteristics of the green alga Haematococcus pluvialis. J Biosci Bioeng 119:345–350. https://doi.org/10.1016/j.jbiosc.2014.09.002
Göksan T, Ak I, Kılıç C (2011) Growth characteristics of the alga Haematococcus pluvialis flotow as affected by nitrogen source, vitamin, light and aeration. Turkish J Fish Aquat Sci 11:377–383. https://doi.org/10.4194/1303-2712-v11
Gouveia L, Rema P, Pereira O, Empis J (2003) Colouring ornamental fish (Cyprinus carpio and Carassius auratus) with microalgal biomass. Aquac Nutr 9:123–129. https://doi.org/10.1046/j.1365-2095.2003.00233.x
Humphrey GF (1979) Photosynthetic characteristics of algae grown under constant illumination and light-dark regimes. J Exp Mar Bio Ecol 40:63–70. https://doi.org/10.1016/0022-0981(79)90034-0
Jeffrey SW, Humphrey GF (1975) New spectrophotometric equations for determining chlorophylls a, b, c1 and c2 in higher plants, algae and natural phytoplankton. Biochem und Physiol der Pflanz 167:191–194. https://doi.org/10.1016/S0015-3796(17)30778-3
Jiang X, Yan Q, Liu F, Jing C, Ding L, Zhang L, Pang C (2018) Chronic trans-astaxanthin treatment exerts antihyperalgesic effect and corrects co-morbid depressive like behaviors in mice with chronic pain. Neurosci Lett 662:36–43. https://doi.org/10.1016/j.neulet.2017.09.064
Jiang J, Nuez-Ortin W, Angell A, Zeng C, de Nys R, Vucko MJ (2019) Enhancing the colouration of the marine ornamental fish Pseudochromis fridmani using natural and synthetic sources of astaxanthin. Algal Res 42:101596. https://doi.org/10.1016/j.algal.2019.101596
Katiyar R, Gurjar BR, Biswas S, Pruthi V, Kumar N, Kumar P (2017) Microalgae: an emerging source of energy based bio-products and a solution for environmental issues. Renew Sust Energ Rev 72:1083–1093. https://doi.org/10.1016/j.rser.2016.10.028
Kim S, Park JE, Cho YB, Hwang SJ (2013) Growth rate, organic carbon and nutrient removal rates of Chlorella sorokiniana in autotrophic, heterotrophic and mixotrophic conditions. Bioresour Technol 144:8–13. https://doi.org/10.1016/j.biortech.2013.06.068
Kobayashi M, Kakizono T, Nagai S (1991) Astaxanthin production by a green alga, Haematococcus pluvialis accompanied with morphological changes in acetate media. J Ferment Bioeng 71:335–339. https://doi.org/10.1016/0922-338X(91)90346-I
Li Y, Sommerfeld M, Chen F, Hu Q (2010) Effect of photon flux densities on regulation of carotenogenesis and cell viability of Haematococcus pluvialis (Chlorophyceae). J Appl Phycol 22(3):253–263
Lourenço SO, Barbarino E, Lavín PL, Lanfer Marquez UM, Aidar E (2004) Distribution of intracellular nitrogen in marine microalgae: calculation of new nitrogen-to-protein conversion factors. Eur J Phycol 39:17–32. https://doi.org/10.1080/0967026032000157156
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275. https://doi.org/10.1016/0304-3894(92)87011-4
Mandotra SK, Kumar P, Suseela MR, Nayaka S, Ramteke PW (2016) Evaluation of fatty acid profile and biodiesel properties of microalga Scenedesmus abundans under the influence of phosphorus, pH and light intensities. Bioresour Technol 201:222–229. https://doi.org/10.1016/j.biortech.2015.11.042
Mazumdar N, Novis PM, Visnovsky G, Gostomski PA (2019) Effect of nutrients on the growth of a new alpine strain of Haematococcus (Chlorophyceae) from New Zealand. Phycol Res 67:21–27. https://doi.org/10.1111/pre.12344
Mustafa Y, Fagiri A, Salleh A, El-nagerabi SAF (2013) Influence of chemical and environmental factors on the growth performance of Spirulina platensis strain SZ100. J Algal Biomass Util 4:7–15
Nahidian B, Ghanati F, Shahbazi M, Soltani N (2018) Effect of nutrients on the growth and physiological features of newly isolated Haematococcus pluvialis TMU. Bioresour Technol 255:229–237. https://doi.org/10.1016/j.biortech.2018.01.130
Nguyen BT, Rittmann BE (2015) Predicting dissolved inorganic carbon in photoautotrophic microalgae culture via the nitrogen source. Environ Sci Technol 49:9826–9831. https://doi.org/10.1021/acs.est.5b01727
Nigam S, Rai MP, Sharma R (2011) Effect of nitrogen on growth and lipid content of Chlorella pyrenoidosa. Am J Biochem Biotechnol 7:126–131. https://doi.org/10.3844/ajbbsp.2011.126.131
Orosa M, Franqueira D, Cid A, Abalde J (2005) Analysis and enhancement of astaxanthin accumulation in Haematococcus pluvialis. Bioresour Technol 96:373–378. https://doi.org/10.1016/j.biortech.2004.04.006
Pancha I, Chokshi K, George B, Ghosh T, Paliwal C, Maurya R, Mishra S (2014) Nitrogen stress triggered biochemical and morphological changes in the microalgae Scenedesmus sp. CCNM 1077. Bioresour Technol 156:146–154. https://doi.org/10.1016/j.biortech.2014.01.025
Pang N, Chen S (2017a) Effects of C5 organic carbon and light on growth and cell activity of Haematococcus pluvialis under mixotrophic conditions. Algal Res 21:227–235. https://doi.org/10.1016/j.algal.2016.12.003
Pang N, Chen S (2017b) Effects of C5 organic carbon and light on growth and cell activity of Haematococcus pluvialis under mixotrophic conditions. Algal Res 21:227–235. https://doi.org/10.1016/j.algal.2016.12.003
Pang N, Gu X, Chen S, Kirchhoff H, Lei H, Roje S (2019a) Exploiting mixotrophy for improving productivities of biomass and co-products of microalgae. Renew Sust Energ Rev 112:450–460. https://doi.org/10.1016/j.rser.2019.06.001
Pang N, Xie Y, Oung HMO, Sonawane BV, Fu X, Kirchhoff H, Cousins AB, Chen S (2019b) Regulation and stimulation of photosynthesis of mixotrophically cultured Haematococcus pluvialis by ribose. Algal Res 39:101443. https://doi.org/10.1016/j.algal.2019.101443
Pan-utai W, Parakulsuksatid P, Phomkaivon N (2017) Effect of inducing agents on growth and astaxanthin production in Haematococcus pluvialis: organic and inorganic. Biocatal Agric Biotechnol 12:152–158. https://doi.org/10.1016/j.bcab.2017.10.004
Perez-Garcia O, Escalante FME, de Bashan LE, Bashan Y (2011) Heterotrophic cultures of microalgae: metabolism and potential products. Water Res 45:11–36
Pindyck RS, Rubinfeld DL (1990) Econometric models and economic forecasts, 3rd edn. Mcgraw-Hill College, New York
Provasoli L (1968) Media and prospects for the cultivation of marine algae. In: Watanabe H, Hattori A (eds) Cultures and collections of algae, proceeding. Hakone, Japanese Society of Plant Physiology, pp 63–75
RCoreTeam (2017) R: a language and environment for statistical computing. R Foundation for Statistical Computing
Roopnarain A, Gray VM, Sym SD (2014) Phosphorus limitation and starvation effects on cell growth and lipid accumulation in Isochrysis galbana U4 for biodiesel production. Bioresour Technol 156:408–411. https://doi.org/10.1016/j.biortech.2014.01.092
Sarada R, Bhattacharya S, Ravishankar GA (2002) Optimization of culture conditions for growth of the green alga Haematococcus pluvialis. World J Microbiol Biotechnol 18:517–521. https://doi.org/10.1023/A:1016349828310
Shah MMR, Liang Y, Cheng JJ, Daroch M (2016) Astaxanthin-producing green microalga Haematococcus pluvialis: from single cell to high value commercial products. Front Plant Sci 7:1–28. https://doi.org/10.3389/fpls.2016.00531
Shen XF, Liu JJ, Chauhan AS, Hu H, Ma LL, Lam PKS, Zeng RJ (2016) Combining nitrogen starvation with sufficient phosphorus supply for enhanced biodiesel productivity of Chlorella vulgaris fed on acetate. Algal Res 17:261–267. https://doi.org/10.1016/j.algal.2016.05.018
Stein JR (1973) Handbook of phycological methods: culture methods and growth measurements, 1rd edn. Cambridge University, Cambridge
Su F, Yu W, Liu J (2020) Comparison of effect of dietary supplementation with Haematococcus pluvialis powder and synthetic astaxanthin on carotenoid composition, concentration, esterification degree and astaxanthin isomers in ovaries, hepatopancreas, carapace, epithelium of adult. Aquaculture 523:735146. https://doi.org/10.1016/j.aquaculture.2020.735146
Tocquin P, Fratamico A, Franck F (2012) Screening for a low-cost Haematococcus pluvialis medium reveals an unexpected impact of a low N/P ratio on vegetative growth. J Appl Phycol 24:365–373. https://doi.org/10.1007/s10811-011-9771-3
Tripathi U, Sarada R, Ramachandra Rao S, Ravishankar GA (1999) Production of astaxanthin in Haematococcus pluvialis cultured in various media. Bioresour Technol 68:197–199. https://doi.org/10.1016/S0960-8524(98)00143-6
Wahidin S, Idris A, Shaleh SRM (2013) The influence of light intensity and photoperiod on the growth and lipid content of microalgae Nannochloropsis sp. Bioresour Technol 129:7–11. https://doi.org/10.1016/j.biortech.2012.11.032
Wan M, Zhang Z, Wang J, Huang J, Fan J, Yu A, Wang W, Li Y (2015) Sequential heterotrophy-dilution-photoinduction cultivation of Haematococcus pluvialis for efficient production of astaxanthin. Bioresour Technol 198:557–563. https://doi.org/10.1016/j.biortech.2015.09.031
Wang J, Curtis WR (2016) Proton stoichiometric imbalance during algae photosynthetic growth on various nitrogen sources: toward metabolic pH control. J Appl Phycol 28:43–52. https://doi.org/10.1007/s10811-015-0551-3
Wang N, Guan B, Kong Q, Duan L (2017) A semi-continuous cultivation method for Haematococcus pluvialis from non-motile cells to motile cells. J Appl Phycol 30:773–781. https://doi.org/10.1007/s10811-017-1337-6
Wang Y, Wang B, Liu M, Jiang K, Wang M, Wang L (2020) Comparative transcriptome analysis reveals the potential influencing mechanism of dietary astaxanthin on growth and metabolism in Litopenaeus vannamei. Aquac Reports 16:100259. https://doi.org/10.1016/j.aqrep.2019.100259
Zhang C, Liu J, Zhang L (2017) Cell cycles and proliferation patterns in Haematococcus pluvialis. Chin J Oceanol Limnol 35:1205–1211. https://doi.org/10.1007/s00343-017-6103-8
Zhang C, Zhang L, Liu J (2019) Exogenous sodium acetate enhances astaxanthin accumulation and photoprotection in Haematococcus pluvialis at the non-motile stage. J Appl Phycol 31:1001–1008. https://doi.org/10.1007/s10811-018-1622-z
Zhekisheva M, Boussiba S, Khozin-Goldberg I, Zarka A, Cohen Z (2002) Accumulation of oleic acid in Haematococcus pluvialis (Chlorophyceae) under nitrogen starvation or high light is correlated with that of astaxanthin esters. J Phycol 38:325–331. https://doi.org/10.1046/j.1529-8817.2002.01107.x
Zhu CJ, Lee YK (1997) Determination of biomass dry weight of marine microalgae. J Appl Phycol 9:189–194. https://doi.org/10.1023/A:1007914806640
Acknowledgements
The authors would like to thank Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) for the financial support provided to this the research and partners involved in its execution and to the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) for the scholarship granted to the first author. We would also like to thank the Labpratório de Bioprocessos and Central Análitica do Centro de Tecnologias Estratégicas do Nordeste (CETENE).
Code availability
Not applicable.
Funding
The financial support granted to this research and the scholarships for the researchers involved were made by the following institutions: Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by Yllana Ferreira Marinho, Carolina Barbosa Malafaia, Katarynna Santos de Araújo, Túlio Diego da Silva, Ana Paula Felipe dos Santos, Laenne Barbara de Moraes, and Alfredo Oliveira Gálvez. The first draft of the manuscript was written by Yllana Ferreira Marinho, and all authors commented on the previous versions of the manuscript.
Corresponding author
Ethics declarations
Ethics approval
This article does not contain any studies with animals performed by any of the authors.
Consent to participate
Not applicable.
Consent for publication
All authors read and approved the final manuscript and are in agreement with the submission and publication of this research.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Marinho, Y.F., Malafaia, C.B., de Araújo, K.S. et al. Evaluation of the influence of different culture media on growth, life cycle, biochemical composition, and astaxanthin production in Haematococcus pluvialis. Aquacult Int 29, 757–778 (2021). https://doi.org/10.1007/s10499-021-00655-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10499-021-00655-z