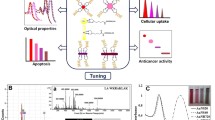
Abstract
Although new cancer therapeutics are discovered at a rapid pace, lack of effective means of delivery and cancer chemoresistance thwart many of the promising therapeutics. We demonstrate a method that confronts both of these issues with the light-activated delivery of a Bcl-2 functional converting peptide, NuBCP-9, using hollow gold nanoshells. This approach has shown not only to increase the efficacy of the peptide 30-fold in vitro but also has shown to reduce paclitaxel resistant H460 lung xenograft tumor growth by 56.4%.




Similar content being viewed by others
References
Allen TM (2002) Ligand-targeted therapeutics in anticancer therapy. Nat Rev Cancer 2(10):750–763
Holohan C, Van Schaeybroeck S, Longley DB, Johnston PG (2013) Cancer drug resistance: an evolving paradigm. Nat Rev Cancer 13(10):714–726
Gupta D, Kumar M, Tyagi P, Kapoor S, Tyagi A, Barman TK et al (2018) Corrigendum to ‘Concomitant delivery of paclitaxel and NuBCP-9 peptide for synergistic enhancement of cancer therapy’ NANO 14 (2018) 1301–1313. Nanomed Nanotechnol Biol Med 14(7):2129
Pearce MC, Gamble JT, Kopparapu PR, O’Donnell EF, Mueller MJ, Jang HS et al (2018) Induction of apoptosis and suppression of tumor growth by Nur77-derived Bcl-2 converting peptide in chemoresistant lung cancer cells. Oncotarget. https://doi.org/10.18632/oncotarget.25437
Li H, Nelson CE, Evans BC, Duvall CL (2011) Delivery of intracellular-acting biologics in pro-apoptotic therapies. Curr Pharm Des 17(3):293–319
Hughes J, Rees S, Kalindjian S, Philpott K (2011) Principles of early drug discovery: principles of early drug discovery. Br J Pharmacol 162(6):1239–1249
Czabotar PE, Lessene G, Strasser A, Adams JM (2014) Control of apoptosis by the BCL-2 protein family: implications for physiology and therapy. Nat Rev Mol Cell Biol 15(1):49–63
Yip KW, Reed JC (2008) Bcl-2 family proteins and cancer. Oncogene 27(50):6398–6406
Bouchalova K, Svoboda M, Kharaishvili G, Vrbkova J, Bouchal J, Trojanec R et al (2015) BCL2 is an independent predictor of outcome in basal-like triple-negative breast cancers treated with adjuvant anthracycline-based chemotherapy. Tumour Biol J Int Soc Oncodev Biol Med 36(6):4243–4252
Honma N, Horii R, Ito Y, Saji S, Younes M, Iwase T et al (2015) Differences in clinical importance of Bcl-2 in breast cancer according to hormone receptors status or adjuvant endocrine therapy. BMC Cancer 15(1):698
Giuliano M, Hu H, Wang Y-C, Fu X, Nardone A, Herrera S et al (2015) Upregulation of ER signaling as an adaptive mechanism of cell survival in HER2-positive breast tumors treated with anti-HER2 therapy. Clin Cancer Res Off J Am Assoc Cancer Res 21(17):3995–4003
Real PJ, Sierra A, De Juan A, Segovia JC, Lopez-Vega JM, Fernandez-Luna JL (2002) Resistance to chemotherapy via Stat3-dependent overexpression of Bcl-2 in metastatic breast cancer cells. Oncogene 21(50):7611–7618
Choudhary GS, Al-harbi S, Mazumder S, Hill BT, Smith MR, Bodo J et al (2015) MCL-1 and BCL-xL-dependent resistance to the BCL-2 inhibitor ABT-199 can be overcome by preventing PI3K/AKT/mTOR activation in lymphoid malignancies. Cell Death Dis 6(1):e1593
Yecies D, Carlson NE, Deng J, Letai A (2010) Acquired resistance to ABT-737 in lymphoma cells that up-regulate MCL-1 and BFL-1. Blood 115(16):3304–3313
Lin B, Kolluri SK, Lin F, Liu W, Han Y-H, Cao X et al (2004) Conversion of Bcl-2 from protector to killer by interaction with nuclear orphan receptor Nur77/TR3. Cell 116(4):527–540
Kolluri SK, Zhu X, Zhou X, Lin B, Chen Y, Sun K et al (2008) A short Nur77-derived peptide converts Bcl-2 from a protector to a killer. Cancer Cell 14(4):285–298
Bodo J, Zhao X, Durkin L, Souers AJ, Phillips DC, Smith MR et al (2016) Acquired resistance to venetoclax (ABT-199) in positive lymphoma cells. Oncotarget 7(43):70000–70010
Huang S, Jiang C, Guo H, Wang J, Liu Y, Li C, Lopez E, Zhang H, Lorence EA, Merolle M et al (2017) Resistance mechanisms underlying venetoclax resistance in mantle cell lymphoma. Blood 130(Suppl 1):2749
Kapoor S, Gupta D, Kumar M, Sharma S, Gupta AK, Misro MM et al (2016) Intracellular delivery of peptide cargos using polyhydroxybutyrate based biodegradable nanoparticles: studies on antitumor efficacy of BCL-2 converting peptide, NuBCP-9. Int J Pharm 511(2):876–889
Kumar M, Gupta D, Singh G, Sharma S, Bhat M, Prashant CK et al (2014) Novel polymeric nanoparticles for intracellular delivery of peptide cargos: antitumor efficacy of the BCL-2 conversion peptide NuBCP-9. Cancer Res 74(12):3271–3281
Kumar M, Singh G, Sharma S, Gupta D, Bansal V, Arora V et al (2014) Intracellular delivery of peptide cargos using iron oxide based nanoparticles: studies on antitumor efficacy of a BCL-2 converting peptide, NuBCP-9. Nanoscale 6(23):14473–14483
Prevo B, Esakoff S, Mikhailovsky A, Zasadzinski J (2008) Scalable routes to gold nanoshells with tunable sizes and response to near-infrared pulsed laser irradiation. Small 4:1183–1195
Kimmel CB, Ballard WW, Kimmel SR, Ullmann B, Schilling TF (1995) Stages of embryonic development of the zebrafish. Dev Dyn 203(3):253–310
Schindelin J, Arganda-Carreras I, Frise E, Kaynig V, Longair M, Pietzsch T et al (2012) Fiji: an open-source platform for biological-image analysis. Nat Methods 9(7):676–682
Wehmas LC, Tanguay RL, Punnoose A, Greenwood JA (2016) Developing a novel embryo–larval zebrafish xenograft assay to prioritize human glioblastoma therapeutics. Zebrafish 13:317–329
Chakraborty C, Sharma AR, Sharma G, Lee S-S (2016) Zebrafish: a complete animal model to enumerate the nanoparticle toxicity. J Nanobiotechnol. https://doi.org/10.1186/s12951-016-0217-6
Liu J, Liu Y, Bu W et al (2014) Ultrasensitive nanosensors based on upconversion nanoparticles for selective hypoxia imaging in vivo upon near-infrared excitation. J Am Chem Soc 136:9701–9709
Liu H-Y, Wu P-J, Kuo S-Y et al (2015) Quinoxaline-based polymer dots with ultrabright red to near-infrared fluorescence for in vivo biological imaging. J Am Chem Soc 137:10420–10429
Acknowledgements
This work was supported by the National Institutes of Health (NIH) Grant R01 EB012637 and in part by Grants from the US Army Medical Research and Material Command (W81XWH-08-1-0600 and W81XWH-12-1-0069), American Cancer Society (RSG-13-132-01-CDD), National Institutes of Health (5RO1ES016651) and Oregon State University Venture Development Fund (http://advantage.oregonstate.edu/funding-opportunities). The authors thank support of the NRI Microscopy Center, the Olympus confocal microscope was funded by the NIH Grant 1S10RR022585-01A1. The authors thank A. Mikhailovsky for helpful conversations and aid of the UCSB Optical Characterization Facility. The ultrafast laser system was funded by DURIP ARO Grant 66886LSRIP. The authors would also like to thank former graduate student Demosthenes Morales for helpful discussions.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing financial interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Morgan, E., Gamble, J.T., Pearce, M.C. et al. Improved in vivo targeting of BCL-2 phenotypic conversion through hollow gold nanoshell delivery. Apoptosis 24, 529–537 (2019). https://doi.org/10.1007/s10495-019-01531-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-019-01531-1