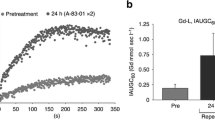
Abstract
In the past two decades advances in the development of targeted nanoparticles have facilitated their application as molecular imaging agents and targeted drug delivery vehicles. Nanoparticle-enhanced molecular imaging of the angiogenic tumor vasculature has been of particular interest. Not only because angiogenesis plays an important role in various pathologies, but also since endothelial cell surface receptors are directly accessible for relatively large circulating nanoparticles. Typically, nanoparticle targeting towards these receptors is studied by analyzing the contrast distribution on tumor images acquired before and at set time points after administration. Although several exciting proof-of-concept studies demonstrated qualitative assessment of relative target concentration and distribution, these studies did not provide quantitative information on the nanoparticle targeting kinetics. These kinetics will not only depend on nanoparticle characteristics, but also on receptor binding and recycling. In this study, we monitored the in vivo targeting kinetics of αvβ3-integrin specific nanoparticles with intravital microscopy and dynamic contrast enhanced magnetic resonance imaging, and using compartment modeling we were able to quantify nanoparticle targeting rates. As such, this approach can facilitate optimization of targeted nanoparticle design and it holds promise for providing more quantitative information on in vivo receptor levels. Interestingly, we also observed a periodicity in the accumulation kinetics of αvβ3-integrin targeted nanoparticles and hypothesize that this periodicity is caused by receptor binding, internalization and recycling dynamics. Taken together, this demonstrates that our experimental approach provides new insights in in vivo nanoparticle targeting, which may proof useful for vascular targeting in general.









Similar content being viewed by others
References
Lobatto ME, Fuster V, Fayad ZA, Mulder WJ (2011) Perspectives and opportunities for nanomedicine in the management of atherosclerosis. Nat Rev Drug Discov 10(11):835–852. doi:10.1038/nrd3578
Choi KY, Liu G, Lee S, Chen X (2012) Theranostic nanoplatforms for simultaneous cancer imaging and therapy: current approaches and future perspectives. Nanoscale 4(2):330–342. doi:10.1039/c1nr11277e
Lanza GM, Wallace KD, Scott MJ, Cacheris WP, Abendschein DR, Christy DH, Sharkey AM, Miller JG, Gaffney PJ, Wickline SA (1996) A novel site-targeted ultrasonic contrast agent with broad biomedical application. Circulation 94(12):3334–3340
Mulder WJ, van der Schaft DW, Hautvast PA, Strijkers GJ, Koning GA, Storm G, Mayo KH, Griffioen AW, Nicolay K (2007) Early in vivo assessment of angiostatic therapy efficacy by molecular MRI. FASEB J 21(2):378–383. doi:10.1096/fj.06-6791com
Medarova Z, Pham W, Farrar C, Petkova V, Moore A (2007) In vivo imaging of siRNA delivery and silencing in tumors. Nat Med 13(3):372–377. doi:10.1038/nm1486
Winter PM, Caruthers SD, Kassner A, Harris TD, Chinen LK, Allen JS, Lacy EK, Zhang H, Robertson JD, Wickline SA, Lanza GM (2003) Molecular imaging of angiogenesis in nascent Vx-2 rabbit tumors using a novel alpha(nu)beta3-targeted nanoparticle and 1.5 tesla magnetic resonance imaging. Cancer Res 63(18):5838–5843
Nel AE, Madler L, Velegol D, Xia T, Hoek EM, Somasundaran P, Klaessig F, Castranova V, Thompson M (2009) Understanding biophysicochemical interactions at the nano-bio interface. Nat Mater 8(7):543–557. doi:10.1038/nmat2442
Choi HS, Frangioni JV (2010) Nanoparticles for biomedical imaging: fundamentals of clinical translation. Mol imaging 9(6):291–310
Lammers T, Kiessling F, Hennink WE, Storm G (2012) Drug targeting to tumors: principles, pitfalls and (pre-) clinical progress. J Control Release 161(2):175–187. doi:10.1016/j.jconrel.2011.09.063
Alberts B, Johnson A, Lewis J, Raff M, Roberts K, Walter P (2007) Molecular biology of the cell, 5th edn. Garland Science, New York
Ocak I, Baluk P, Barrett T, McDonald DM, Choyke P (2007) The biologic basis of in vivo angiogenesis imaging. Front Biosci 12:3601–3616
O’Connor JP, Jackson A, Parker GJ, Roberts C, Jayson GC (2012) Dynamic contrast-enhanced MRI in clinical trials of antivascular therapies. Nat Rev Clin oncol 9(3):167–177. doi:10.1038/nrclinonc.2012.2
Larsson HB, Stubgaard M, Frederiksen JL, Jensen M, Henriksen O, Paulson OB (1990) Quantitation of blood-brain barrier defect by magnetic resonance imaging and gadolinium-DTPA in patients with multiple sclerosis and brain tumors. Magn Reson Med 16(1):117–131
Patlak CS, Blasberg RG (1985) Graphical evaluation of blood-to-brain transfer constants from multiple-time uptake data. Generalizations. J Cereb Blood Flow Metab 5(4):584–590. doi:10.1038/jcbfm.1985.87
Tofts PS, Kermode AG (1991) Measurement of the blood-brain barrier permeability and leakage space using dynamic MR imaging. 1. Fundamental concepts. Magn Reson Med 17(2):357–367
Backer MV, Backer JM (2012) Imaging key biomarkers of tumor angiogenesis. Theranostics 2(5):502–515. doi:10.7150/thno.3623
Folkman J (2002) Role of angiogenesis in tumor growth and metastasis. Semin Oncol 29(6, Supplement 16):15–18. doi:10.1016/S0093-7754(02)70065-1
McKeage MJ, Baguley BC (2010) Disrupting established tumor blood vessels: an emerging therapeutic strategy for cancer. Cancer 116(8):1859–1871. doi:10.1002/cncr.24975
Liu Z, Wang F, Chen X (2008) Integrin alpha(v)beta(3)-targeted cancer therapy. Drug Dev Res 69(6):329–339. doi:10.1002/ddr.20265
Sipkins DA, Cheresh DA, Kazemi MR, Nevin LM, Bednarski MD, Li KC (1998) Detection of tumor angiogenesis in vivo by alphaVbeta3-targeted magnetic resonance imaging. Nat Med 4(5):623–626
Tateishi U, Oka T, Inoue T (2012) Radiolabeled RGD peptides as integrin alpha(v)beta3-targeted PET tracers. Curr Med Chem 19(20):3301–3309
Caswell PT, Norman JC (2006) Integrin trafficking and the control of cell migration. Traffic 7(1):14–21. doi:10.1111/j.1600-0854.2005.00362.x
Kok MB, Hak S, Mulder WJ, van der Schaft DW, Strijkers GJ, Nicolay K (2009) Cellular compartmentalization of internalized paramagnetic liposomes strongly influences both T1 and T2 relaxivity. Magn Reson Med 61(5):1022–1032. doi:10.1002/mrm.21910
Roberts M, Barry S, Woods A, van der Sluijs P, Norman J (2001) PDGF-regulated rab4-dependent recycling of alphavbeta3 integrin from early endosomes is necessary for cell adhesion and spreading. Curr Biol 11(18):1392–1402
Janssen APCA, Schiffelers RM, ten Hagen TLM, Koning GA, Schraa AJ, Kok RJ, Storm G, Molema G (2003) Peptide-targeted PEG-liposomes in anti-angiogenic therapy. Int J Pharm 254(1):55–58. doi:10.1016/S0378-5173(02)00682-8
Hak S, Helgesen E, Hektoen HH, Huuse EM, Jarzyna PA, Mulder WJ, Haraldseth O, Davies C de L (2012) The effect of nanoparticle polyethylene glycol surface density on ligand-directed tumor targeting studied in vivo by dual modality imaging. ACS Nano 6(6):5648–5658. doi:10.1021/nn301630n
Seymour LW (1992) Passive tumor targeting of soluble macromolecules and drug conjugates. Crit Rev Ther Drug Carr Syst 9(2):135–187
Jarzyna PA, Skajaa T, Gianella A, Cormode DP, Samber DD, Dickson SD, Chen W, Griffioen AW, Fayad ZA, Mulder WJ (2009) Iron oxide core oil-in-water emulsions as a multifunctional nanoparticle platform for tumor targeting and imaging. Biomaterials 30(36):6947–6954. doi:10.1016/j.biomaterials.2009.09.004
Erikson A, Tufto I, Bjonnum AB, Bruland OS, Davies C de L (2008) The impact of enzymatic degradation on the uptake of differently sized therapeutic molecules. Anticancer Res 28(6A):3557–3566
Tofts PS (1997) Modeling tracer kinetics in dynamic Gd-DTPA MR imaging. J Magn Reson Imaging 7(1):91–101
Larsson HB, Fritz-Hansen T, Rostrup E, Sondergaard L, Ring P, Henriksen O (1996) Myocardial perfusion modeling using MRI. Magn Reson Med 35(5):716–726
Parker GJM, Padhani AR (2004) T1-W DCE-MRI: T1-weighted dynamic contrast-enhanced MRI. In: Quantitative MRI of the brain. Wiley, Ltd, pp 341–364. doi:10.1002/0470869526.ch10
Larsson HB, Courivaud F, Rostrup E, Hansen AE (2009) Measurement of brain perfusion, blood volume, and blood-brain barrier permeability, using dynamic contrast-enhanced T(1)-weighted MRI at 3 tesla. Magn Reson Med 62(5):1270–1281. doi:10.1002/mrm.22136
Patlak CS, Blasberg RG, Fenstermacher JD (1983) Graphical evaluation of blood-to-brain transfer constants from multiple-time uptake data. J Cereb Blood Flow Metab 3(1):1–7. doi:10.1038/jcbfm.1983.1
Mulder WJ, Strijkers GJ, Habets JW, Bleeker EJ, van der Schaft DW, Storm G, Koning GA, Griffioen AW, Nicolay K (2005) MR molecular imaging and fluorescence microscopy for identification of activated tumor endothelium using a bimodal lipidic nanoparticle. FASEB J 19(14):2008–2010. doi:10.1096/fj.05-4145fje
Jarzyna PA, Deddens LH, Kann BH, Ramachandran S, Calcagno C, Chen W, Gianella A, Dijkhuizen RM, Griffioen AW, Fayad ZA, Mulder WJ (2012) Tumor angiogenesis phenotyping by nanoparticle-facilitated magnetic resonance and near-infrared fluorescence molecular imaging. Neoplasia 14(10):964–973
Schiffelers RM, Koning GA, ten Hagen TL, Fens MH, Schraa AJ, Janssen AP, Kok RJ, Molema G, Storm G (2003) Anti-tumor efficacy of tumor vasculature-targeted liposomal doxorubicin. J Control Release 91(1–2):115–122
Neubauer AM, Sim H, Winter PM, Caruthers SD, Williams TA, Robertson JD, Sept D, Lanza GM, Wickline SA (2008) Nanoparticle pharmacokinetic profiling in vivo using magnetic resonance imaging. Magn Reson Med 60(6):1353–1361. doi:10.1002/mrm.21795
Montet X, Montet-Abou K, Reynolds F, Weissleder R, Josephson L (2006) Nanoparticle imaging of integrins on tumor cells. Neoplasia 8(3):214–222. doi:10.1593/neo.05769
van de Ven AL, Kim P, Haley O, Fakhoury JR, Adriani G, Schmulen J, Moloney P, Hussain F, Ferrari M, Liu X, Yun SH, Decuzzi P (2012) Rapid tumoritropic accumulation of systemically injected plateloid particles and their biodistribution. J Control Release 158(1):148–155. doi:10.1016/j.jconrel.2011.10.021
Terreno E, Geninatti Crich S, Belfiore S, Biancone L, Cabella C, Esposito G, Manazza AD, Aime S (2006) Effect of the intracellular localization of a Gd-based imaging probe on the relaxation enhancement of water protons. Magn Reson Med 55(3):491–497. doi:10.1002/mrm.20793
Strijkers GJ, Hak S, Kok MB, Springer CS Jr, Nicolay K (2009) Three-compartment T1 relaxation model for intracellular paramagnetic contrast agents. Magn Reson Med 61(5):1049–1058. doi:10.1002/mrm.21919
Oostendorp M, Douma K, Hackeng TM, van Zandvoort MA, Post MJ, Backes WH (2010) Pharmacokinetics of contrast agents targeted to the tumor vasculature in molecular magnetic resonance imaging. Contrast Media Mol Imaging 5(1):9–17. doi:10.1002/cmmi.361
Pasqualini R, Koivunen E, Kain R, Lahdenranta J, Sakamoto M, Stryhn A, Ashmun RA, Shapiro LH, Arap W, Ruoslahti E (2000) Aminopeptidase N is a receptor for tumor-homing peptides and a target for inhibiting angiogenesis. Cancer Res 60(3):722–727
Wang X, Wang Y, Chen X, Wang J, Zhang X, Zhang Q (2009) NGR-modified micelles enhance their interaction with CD13-overexpressing tumor and endothelial cells. J Control Release 139(1):56–62. doi:10.1016/j.jconrel.2009.05.030
Negussie AH, Miller JL, Reddy G, Drake SK, Wood BJ, Dreher MR (2010) Synthesis and in vitro evaluation of cyclic NGR peptide targeted thermally sensitive liposome. J Control Release 143(2):265–273. doi:10.1016/j.jconrel.2009.12.031
Lakhotia S, Bauer KD, Papoutsakis ET (1993) Fluid-mechanical forces in agitated bioreactors reduce the CD13 and CD33 surface protein content of HL60 cells. Biotechnol Bioeng 41(9):868–877. doi:10.1002/bit.260410906
Acknowledgments
We gratefully thank K. Sæterbø for implanting the dorsal window chambers in mice and culturing cells, S. Andersen, K. Pettersen and S. Moestue for providing and taking care of the mice with subcutaneous xenografts used in the in vivo MRI experiments, and B. Ytterhus and L. Dyrnes for preparation of the histological tumor sections. The work was supported by the Norwegian Cancer Society and Medical Imaging Laboratory (MI-lab, NTNU, Norway) and the National Institute of Health National Cancer Institute Grant R01 CA155432 (W.J.M.M.).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hak, S., Cebulla, J., Huuse, E.M. et al. Periodicity in tumor vasculature targeting kinetics of ligand-functionalized nanoparticles studied by dynamic contrast enhanced magnetic resonance imaging and intravital microscopy. Angiogenesis 17, 93–107 (2014). https://doi.org/10.1007/s10456-013-9380-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10456-013-9380-7