Abstract
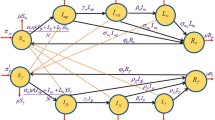
A new multi-stage deterministic model for the transmission dynamics of syphilis, which incorporates disease transmission by individuals in the early latent stage of syphilis infection and the reversions of early latent syphilis to the primary and secondary stages, is formulated and rigorously analysed. The model is used to assess the population-level impact of preventive (condom use) and therapeutic measures (treatment using antibiotics) against the spread of the disease in a community. It is shown that the disease-free equilibrium of the model is globally-asymptotically stable whenever the associated control reproduction number (denoted by \(\mathcal {R}_T\)) is less than unity. A special case of the model is shown to have a unique and globally-asymptotically stable endemic equilibrium whenever the associated reproduction number (denoted by \({\tilde{\mathcal {R}}}_T\)) exceeds unity. Uncertainty and sensitivity analysis of the model, using parameter values and ranges relevant to syphilis transmission dynamics in Nigeria, show that the top three parameters that drive the syphilis infection (with respect to \(\mathcal {R}_T\)) are the disease transmission rate (\(\beta\)), compliance in condom use (c) and efficacy of condom (\(\epsilon _c\)). Numerical simulations of the model show that the targeted treatment of secondary syphilis cases is more effective than the targeted treatment of individuals in the primary or early latent stage of syphilis infection.









Similar content being viewed by others
Notes
www.worldometers.info. Accessed 20 May 2016.
References
Centres for Disease Control and Prevention (2014) Primary and secondary syphilis—United States, 2005–2013. Morb Mortal Wkly Rep 63(18):402–406
Aadland D, Finnoff D, Huang K (2013) Syphilis cycles. J Econ Financ 13(1):297–348
Bartlett JG, Auwaerter PG, Pham PA (2010) John Hopkins POC-IT Center ABX guide: diagnosis and treatment of infectious diseases. Jones and Bartlett Publishers, Burlington
Baughn R, Musher D (2005) Secondary syphilitic lesions. Clin Microbiol Rev 18(1):205–216
Berman SM (2004) Maternal syphilis: pathophysiology and treatment. Bull WHO 82(6):433–438
Birnbaum NR, Goldschmidt RH, Buffett WO (1999) Resolving the common clinical dilemmas of syphilis. Am Fam Physician 59(8):2233–2240
Blower SM, Dowlatabadi H (1994) Sensitivity and uncertainty analysis of complex models of disease transmission: an HIV model, as an example. Int Stat Rev 2:229–243
Breban R, Supervie V, Okano JT, Vardavas R, Blower S (2008) Is there any evidence that syphilis epidemics cycle? Lancet Infect Dis 8(9):577–581
British Columbia HealthLinkBC: stages of syphilis. www.healthlinkbc.ca. Accessed 8 Apr 2018
Cameron CE, Lukehart SA (2014) Current status of syphilis vaccine development: need, challenges, prospects. Vaccine 32(14):1609–9
Center for Substance Abuse Treatment (1993) Screening for infectious diseases among substance abusers. Rockville, MD. Substance abuse and mental health services administration (US): chapter 10, syphilis
Centres for Disease Control and Prevention (2016) Sexually transmitted diseases: syphilis. www.cdc.gov/std/syphilis. Accessed 24 Jan 2016 and 13 June 2016
Centres for Disease Control and Prevention (2018) Syphilis—CDC fact sheet (detailed). www.cdc.gov/std/syphilis. Accessed 8 Apr 2018
Chen Z, Zhang G, Gong X, Lin C, Gao X, Liang G, Yue X, Chen X, Cohen MS (2007) Syphilis in China: results of a national surveillance programme. Lancet 369:132–138
Diekmann O, Heesterbeek J, Metz J (1990) On the definition and the computation of the basic reproduction ratio \(R_0\) in models for infectious diseases in heterogeneous populations. J Math Biol 28:365–382
Doherty L, Fenton K, Jones J, Paine T, Higgins S, Williams D, Palfreeman A (2002) Syphilis: old problem, new strategy. BMJ 325:153–156
Fenton KA, Breban R, Vardavas R, Okano JT, Martin T, Aral S, Blower S (2008) Infectious syphilis in high-income settings in the 21st century. Lancet Infect Dis 8(4):244–253
Garnett GP, Aral SO, Hoyle DV, Cates W, Anderson RM (1997) The natural history of syphilis: implications for the transition dynamics and control of infection. Sex Transm Dis 24(4):185–200
Grassly C, Fraser C, Garnett GP (2005) Host immunity and synchronized epidemics of syphilis across the United States. Nature 433:417–421
Gunn RA, Harper SL, Borntrager DE (2000) Implementing a syphilis elimination and importation control strategy in a low-incidence urban area: san Diego County, california, 1997–1998. Am J Public Health 90:1540–1544
Gutierrez J-P, McPherson S, Fakoya A, Matheou A, Bertozzi SM (2010) Community-based prevention leads to an increase in condom use and a reduction in sexually transmitted infections (STIs) among men who have sex with men (MSM) and female sex workers (FSW): the Frontiers Prevention Project (FPP) evaluation results. BMC Public Health 10:497–508
Hegamin-Younger C, Jeremiah R, Bilbro N (2014) Patterns of Caribbean masculinities and condom compliance among males in Grenada. Am J Men’s Health 8(4):335–338
Hong FC, Yang YZ, Liu XL, Feng TJ, Liu JB, Zhang CL, Lan LN, Yao MZ, Zhou H (2014) Reduction in mother-to-child tranmission of syphilis for 10 years in Shenzhen, China. Sex Transm Dis 41(3):188–193
Iboi E, Okuonghae D (2016) Population dynamics of a mathematical model for syphilis. Appl Math Model 40:3573–3590
Jia Z, Huang X, Wu H, Zhang T, Li N et al (2015) HIV burden in men who have sex with men: a prospective cohort study 2007–2012. Sci Rep 5:11205. https://doi.org/10.1038/srep11205
Karen RD, Susan CW (1999) The effectiveness of condoms in reducing heterosexual transmission of HIV. Fam Plan Perspect 31(6):272–279
LaFond RE, Lukehart SA (2006) Biological basis for syphilis. Clin Microbiol Rev 19:29–49
LaSalle J, Lefschetz S (1976) The stability of dynamical systems. SIAM, Philadelphia
Marfatis YS, Pandya I, Mehta K (2015) Condoms: past, present, and future. Indian J Sex Transm Dis 36(2):133–139
Mercure SA, Savard N (2015) Effectiveness of interventions for syphilis prevention and control in MSM: a summary of published research findings. Report of the National Collaborating Center for Infectious Diseases, Canada
Milner F, Zhao R (2010) A new mathematical model of syphilis. Math Model Nat Phenom 5(6):96–108
Mutagoma M, Remera E, Sebuhoro D, Kanters S, Riedel DJ, Nsanzimana S (2016) The prevalence of syphilis infection and its associated factors in the general population of Rwanda: a national household-based survey. J Sex Transm Dis. https://doi.org/10.1155/2016/4980417
Myint M, Bashiri H, Harrington R, Marra C (2004) Relapse of secondary syphilis after Benzathine Penicillin G: molecular analysis. Sex Transm Dis 31:196–199
Ophori EA, Atanunu O, Johnny EJ, Adu M (2010) Seroprevalence of syphilis in apparently healthy students from a tertiary institution in Benin City, Nigeria. Jpn J Infect Dis 63:437–439
Oswalt R, Matsen K (1993) Sex, AIDS, and the use of condoms: a survey of compliance in college students. Psychol Rep 72(3):764–766
Oxman GL, Smolkowski K, Noell J (1996) Mathematical modelling of epidemic syphilis transmission: implication for syphilis control. Sex Transm Dis 23(1):30–39
Peterman TA, Furness BW (2015) Public health interventions to control syphilis. Sex Health 12:126–134
Pourbohloul B, Rekart ML, Brunham RC (2003) Impact of mass treatment on syphilis transmission: a mathematical modeling approach. Sex Transm Dis 30:297–305
Public Health Agency of Canada (2016) Canadian guidelines on sexually transmitted infections, section 5: management and treatment of specific infections—syphilis. www.phac.aspc.gc.ca. Accessed 28 May 2016
Saad-Roy CM, Shuai Z, van den Driessche P (2016) A mathematical model of syphilis transmission in an MSM population. Math Biosci 277:59–70
Saloojee H, Velaphi S, Goga Y, Afadapa N, Steen R, Lincetto O (2004) The prevention and management of congenital syphilis: an overview and recommendations. Bull WHO 82:424–30
Smolak A, Rowley J, Nagelkerke N, Kassebaum NJ, Chico RM, Korenromp EL, Abu-Raddad LJ (2018) Trends and predictors of syphilis prevalence in the general population: global pooled analyses of 1103 prevalence measures including 136 million syphilis tests. Clin Infect Dis 66(8):1184–1191
Stamm LV (2010) Global challenge of antibiotic-resistant Treponema pallidum. Antimacrob Agents Chemother 54(2):583–589
Stoltey JE, Cohen SE (2015) Syphilis transmission: a review of the current evidence. Sex Health 12:103–109
This Day Newspaper (2016) Borno with highest cases of syphilis, 24 Mar 2016
Tuddenham S, Ghanem KG (2015) Emerging trends and persistent challenges in the management of adult syphilis. BMC Infect Dis 15:351
van den Driessche P, Watmough J (2002) Reproduction numbers and sub-threshold endemic equilibria for compartmental models of disease transmission. Math Biosci 180:29–48
World Bank (2016) Life expectancy at birth. data.worldbank.org. Accessed 12 May 2016
World Health Organization (2014a) Eliminating congenital syphilis. www.who.int/reproductive-health/stis/syphilis.html. Accessed 6 Mar 2014
World Health Organization (2014b) Sexually transmitted infections: syphilis. www.data.euro.who.int. Assessed 17 Apr 2014
World Health Organization (2018) Sexual and reproductive health report: mother-to-child transmission of syphilis. www.who.int. Accessed 8 Apr 2018
Yang L-G, Tucker JD, Yang B, Shen S-Y, Sun X-F, Chen Y-F, Chen X-S (2010) Primary syphilis cases in Guangdong Province 1995–2008: opportunities for linking syphilis control and regional development. BMC Public Health 10:793–799
Zhou Y, Li X, Yang H, Hong Y, Fang X, Wang B, Stanton B (2012) Effect of condom-use measures in predicting sexually transmitted diseases: variation by individuals and contextual factors of sexual risk. Int J STD AIDS 23:27–34
Acknowledgements
The authors are grateful to the anonymous reviewers for their constructive comments.
Author information
Authors and Affiliations
Corresponding author
Appendices
Appendix 1: Proof of Theorem 3.1
Proof
Consider the following linear Lyapunov function
where,
The Lyapunov derivative (where a dot represents differentiation with respect to time t) is given by:
Thus, \(\dot{\mathcal {F}}\le 0\) whenever \(\mathcal {R}_T\le 1\). Hence, \(\dot{\mathcal {F}}\le 0\) if \(\mathcal {R}_T \le 1\) with \(\dot{\mathcal {F}}=0\) if and only if \(I_p\) = \(I_s\) = \(L_e =\) 0. Therefore, \(\mathcal {F}\) is a Lyapunov function in \(\mathcal {D}\). Thus,
as \(t\rightarrow \infty\). Substituting \({E(t)=I_p(t)=I_s(t)=L_e(t)=L_l(t)=I_t(t)=R(t)=0}\) into the first two equation of the model (2.2) gives (\(S_u(t),S_r(t))\rightarrow (\frac{\Lambda }{\mu },0)\), as \(t\rightarrow \infty\). Hence, it follows from the LaSalle’s Invariance Principle (LaSalle and Lefschetz 1976), that
as \(t\rightarrow \infty\), for \(\mathcal {R}_T\le 1\). \(\square\)
Appendix 2: Proof of Theorem 3.2
Consider the model (2.2) with \(\omega = \alpha = 0\) and \(\eta _l = \eta _s = 1\), given by (3.2). Recall that \(\tilde{\mathcal {R}}_T = \mathcal {R}_T|_{\omega =\alpha =0,\eta _l=\eta _s=1}\). The infection rate (\(\lambda\)), given by (2.1), can be expressed at endemic steady-state as:
Solving the variables of the model (in this case, model (3.2)) at steady state gives:
where,
and,
Substituting the expressions in (7.2) into (7.1) (noting the expressions in (7.3) and (7.4)) results in the following polynomial (in \(\lambda ^{**}\)):
where,
It follows from (7.6) that, when \(\tilde{\mathcal {R}}_T > 1\), the coefficient \(c_0\) is negative. Since \(a_0 > 0\), regardless of the sign of \(b_0\), the polynomial (7.5) will have only one positive root when \(\tilde{\mathcal {R}}_T > 1\). Hence, the reduced model (3.2) has a unique endemic equilibrium whenever \(\tilde{\mathcal {R}}_T > 1\).
Remark 7.1
It can be seen that the polynomial (7.5) will not have a positive root when \(\tilde{\mathcal {R}}_T < 1\). In particular, it follows from (7.5), with (7.6), that when \(\tilde{\mathcal {R}}_T < 1\), the coefficient \(c_0\) is positive. For the sign of the coefficient \(b_0\), when \(\tilde{\mathcal {R}}_T < 1\), it is convenient to solve for \(b_0\) when \(\tilde{\mathcal {R}}_T < 1\). It should, first of all, be noted that, the requirement \(\tilde{\mathcal {R}}_T < 1\) implies that
Substituting (7.7) into the expression for \(b_0\) in (7.6), and simplifying, gives
Thus, for the case with \(\tilde{\mathcal {R}}_T < 1\), the quadratic (7.5) has no positive root when \(\tilde{\mathcal {R}}_T < 1\) (since the coefficients \(a_0\), \(b_0\) and \(c_0\) are positive in this case); hence, there is no endemic equilibrium point when \(\tilde{\mathcal {R}}_T < 1\).
To prove the global asymptotic stability of the unique endemic equilibrium of the reduced model (3.2), consider the following nonlinear Lyapunov function (of Goh-Volterra type) defined in \(\mathcal {D}_r\backslash \mathcal {D}_0\).
where, now, \(K_5 = \gamma _4 + \tau _3 + \mu\). The Lyapunov derivative is
Substituting the derivatives in (3.2) into (7.10) gives
It follows from the equations of the reduced model (3.2), at steady-state, that
Substituting the expressions in (7.12) into (7.11) gives
from which it follows that, since the arithmetic mean exceeds the geometric mean,
Thus, \(\dot{V} \le 0\) for \(\tilde{\mathcal {R}}_T > 1\). Hence, V is a lyapunov function for the reduced model (3.2) in \(\mathcal {D}_r\backslash \mathcal {D}_0\). The proof is concluded as in the proof of Theorem 3.1. \(\square\)
Appendix 3: Cumulative Number of New Cases When \(\eta _s < 1\)
Here, we present some numerical simulations of the model (2.2) showing the cumulative number of new syphilis cases when \(\eta _s < 1\). See Sect. 4.3 for discussions on the figures shown in this “Appendix”.
Simulations of the model (2.2) for the treatment-only strategy, showing the cumulative number of new cases, as a function of time, where treatment is applied to only one stage of syphilis infection while there are no treatments for the other stages of infection, in the absence of condom use. a Low treatment rate and b high treatment rate. Parameter values used are as in Table 2, with \(\beta = 0.0069\), \(c = \varepsilon _c = 0\) and \(\eta _s = 0.5\)
Simulations of the model (2.2) for the treatment-only strategy, showing the cumulative number of new cases, as a function of time, in the absence of condom use, varying treatment rate for the targeted infection group while the treatment rates for the other four infected groups are set to zero. Parameter values used are as in Table 2, with \(\beta = 0.0069\), \(c = \varepsilon _c = 0\) and \(\eta _s = 0.5\). a\(\tau _1\), b\(\tau _2\) and c\(\tau _3\)
Simulations of the model (2.2) for the universal strategy, showing the cumulative number of new cases, as a function of time, in the presence of condom use, varying treatment rate for the targeted infection group, while treatment rates for the other four infected groups are set to zero. Parameter values used are as in Table 2, with \(\beta = 0.0069\), \(c = 0.54\), \(\varepsilon _c = 0.8\) and \(\eta _s = 0.5\). a\(\tau _1\), b\(\tau _2\) and c\(\tau _3\)
Rights and permissions
About this article
Cite this article
Okuonghae, D., Gumel, A.B., Ikhimwin, B.O. et al. Mathematical Assessment of the Role of Early Latent Infections and Targeted Control Strategies on Syphilis Transmission Dynamics. Acta Biotheor 67, 47–84 (2019). https://doi.org/10.1007/s10441-018-9336-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10441-018-9336-9