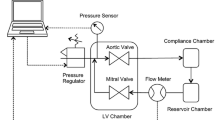
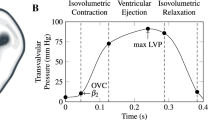
Abstract
Functional mitral regurgitation (FMR) is an insidious and poorly understood condition affecting patients with myocardial disease. While current treatments reduce regurgitation, their ability to reverse mitral valve pathology is unclear. We utilized a pseudo-physiological flow loop to study how repair impacted valve composition. Porcine mitral valves were cultured in control geometry (native papillary muscle position and annular area) or high-tension FMR geometry (5 mm apical and 5 mm lateral displacement of papillary muscles, 65% increased annular area) for 2 weeks. To mimic repair, a reversal condition was created by returning one-week FMR conditioned valves to a non-regurgitant geometry and culturing for 1 week. Valve composition and material properties were analyzed. After two-week culture, FMR conditioned tissues were stiffer and stronger than control and underwent extensive fibrotic remodeling, with increased prolyl-4-hydroxylase, lysyl oxidase, matrix metalloproteinase-1, and decorin. The reversal condition displayed a heterogeneous, leaflet- and orientation-dependent response. Reversal-conditioned anterior leaflets and circumferential tissue sections continued to have significant fibrotic remodeling compared to control, whereas reversal-conditioned posterior leaflets, chordae tendineae, and radial tissue sections had significantly decreased remodeling compared to FMR-conditioned tissues. These findings suggest current repairs only partially reverse pathology, underscoring the need for innovation in the treatment of FMR.








Similar content being viewed by others
Abbreviations
- MV:
-
Mitral valve
- FMR:
-
Functional mitral regurgitation
- PM:
-
Papillary muscle
- GAG:
-
Glycosaminoglycan
- LOX:
-
Lysyl oxidase
- MMP-1:
-
Matrix metalloproteinase-1
- RUFLS:
-
Rice University flow loop system
- VIC:
-
Valve interstitial cell
- ECM:
-
Extracellular matrix
- LVAD:
-
Left ventricular assist device
References
Bail, D. H. L. Treatment of functional mitral regurgitation by percutaneous annuloplasty using the carillon mitral contour system-currently available data state. J. Interv. Cardiol. 30:156–162, 2017.
Balaoing, L. R., A. D. Post, H. Liu, K. T. Minn, and K. J. Grande-Allen. Age-related changes in aortic valve hemostatic protein regulation. Arterioscler. Thromb. Vasc. Biol. 34:72–80, 2014.
Carew, E. O., and I. Vesely. A new method of estimating gauge length for porcine aortic valve test specimens. J. Biomech. 36:1039–1042, 2003.
Connell, P. S., A. F. Azimuddin, S. E. Kim, F. Ramirez, M. S. Jackson, S. H. Little, and K. J. Grande-Allen. Regurgitation hemodynamics alone cause mitral valve remodeling characteristic of clinical disease states in vitro. Ann. Biomed. Eng. 44:954–967, 2016.
Connell, P. S., D. P. Vekilov, and K. J. Grande-Allen. Control of environment—redesign of flow loop bioreactor to control mitral valve regurgitation. In: Engineering 3D Tissue Test Systems, edited by K. J. L. Burg, D. Dreau, and T. Burg. Boca Raton: Taylor & Francis, 2017, pp. 61–73.
Crick, S. J., M. N. Sheppard, S. Y. Ho, L. Gebstein, and R. H. Anderson. Anatomy of the pig heart : comparisons with normal human cardiac structure. J. Anat. 193:105–119, 1998.
De Bonis, M., and O. Alfieri. The edge-to-edge technique for mitral valve repair. HSR Proc. Intensive Care Cardiovasc. Anesth. 2:7–17, 2010.
Fattouch, K., G. Murana, S. Castrovinci, C. Mossuto, R. Sampognaro, M. G. Borruso, E. C. Bertolino, G. Caccamo, G. Ruvolo, and P. Lancellotti. Mitral valve annuloplasty and papillary muscle relocation oriented by 3-dimensional transesophageal echocardiography for severe functional mitral regurgitation. J. Thorac. Cardiovasc. Surg. 143:S38–S42, 2012.
Feldman, T., S. Kar, M. Rinaldi, P. Fail, J. Hermiller, R. Smalling, P. L. Whitlow, W. Gray, R. Low, H. C. Herrmann, S. Lim, E. Foster, and D. Glower. Percutaneous mitral repair with the mitraclip system: safety and midterm durability in the initial everest (endovascular valve edge-to-edge repair study) cohort. J. Am. Coll. Cardiol. 54:686–694, 2009.
Fisher, L. W., J. T. Stubbs, and M. F. Young. Antisera and cdna probes to human and certain animal model bone matrix noncollagenous proteins. Acta Orthop. Scand. Suppl. 266:61–65, 1995.
Fukamachi, K. Percutaneous and off-pump treatments for functional mitral regurgitation. J. Artif. Organs 11:12–18, 2008.
Fukamachi, K., Z. B. Popović, M. Inoue, K. Doi, S. Schenk, Y. Ootaki, M. W. Kopcak, and P. M. McCarthy. Changes in mitral annular and left ventricular dimensions and left ventricular pressure-volume relations after off-pump treatment of mitral regurgitation with the coapsys device. Eur. J. Cardio-Thorac. Surg. 25:352–357, 2004.
Gheewala, N., K. A. Schwarz, and K. J. Grande-Allen. Organ culture of porcine mitral valves as a novel experimental paradigm. Cardiovasc. Eng. Technol. 4:139–150, 2013.
Gheewala, N., and K. J. Grande-Allen. Design and mechanical evaluation of a physiological mitral valve organ culture system. Cardiovasc. Eng. Technol. 1:123–131, 2010.
Grande-Allen, K. J., J. E. Barber, K. M. Klatka, P. L. Houghtaling, I. Vesely, C. S. Moravec, and P. M. McCarthy. Mitral valve stiffening in end-stage heart failure: evidence of an organic contribution to functional mitral regurgitation. J. Thorac. Cardiovasc. Surg. 130:783–790, 2005.
Grande-Allen, K. J., A. G. Borowski, R. W. Troughton, P. L. Houghtaling, N. R. Dipaola, C. S. Moravec, I. Vesely, and B. P. Griffin. Apparently normal mitral valves in patients with heart failure demonstrate biochemical and structural derangements: an extracellular matrix and echocardiographic study. J. Am. Coll. Cardiol. 45:54–61, 2005.
Harnek, J., J. G. Webb, K. H. Kuck, C. Tschope, A. Vahanian, C. E. Buller, S. K. James, C. P. Tiefenbacher, and G. W. Stone. Transcatheter implantation of the monarc coronary sinus device for mitral regurgitation. JACC Cardiovasc. Interv. 4:115–122, 2011.
He, S., A. A. Fontaine, E. Schwammenthal, A. P. Yoganathan, and R. A. Levine. Integrated mechanism for functional mitral regurgitation: leaflet restriction versus coapting force: in vitro studies. Circulation 96:1826–1834, 1997.
Herovici, C. A polychrome stain for differentiating precollagen from collagen. Stain Technol. 38:204, 1963.
Jimenez, J. H., D. D. Soerensen, Z. He, S. He, and A. P. Yoganathan. Effects of a saddle shaped annulus on mitral valve function and chordal force distribution: an in vitro study. Ann. Biomed. Eng. 31:1171–1181, 2003.
Jimenez, J. H., D. D. Soerensen, Z. He, J. Ritchie, and A. P. Yoganathan. Effects of papillary muscle position on chordal force distribution: an in vitro study. J. Heart Valve Dis. 14:295–302, 2005.
Klotz, S., and A. H. Jan. Danser, and D. Burkhoff. Impact of left ventricular assist device (lvad) support on the cardiac reverse remodeling process. Prog. Biophys. Mol. Biol. 97:479–496, 2008.
Levine, R. A., J. Hung, Y. Otsuji, E. Messas, N. Liel-Cohen, N. Nathan, M. D. Handschumacher, J. L. Guerrero, S. He, A. P. Yoganathan, and G. J. Vlahakes. Mechanistic insights into functional mitral regurgitation. Curr. Cardiol. Rep. 4:125–129, 2002.
Liel-Cohen, N., J. L. Guerrero, Y. Otsuji, M. D. Handschumacher, L. G. Rudski, P. R. Hunziker, H. Tanabe, M. Scherrer-Crosbie, S. Sullivan, and R. A. Levine. Design of a new surgical approach for ventricular remodeling to relieve ischemic mitral regurgitation: insights from 3-dimensional echocardiography. Circulation 101:2756–2763, 2000.
MacHaalany, J., L. Bilodeau, R. Hoffmann, S. Sack, H. Sievert, J. Kautzner, C. Hehrlein, P. Serruys, M. Sénéchal, P. Douglas, and O. F. Bertrand. Treatment of functional mitral valve regurgitation with the permanent percutaneous transvenous mitral annuloplasty system: results of the multicenter international percutaneous transvenous mitral annuloplasty system to reduce mitral valve regurgitation in patients with heart failure trial. Am. Heart J. 165:761–769, 2013.
Mishra, Y. K., S. Mittal, P. Jaguri, and N. Trehan. Coapsys mitral annuloplasty for chronic functional ischemic mitral regurgitation: 1-year results. Ann. Thorac. Surg. 81:42–46, 2006.
Morgan, J. A., R. J. Brewer, H. W. Nemeh, R. Murthy, C. T. Williams, D. E. Lanfear, C. Tita, and G. Paone. Left ventricular reverse remodeling with a continuous flow left ventricular assist device measured by left ventricular end-diastolic dimensions and severity of mitral regurgitation. ASAIO J. 58:574–577, 2012.
Mozaffarian, D., et al. Heart disease and stroke statistics-2016 update a report from the american heart association. Circulation 133:e38–e48, 2016.
Padala, M., L. Gyoneva, and A. P. Yoganathan. Effect of anterior strut chordal transection on the force distribution on the marginal chordae of the mitral valve. J. Thorac. Cardiovasc. Surg. 144:624–633, 2012.
Padala, M., R. A. Hutchison, L. R. Croft, J. H. Jimenez, R. C. Gorman, J. H. Gorman, M. S. Sacks, A. P. Yoganathan, and A. P. Yoganathan. Saddle shape of the mitral annulus reduces systolic strains on the p2 segment of the posterior mitral leaflet. Ann. Thorac. Surg. 88:1499–1504, 2009.
Pedersen, W. R., P. Block, M. Leon, P. Kramer, S. Kapadia, V. Babaliaros, S. Kodali, E. M. Tuzcu, and T. Feldman. Icoapsys mitral valve repair system: percutaneous implantation in an animal model. Catheter. Cardiovasc. Interv. 72:125–131, 2008.
Rich, L., and P. Whittaker. Collagen and picrosirius red staining: a polarized light assessment of fibrillar hue and spatial distribution. Braz. J. Morphol. Sci. 22:97–104, 2005.
Rossi, A., F. L. Dini, P. Faggiano, E. Agricola, M. Cicoira, S. Frattini, A. Simioniuc, M. Gullace, S. Ghio, M. Enriquez-Sarano, and P. L. Temporelli. Independent prognostic value of functional mitral regurgitation in patients with heart failure. a quantitative analysis of 1256 patients with ischaemic and non-ischaemic dilated cardiomyopathy. Heart 97:1675–1680, 2011.
Sakamuri, S. S. V. P., A. Takawale, R. Basu, P. W. M. Fedak, D. Freed, C. Sergi, G. Y. Oudit, and Z. Kassiri. Differential impact of mechanical unloading on structural and nonstructural components of the extracellular matrix in advanced human heart failure. Transl. Res. 172:30–44, 2016.
Stephens, E. H., and K. J. Grande-Allen. Age-related changes in collagen synthesis and turnover in porcine heart valves. J. Heart Valve Dis. 16:672–682, 2007.
Stephens, E. H., T. C. Nguyen, A. Itoh, N. B. Ingels, D. C. Miller, and K. J. Grande-Allen. The effects of mitral regurgitation alone are sufficient for leaflet remodeling. Circulation 118:S243–S249, 2008.
Taramasso, M., P. Denti, N. Buzzatti, M. De Bonis, G. La Canna, A. Colombo, O. Alfieri, and F. Maisano. Mitraclip therapy and surgical mitral repair in patients with moderate to severe left ventricular failure causing functional mitral regurgitation: a single-centre experience. Eur. J. Cardiothorac. Surg. 42:920–926, 2012.
Timek, T. A., P. Dagum, D. T. Lai, D. Liang, G. T. Daughters, N. B. Ingels, and D. C. Miller. Pathogenesis of mitral regurgitation in tachycardia-induced cardiomyopathy. Circulation 104:i47–i53, 2001.
Acknowledgements
The authors would like to acknowledge Dr. Larry Fisher, NIH for his gift of decorin antibody used in this research.
Conflicts of interest
Dr. Connell reports personal fees from Polyvascular Corporation, outside the submitted work. Other authors have no disclosures.
Funding
This work was supported by the National Science Foundation Graduate Research Fellowship Program [1450681 to D.V.]; and an American Heart Association Predoctoral Fellowship [13PRE14110003 to P.C.].
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Arash Kheradvar oversaw the review of this article.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Connell, P.S., Vekilov, D.P., Diaz, C.M. et al. Eliminating Regurgitation Reduces Fibrotic Remodeling of Functional Mitral Regurgitation Conditioned Valves. Ann Biomed Eng 46, 670–683 (2018). https://doi.org/10.1007/s10439-018-1987-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-018-1987-9