Abstract
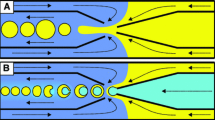
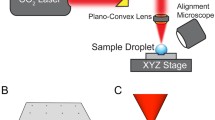
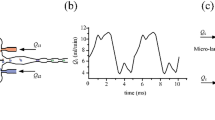
We demonstrated the induced coalescence of droplets under a highly accurate optical force control. Optical scattering and gradient forces were used to push and trap the droplets prior to coalescence within a microfluidic channel. The behavior of the droplets under the influence of an optical force was predicted using an analytical model that agreed well with the experimental data. The optical gradient force accelerated and decelerated the droplet within the laser beam region, and the drag force acting on the droplet was thoroughly characterized. A description of the optical trap was presented in terms of the momentum transfer from the photons to the droplet, effectively restricting droplet motion inside the microfluidic channel prior to coalescence. A phase diagram was plotted to distinguish between the three regimes of droplet coalescence, including the absence of coalescence, coalescence, and multiple coalescence events. The phase diagram permitted the laser power input and the net flow rate in the microfluidic channel to be estimated. This technique was applied to the synthesis of biodegradable gel microparticles.





Similar content being viewed by others
References
Abate AR, Hung T, Mary P, Agresti JJ, Weitz DA (2010) High-throughput injection with microfluidics using picoinjectors. Proc Natl Acad Sci USA 107:19163–19166
Ahn K, Agresti J, Chong H, Marquez M, Weitz DA (2006) Dielectrophoretic manipulation of drops for high-speed microfluidic sorting devices. Appl Phys Lett 88:264105
Ashkin A, Dziedzic JM (1971) Optical levitation by radiation pressure. Appl Phys Lett 19:283–285
Baroud CN, de Saint Vincent MR, Delville JP (2007) An optical toolbox for total control of droplet microfluidics. Lab Chip 7:1029–1033
Baroud CN, Gallaire F, Dangla R (2010) Dynamics of microfluidic droplets. Lab Chip 10:2032–2045
Chang SC, Su YC (2010) On-demand micro-encapsulation utilizing on-chip synthesis of semi-permeable alginate-PLL capsules. Microfluid Nanofluid 10:1165–1174
Christopher GF, Bergstein J, End NB, Poon M, Nguyen C, Anna SL (2009) Coalescence and splitting of confined droplets at microfluidic junctions. Lab Chip 9:1102–1109
Churski K, Kaminski TS, Jakiela S, Kamysz W, Baranska-Rybak W, Weibel DB, Garstecki P (2012) Rapid screening of antibiotic toxicity in an automated microdroplet system. Lab Chip 12:1629–1637
Dholakia K, Cizmar T (2011) Shaping the future of manipulation. Nat Photonics 5:335–342
Dholakia K, Reece P (2006) Optical micromanipulation takes hold. Nano Today 1:18–27
Grier DG (2003) A revolution in optical manipulation. Nature 424:810–816
Hoang PH, Park H, Kim DP (2011) Ultrafast and continuous synthesis of unaccommodating inorganic nanomaterials in droplet- and ionic liquid-assisted microfluidic system. J Am Chem Soc 133:14765–14770
Hung LH, Choi KM, Tseng WY, Tan YC, Shea KJ, Lee AP (2006) Alternating droplet generation and controlled dynamic droplet fusion in microfluidic device for CdS nanoparticle synthesis. Lab Chip 6:174–178
Jung JH, Lee KH, Lee KS, Ha BH, Oh YS, Sung HJ (2014) Optical separation of droplets on a microfluidic platform. Microfluid Nanofluid 16:635–644
Kim SB, Kim SS (2006) Radiation forces on spheres in loosely focused Gaussian beam: ray-optics regime. J Opt Soc Am B 23:897–903
Kotz KT, Gu Y, Faris GW (2005) Optically addressed droplet-based protein assay. J Am Chem Soc 127:5736–5737
Lee DH, Lee W, Um E, Park JK (2011) Microbridge structures for uniform interval control of flowing droplets in microfluidic networks. Biomicrofluidics 5:34117–341179
Lee KH, Kim SB, Yoon SY, Lee KS, Jung JH, Sung HJ (2012) Behavior of double emulsions in a cross-type optical separation system. Langmuir 28:7343–7349
Lorenz RM, Edgar JS, Jeffries GD, Zhao Y, McGloin D, Chiu DT (2007) Vortex-trap-induced fusion of femtoliter-volume aqueous droplets. Anal Chem 79:224–228
Luong TD, Nguyen NT, Sposito A (2012) Thermocoalescence of microdroplets in a microfluidic chamber. Appl Phys Lett 100:254105
Niu X, Gulati S, Edel JB, deMello AJ (2008) Pillar-induced droplet merging in microfluidic circuits. Lab Chip 8:1837–1841
Song H, Chen DL, Ismagilov RF (2006) Reactions in droplets in microfluidic channels. Angew Chem Int Ed 45:7336–7356
Srinivasan V, Pamula VK, Fair RB (2007) An integrated digital microfluidic lab-on-a-chip for clinical diagnostics on human physiological fluids. Lab Chip 4:310–315
Sugiura S, Oda T, Izumida Y, Aoyagi Y, Satake M, Ochiai A, Ohkohchi N, Nakajima M (2005) Size control of calcium alginate beads containing living cells using micro-nozzle array. Biomaterials 26:3327–3331
Tan YC, Ho YL, Lee AP (2006) Droplet coalescence by geometrically mediated flow in microfluidic channels. Microfluid Nanofluid 3:495–499
Teh SY, Lin R, Hung LH, Lee AP (2008) Droplet microfluidics. Lab Chip 8:198–220
Tewhey R, Warner JB, Nakano M, Libby B, Medkova M, David PH, Kotsopoulos SK, Samuels ML, Hutchison JB, Larson JW, Topol EJ, Weiner MP, Harismendy O, Olson J, Link DR, Frazer KA (2009) Microdroplet-based PCR enrichment for large-scale targeted sequencing. Nat Biotechnol 27:1025–1031
Theberge AB, Mayot E, El Harrak A, Kleinschmidt F, Huck WT, Griffiths AD (2012) Microfluidic platform for combinatorial synthesis in picolitre droplets. Lab Chip 12:1320–1326
Um E, Rha E, Choi SL, Lee SG, Park JK (2012) Mesh-integrated microdroplet array for simultaneous merging and storage of single-cell droplets. Lab Chip 12:1594–1597
van der Sman RG (2010) Drag force on spheres confined on the center line of rectangular microchannels. J Colloid Interface Sci 351:43–49
Whitesides GM (2006) The origins and the future of microfluidics. Nature 442:368–373
Acknowledgments
This work was supported by the Creative Research Initiatives Program (No. 2014-001493) of the National Research Foundation of Korea (MSIP).
Author information
Authors and Affiliations
Corresponding author
Additional information
Jin Ho Jung and Kyung Heon Lee have contributed equally to this article.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary material 1 (AVI 5178 kb)
Rights and permissions
About this article
Cite this article
Jung, J.H., Lee, K.H., Destgeer, G. et al. In situ seriate droplet coalescence under an optical force. Microfluid Nanofluid 18, 1247–1254 (2015). https://doi.org/10.1007/s10404-014-1522-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10404-014-1522-8