Abstract
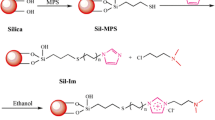
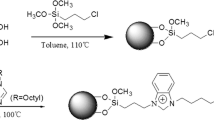
An octyl-quaternary ammonium (Sil-C8-QA)-modified silica stationary phase was prepared by a two-step vapor deposition method which can substantially reduce the consumption of toxic organic solvents. The structure of the dual-functional ligands stationary phase was characterized by Fourier transform infrared spectroscopy and solid-state 13C CP/MAS NMR. Following elemental analysis, the surface density of octyl and quaternary ammonium bonding amount is 2.13 µmol m−2 and 0.77 µmol m−2, respectively. The efficiency and selectivity of the two-ligand phases were compared with two sole functional ligand phases: quaternary ammonium (Sil-QA)- and octyl-modified silica (Sil-C8). Chromatographic evaluation demonstrated a mixed-mode reversed-phase/anion-exchange retention property on the Sil-C8-QA column. The retention behavior of a homologous series of alkylbenzoic acids on the synthesized phase was systematically studied by changing the pH, ionic, and solvent strength of the mobile phase. A well-established mixed-model mechanism was confirmed. The Sil-C8-QA phase can predominantly enhance the retention of acidic drugs and improve their selectivity. It was also successfully applied to the simultaneous separation and determination of three kinds of compound preparations with large differences in component polarity.
Graphic abstract







Similar content being viewed by others
References
Yan JL, Li M, Li RY, Wu LP, Sun YF, Li HL (2013) Chin J Tissue Eng Res 33:6055–6060
Li QP, Liu XM, Dai JD (2014) China J Chin Mater Med 39:402–406
Snyder LR, Kirkland JJ, Dolan JW (2010) Introduction to modern liquid chromatography, 3rd edn. Wiley, Weinheim
Fekete S, Oláh E, Fekete J (2012) J Chromatogr A 1228:57–71
Qiu HD, Liang XJ, Sun M, Jiang SX (2011) Anal Bioanal Chem 399:3307–3322
Yang Y, Geng XD (2011) J Chromatogr A 1218:8813–8825
Hu K, Feng SX, Wu MX, Wang S, Zhao WJ, Jiang Q, Yu AJ, Zhang SS (2014) Talanta 130:63–70
Sun M, Qiu HD, Wang LC, Liu X, Jiang SX (2009) J Chromatogr A 1216:3904–3909
Sun M, Feng JJ, Luo CN, Liu X, Jiang SX (2013) Talanta 105:135–141
Sun M, Feng JJ, Liu SJ, Xiong CM, Liu X, Jiang SX (2011) J Chromatogr A 1218:3743–3749
Zimmermann A, Greco R, Walker I, Horak J, Cavazzini A (2014) J Chromatogr A 1354:43–55
Yang F, Bai Q, Zhao KL, Gao D, Tian L (2015) Anal Bioanal Chem 407:1721–1734
Shen GB, Zhang FF, Yang BC, Chu CG, Liang XM (2013) Talanta 115:129–132
Qiao LZ, Wang SY, Li H, Shan YH, Dou A, Shi XZ, Xu GW (2014) J Chromatogr A 1360:240–247
Iverson CD, Lucy CA (2014) J Chromatogr A 1373:17–24
Sun M, Feng JJ, Wang XW, Duan HM, Li LL, Luo CN (2014) J Sep Sci 37:2153–2159
Abbood A, Smadja C, Herrenknecht C, Alahmad Y, Tchapla A, Taverna M (2009) J Chromatogr A 1216:3244–3251
Abbood A, Smadja C, Taverna M, Herrenknecht C (2010) J Chromatogr A 1217:450–458
Liu QX, Liang T, Li KY, Ke YX, Jin Y, Liang XM (2012) J Sep Sci 35:2685–2692
Wikström P, Mandenius CF, Larsson PO (1988) J Chromatogr A 455:105–117
Li RS, Wan QH (2018) Chromatographia 81:1623–1630
Chen M, Chen L (2018) Anal Methods 10:1538–1546
Guo ZQ, Chen F, Deng Y, Wan QH, Chen L (2009) Chem J Chin Univ 30:2017–2023
Giri S, Inostroza-Rivera R, Herrera B, Núñez AS, Lund F, Toro-Labbé A (2014) J Mol Model 20:2353
Cao W, Wang ZQ, Zeng QL, Shen CH (2016) Appl Surf Sci 389:404–410
Huang XJ, Wang JD, Liu XL, Shang ZH (2001) Chromatographia 54:350–354
Berendsen G, Galan LD (1978) J Liq Chromatogr 5:561–586
Zhang MY, Wang HB, Chen H, Sun XL, Jiang R (2011) Acta Chim Slov 58:60–68
Hu K, Zhang WF, Yang HX, Cui YX, Zhang JY, Zhao WJ, Yu AJ, Zhang SS (2016) Talanta 152:392–400
Ståhlberg J (1999) J Chromatogr A 855:3–55
Ford MS (1969) Analyst 91:15–22
Yang X, Dai J, Carr PW (2003) J Chromatogr A 996:13–31
Neue UD, Tran KV, Méndez A, Carr PW (2005) J Chromatogr A 1063:35–45
Li LX, Lai CY, Xuan XY, Gao CK, Li N (2016) J Chromatogr Sci 54:1415–1420
Wang YH, Liu GR, Fu CG, Wang XS, Cao XQ, Wang SE (2006) Chin J Pharm Anal 26:692–695
Burdan F (2002) Hum Exp Toxicol 21:25–31
Acknowledgements
We thank Prof. Qian-Hong Wan from Tianjin University for his technical assistance with chromatography evaluation. We also thank Dr. Xinghua Jin of Tianjin University for performing the elemental analysis.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhang, F., Chen, L. Synthesis of Octyl-Quaternary Ammonium Mixed-Mode Stationary Phase by Vapor Deposition Approach and Its Application in Compound Preparation Separation. Chromatographia 82, 1437–1447 (2019). https://doi.org/10.1007/s10337-019-03774-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10337-019-03774-1