Abstract
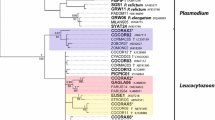
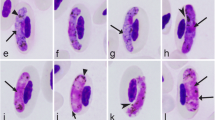
Blood samples from 94 coal tits (Parus ater), 56 great tits (Parus major) and 219 pied flycatchers (Ficedula hypoleuca), caught between 1993 and 2002 at two localities in Lower Saxony, Germany, were examined for haemosporidian infection by parasite-specific polymerase chain reaction (PCR). A simple PCR targeting the 18 SSU rRNA gene of the parasites was used for rapid screening of the samples and generated a total infection prevalence of 20.6% (76/369): 6.8% (n = 15) of the pied flycatchers, 19.1% (n = 18) of the coal tits and 76.8% (n = 43) of the great tits were infected. The positive specimens were re-examined by a cytochrome b gene-directed nested PCR producing significantly longer DNA fragments (approx. 520 bp) that were sequenced and analysed against GenBank-deposited nucleotide sequences. In various numbers (once to 30 times), a total of 13 parasitic DNA sequences differing from 2.9 to 8.5% (13–45 nucleotides) were demonstrated in the three bird species. Due to similarities of 98–100% with GenBank entries, 11 sequences could be assigned to Plasmodium sp. and two to the genus Haemoproteus. In summary, 57 birds were infected with Plasmodium and 19 with Haemoproteus, corresponding to 15.4 and 5.1% of all birds examined, and to 75 and 25% of all birds tested positive. As the only defined species, Haemoproteus majoris was identified in 17 great tits.
Similar content being viewed by others
References
Arctander P (1988) Comparative studies on avian DNA restriction fragment length polymorphism analysis: convenient procedures based on blood samples from live birds. J Ornithol 129:205–216
Bennett GF, Bishop MA, Peirce MA (1993) Checklist for the avian species of Plasmodium Marchiafava & Celli, 1885 (Apicomplexa) and their distribution by avian family and Wallacean life zones. Syst Parasitol 26:171–179
Bennett GF, Peirce MA, Earlé RA (1994) An annotated checklist of the valid avian species of Haemoproteus, Leucocytozoon (Apicomplexa: Haemosporida) and Hepatozoon (Apicomplexa: Haemogregarinidae). Syst Parasitol 29:61–73
Bensch S, Stjernman M, Hasselquist D, Östman Ö, Hansson B, Westerdahl H, Pinheiro RT (2000) Host specificity in avian blood parasites: a study of Plasmodium and Haemoproteus mitochondrial DNA amplified from birds. Proc R Soc Lond B 267:1583–1589
Bensch S, Perez-Tris J, Waldenström J, Hellgren O (2004) Linkage between nuclear and mitochondrial DNA sequences in avian malaria parasites: multiple cases of cryptic speciation. Evolution 58:1617–1621
Bishop MA, Bennett GF (1992) Host–parasite catalogue of the avian haematozoa, supplement 1. Occas Pap Biol (Mem Univ Newfoundland) 15:1–211
Brün J, Winkel W, Epplen JT, Lubjuhn T (1996) Elternschaftsnachweise bei Trauerschnäppern Ficedula hypoleuca am Westrand ihres mitteleuropäischen Verbreitungsareals. J Ornithol 137:435–446
Corradetti A, Garnham PCC, Laird M (1963) New classification of the avian malaria parasites. Parassitologia 5:1–4
Fallis AM, Desser SS, Khan RA (1974) On species of Leucocytozoon. Adv Parasitol 12:1–67
Gabaldon A, Ulloa G, de Montcourt AG (1974) Encuesta sobre malaria aviara en Venezuela: resultados del primer año. Bol Direc Malariol Saneam Ambient 14:80–104
Gabaldon A, Ulloa G, de Montcourt AG (1975) Encuesta sobre malaria aviara en Venezuela: resultados del segundo año. Bol Direc Malariol Saneam Ambient 15:73–92
Gabaldon A, Ulloa G, de Montcourt AG (1976) Encuesta sobre malaria aviara en Venezuela: resultados del tercer y último año. Bol Direc Malariol Saneam Ambient 16:107–118
Garnham PCC (1966) Malaria parasites and other haemosporidia. Blackwell, Oxford
Greiner EC, Bennett GF, White EM, Coombs RF (1975) Distribution of the avian hematozoa of North America. Can J Zool 53:1762–1787
Haberkorn A (1984) Observations on malaria in European perching birds (Passeriformes). Zbl Bakt Hyg A 256:288–295
Hellgren O, Waldenström J, Bensch S (2004) A new PCR assay for simultaneous studies of Leucocytozoon, Plasmodium, and Haemoproteus from avian blood. J Parasitol 90:797–802
Hulier E, Petour P, Snounou G, Nivez M-P, Miltgen F, Mazier D, Renia L (1996) A method for the quantitative assessment of malaria parasite development in organs of the mammalian host. Mol Biochem Parasitol 77:127–135
Kissinger JC, Souza PC, Soares CO, Paul R, Wahl AM, Rathore D, McCutchan TF, Krettli AU (2002) Molecular phylogenetic analysis of the avian malarial parasite Plasmodium (Novyella) juxtanucleare. J Parasitol 88:769–773
Kronberger H, Schüppel K-F (1977) Zwanzig Jahre postmortale Untersuchungen von Vögeln. Verhandlungsber Erkr Zootiere 19:153–169
Krone O, Priemer J, Streich J, Sömmer J, Langgemach T, Lessow O (2001) Haemosporida of birds of prey and owls from Germany. Acta Protozool 40:281–289
Kučera J (1981) Blood parasites of central Europe. 1. Survey of literature. The incidence in domestic birds and general remarks to the incidence in wild birds. Folia Parasitol 28:13–22
Lindt S, Hörning B (1966) Über Malaria und Pinguine. Verhandlungsber Erkr Zootiere 8:223–231
Lubjuhn T, Sauer KP (1999) DNA fingerprinting and profiling in behavioural ecology. In: Epplen JT, Lubjuhn T (eds) DNA profiling and DNA fingerprinting. Birkhäuser, Basel, pp 39–52
Lubjuhn T, Brün J, Winkel W, Muth S (1998) Effects of blood sampling in great tits. J Field Ornithol 69:595–602
Lubjuhn T, Gerken T, Brün J, Epplen JT (1999a) High frequency of extra-pair paternity in the coal tit. J Avian Biol 30:229–233
Lubjuhn T, Strohbach S, Brün J, Gerken T, Epplen JT (1999b) Extra-pair paternity in great tits (Parus major)—a long term study. Behaviour 136:1157–1172
Lubjuhn T, Winkel W, Epplen JT, Brün J (2000) Reproductive success of monogamous and polygynous pied flycatchers (Ficedula hypoleuca). Behav Ecol Sociobiol 48:12–17
Manwell RD (1935) How many species of avian malaria parasites are there? Am J Trop Med 15:265–282
Massey JG, Graczyk TK, Cranfield MR (1996) Characteristics of naturally acquired Plasmodium relictum capistranoae infections in naïve Hawaiian crows (Corvus hawaiiensis) in Hawaii. J Parasitol 82:182–185
McClure HE, Poonswad P, Greiner EC, Laird M (1978) Haematozoa in the birds of eastern and southern Asia. Mem Univ Newfoundland, St. John’s, Newfoundland
Miller SA, Dykes DD, Polesky HF (1988) A simple salting procedure for extracting DNA from human nucleated cells. Nucleic Acids Res 16:1215
Olsen OW (1974) Animal parasites, their life cycles and ecology. University Park Press, Baltimore
Perkins SL (2000) Species concepts and malaria parasites: detecting a cryptic species of Plasmodium. Proc R Soc Lond B 267:2345–2350
Perkins SL, Schall JJ (2002) A molecular phylogeny of malarial parasites recovered from cytochrome b gene sequences. J Parasitol 88:972–978
Pérez-Tris J, Hasselquist D, Hellgren O, Krizanauskiene A, Waldenström J, Bensch S (2005) What are malaria parasites? Trends Parasitol 21:209–211
Rathore D, Wahl AM, Sullivan M, McCutchan TF (2001) A phylogenetic comparison of gene trees constructed from plastid, mitochondrial and genomic DNA of Plasmodium species. Mol Biochem Parasitol 114:89–94
Ricklefs RE, Fallon SM (2002) Diversification and host switching in avian malaria parasites. Proc R Soc Lond B 269:885–892
Rintamäki PT, Halonen M, Kilpimaa J, Lundberg A. (1997) Blood parasites found in three passerine species during spring migration. Ornis Fenn 74:195–200
Rintamäki PT, Ojanen M, Pakkala H, Tynjälä M, Lundberg A (2000) Blood parasites of juvenile willow tits Parus montanus during autumn migration in northern Finland. Ornis Fenn 77:83–87
Schmoll T, Dietrich V, Winkel W, Epplen JT, Lubjuhn T (2003) Longterm fitness consequences of female extra-pair matings in a socially monogamous passerine. Proc R Soc Lond B 270:259–264
Schmoll T, Dietrich V, Winkel W, Lubjuhn T (2004) Blood sampling does not affect fledging success and fledgling local recruitment in coal tits (Parus ater). J Ornithol 145:79–80
Seed TM, Manwell RD (1977) Plasmodia of birds. In: Kreier JP (ed) Parasitic protozoa, vol 3. Academic, New York, pp 311–357
Smyth JD (1976) Introduction to animal parasitology, 2nd edn. Halsted Press, Wiley, New York, pp 112–121
Szymanski MM, Lovette IJ (2005) High lineage diversity and host sharing of malarial parasites in a local avian assemblage. J Parasitol 91:768–774
Valentin A, Haberkorn A, Hensch B, Jakob W (1994) Massive Malaria-Infektionen mit Parahaemoproteus spec. in Schnee-eulen (Nyctea scandiaca) und deren Behandlung mit Primaquin. Verhandlungsber Erkr Zootiere 36:401–404
Valkiūnas G (2005) Avian malaria parasites and other haemosporidia. CRC Press, Boca Raton
Valkiūnas G, Iezhova TA (2001) A comparison of the blood parasites in three subspecies of the yellow wagtail Motacilla flava. J Parasitol 87:930–934
Valkiūnas G, Anwar AM, Atkinson CT, Greiner EC, Paperna I, Peirce MA (2005) What distinguishes malaria parasites from other pigmented haemosporidians? Trends Parasitol 21:357–358
Waldenström J, Bensch S, Hasselquist D, Östman Ö (2004) A new nested polymerase chain reaction method very efficient in detecting Plasmodium and Haemoproteus infections from avian blood. J Parasitol 90:191–194
Woodworth BL, Atkinson CT, LaPointe DA, Hart PJ, Spiegell CS, Tweed EJ, Hennemann C, LeBrun J, Denette T, de Mots R, Kozar KL, Triglia D, Lease D, Gregor A, Smith T, Duffy D (2005) Host population persistence in the face of introduced vector-borne diseases: Hawaii amakihi and avian malaria. Proc Natl Acad Sci USA 102:1531–1536
Acknowledgements
We thank all people who participated in collecting avian blood samples (they are acknowledged in the publications cited in the respective part of the Methods section) and S. Bleidissel, M. Orland and C. Wallnisch (Institute for Evolutionary Biology and Ecology, University of Bonn) for technical laboratory assistance. The studies for which the blood samples were originally obtained were supported by the Deutsche Forschungsgemeinschaft (Lu572/1 and Lu572/2), while the specific aims of this study were supported by the Deutsche Ornithologen-Gesellschaft.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by F. Bairlein.
Rights and permissions
About this article
Cite this article
Wiersch, S.C., Lubjuhn, T., Maier, W.A. et al. Haemosporidian infection in passerine birds from Lower Saxony. J Ornithol 148, 17–24 (2007). https://doi.org/10.1007/s10336-006-0094-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10336-006-0094-0