Abstract
Objectives
Synthetic magnetic resonance imaging (SyMRI) allows to obtain different weighted-images using the multiple-dynamic multiple-echo sequence lasting 6 min. The aim is to compare quantitatively and qualitatively synthetic- and conventional MRI in patients with multiple sclerosis (MS) and controls assessing the contrast (C), the signal to noise ratio (SNR), and the contrast to noise ratio (CNR). We evaluated the lesion count and lesion-to-white matter contrast (\({\text{C}}_{{\text{l } - \text{ WM}}} {)}\) in the MS patients.
Methods and methods
51 patients underwent synthetic- and conventional MRI. Qualitative analysis was evaluated by assigning scores to all synthetic- and conventional MRI sequences by two neuroradiologists. Lesions were counted in MS patients both in the conventional- and synthetic T2-FLAIR. Regions of interest were placed in the cerebrospinal fluid, in the white- and grey matter. For the sequences were evaluated: C, CNR, and SNR.
Results
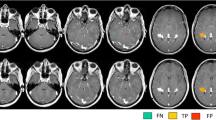
Synthetic T2-FLAIR images are qualitatively inferior. C and CNR were significantly higher in synthetic T1W and T2W images compared to conventional images, but not for T2-FLAIR. The SNR value was always lower in synthetic images than in conventional ones.
Conclusions
SyMRI can be used in clinical practice because it has a similar diagnostic accuracy which reduces the scanning time compared to the conventional one. However, synthetic T2-FLAIR images need to be improved.



Similar content being viewed by others
References
Maitra R, Riddles JJ (2010) Synthetic magnetic resonance imaging revisited. IEEE Trans Med Imaging. https://doi.org/10.1109/TMI.2009.2039487
Riederer SJ, Lee JN, Farzaneh F, Wang HZ, Wright RC (1986) Magnetic resonance image synthesis. Clinical implementation. Acta Radiol Suppl 369:466–468
Betts AM, Leach JL, Jones BV, Zhang B, Serai S (2016) Brain imaging with synthetic MR in children: clinical quality assessment. Neuroradiology. https://doi.org/10.1007/s00234-016-1723-9
West H, Leach JL, Jones BV, Care M, Radhakrishnan R, Merrow AC, Alvarado E, Serai SD (2017) Clinical validation of synthetic brain MRI in children: initial experience. Neuroradiology. https://doi.org/10.1007/s00234-016-1765-z
Lee SM, Choi YH, Cheon JE, Kim IO, Cho SH, Kim WH, Kim HJ, Cho HH, You SK, Park SH, Hwang MJ (2017) Image quality at synthetic brain magnetic resonance imaging in children. Pediatr Radiol. https://doi.org/10.1007/s00247-017-3913-y
McAllister A, Leach J, West H, Jones B, Zhang B, Serai S (2017) Quantitative synthetic MRI in children: normative intracranial tissue segmentation values during development. Am J Neuroradiol. https://doi.org/10.3174/ajnr.A5398
Park S, Kwack KS, Lee YJ, Gho SM, Lee HY (2017) Initial experience with synthetic MRI of the knee at 3T: comparison with conventional T1weighted imaging and T2mapping. Br J Radiol. https://doi.org/10.1259/bjr.20170350
Boudabbous S, Neroladaki A, Bagetakos I, Hamard M, Delattre BM, Vargas MI (2018) Feasibility of synthetic MRI in knee imaging in routine practice. Acta Radiol Open. https://doi.org/10.1177/2058460118769686
Larsson HBW, Frederiksen J, Kjær L, Henriksen O, Olesen J (1988) In vivo determination of T1and T2in the brain of patients with severe but stable multiple sclerosis. Magn Reson Med. https://doi.org/10.1002/mrm.1910070106
Hasan KM, Walimuni IS, Abid H, Wolinsky JS, Narayana PA (2012) Multi-modal quantitative MRI investigation of brain tissue neurodegeneration in multiple sclerosis. J Magn Reson Imaging. https://doi.org/10.1002/jmri.23539
Townsend TN, Bernasconi N, Pike GB, Bernasconi A (2004) Quantitative analysis of temporal lobe white matter T2 relaxation time in temporal lobe epilepsy. Neuroimage. https://doi.org/10.1016/j.neuroimage.2004.06.009
Mamere AE, Saraiva LAL, Matos ALM, Carneiro AAO, Santos AC (2009) Evaluation of delayed neuronal and axonal damage secondary to moderate and severe traumatic brain injury using quantitative MR imaging techniques. Am J Neuroradiol. https://doi.org/10.3174/ajnr.A1477
Granziera C, Daducci A, Donati A, Bonnier G, Romascano D, Roche A, Bach Cuadra M, Schmitter D, Klöppel S, Meuli R, Von Gunten A, Krueger G (2015) A multi-contrast MRI study of microstructural brain damage in patients with mild cognitive impairment. NeuroImage Clin. https://doi.org/10.1016/j.nicl.2015.06.003
Bobman SA, Riederer SJ, Lee JN, Suddarth SA, Wang HZ, Drayer BP, MacFall JR (1985) Cerebral magnetic resonance image synthesis. Am J Neuroradiol 6:265–269
Granberg T, Uppman M, Hashim F, Cananau C, Nordin LE, Shams S, Berglund J, Forslin Y, Aspelin P, Fredrikson S, Kristoffersen-Wiberg M (2016) Clinical feasibility of synthetic MRI in multiple sclerosis: a diagnostic and volumetric validation study. Am J Neuroradiol. https://doi.org/10.3174/ajnr.A4665
Krauss W, Gunnarsson M, Nilsson M, Thunberg P (2018) Conventional and synthetic MRI in multiple sclerosis: a comparative study. Eur Radiol. https://doi.org/10.1007/s00330-017-5100-9
Blystad I, Warntjes JBM, Smedby O, Landtblom AM, Lundberg P, Larsson EM (2012) Synthetic MRI of the brain in a clinical setting. Acta Radiol. https://doi.org/10.1258/ar.2012.120195
Tanenbaum LN, Tsiouris AJ, Johnson AN, Naidich TP, DeLano MC, Melhem ER, Quarterman P, Parameswaran SX, Shankaranarayanan A, Goyen M, Field AS (2017) Synthetic MRI for clinical neuroimaging: results of the magnetic resonance image compilation (MAGiC) prospective, multicenter, multireader trial. Am J Neuroradiol. https://doi.org/10.3174/ajnr.A5227
Fedorov A, Beichel R, Kalpathy-Cramer J, Finet J, Fillion-Robin JC, Pujol S, Bauer C, Jennings D, Fennessy F, Sonka M, Buatti J, Aylward S, Miller JV, Pieper S, Kikinis R (2012) 3D slicer as an image computing platform for the Quantitative Imaging Network. Magn Reson Imaging. https://doi.org/10.1016/j.mri.2012.05.001
Hagiwara A, Warntjes M, Hori M, Andica C, Nakazawa M, Kumamaru KK, Abe O, Aoki S (2017) SyMRI of the brain: rapid quantification of relaxation rates and proton density, with synthetic MRI, automatic brain segmentation, and myelin measurement. Invest Radiol. https://doi.org/10.1097/RLI.0000000000000365
Hagiwara A, Hori M, Yokoyama K, Takemura MY, Andica C, Tabata T, Kamagata K, Suzuki M, Kumamaru KK, Nakazawa M, Takano N, Kawasaki H, Hamasaki N, Kunimatsu A, Aoki S (2017) Synthetic MRI in the detection of multiple sclerosis plaques. Am J Neuroradiol. https://doi.org/10.3174/ajnr.A5012
Ryu K, Nam Y, Gho S, Jang J, Lee H, Cha J, Baek HJ, Park J, Kim D (2019) Data-driven synthetic MRI FLAIR artifact correction via deep neural network. J Magn Reson Imaging. https://doi.org/10.1002/jmri.26712
Hagiwara A, Otsuka Y, Hori M, Tachibana Y, Yokoyama K, Fujita S, Andica C, Kamagata K, Irie R, Koshino S, Maekawa T, Chougar L, Wada A, Takemura MY, Hattori N, Aoki S (2019) Improving the quality of synthetic FLAIR images with deep learning using a conditional generative adversarial network for pixel-by-pixel image translation. Am J Neuroradiol. https://doi.org/10.3174/ajnr.A5927
Bedell BJ, Narayana PA (1998) Implementation and evaluation of a new pulse sequence for rapid acquisition of double inversion recovery images for simultaneous suppression of white matter and CSF. J Magn Reson Imaging. https://doi.org/10.1002/jmri.1880080305
Nelson F, Poonawalla AH, Hou P, Huang F, Wolinsky JS, Narayana PA (2007) Improved identification of intracortical lesions in multiple sclerosis with phase-sensitive inversion recovery in combination with fast double inversion recovery MR imaging. Am J Neuroradiol. https://doi.org/10.3174/ajnr.A0645
Blystad I, Håkansson I, Tisell A, Ernerudh J, Smedby LP, Larsson EM (2016) Quantitative MRI for analysis of active multiple sclerosis lesions without gadolinium-based contrast agent. Am J Neuroradiol. https://doi.org/10.3174/ajnr.A4501
Hagiwara A, Hori M, Suzuki M, Andica C, Nakazawa M, Tsuruta K, Takano N, Sato S, Hamasaki N, Yoshida M, Kumamaru KK, Ohtomo K, Aoki S (2016) Contrast-enhanced synthetic MRI for the detection of brain metastases. Acta Radiol Open. https://doi.org/10.1177/2058460115626757
Funding
Supported by research project "Magic-MRI"—Villa Benedetta Group.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no financial activities related to the present article.
Ethical approval
This prospective, single-centre, Health Insurance Portability and Accountability Act-compliant study was approved by the Institutional Review Board.
Informed consent
Patients gave written informed consent before enrolment.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Di Giuliano, F., Minosse, S., Picchi, E. et al. Comparison between synthetic and conventional magnetic resonance imaging in patients with multiple sclerosis and controls. Magn Reson Mater Phy 33, 549–557 (2020). https://doi.org/10.1007/s10334-019-00804-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10334-019-00804-9