Abstract
Objective
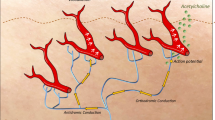
The aim of this study was to report a method that quantifies axon reflex sweating from individual sweat glands with nanoliter precision. Measurement of the axon reflex is generally expressed as a single variable (e.g., the flare area or total sweat volume). High-definition videography enables precise measurement of sweating from single, axon reflex-stimulated sweat glands (SGs).
Methods
The sudomotor axon reflex was activated in healthy subjects and subjects with peripheral neuropathy by iontophoresis of 10% acetylcholine. Sweating was simultaneously imaged for 5 min in a 2.5-cm2 area of iodine-coated skin to one side of the stimulus, using a customized high-resolution camera with starch-coated transparent tape over a rigid viewing screen. A second video then imaged the directly stimulated sweating. The indirect sweat response was quantified in terms of sweat gland number and distance from the stimulation site (radius), sweat rate per gland, and total sweat.
Results
Fifty-two healthy control and twenty subjects with neuropathy underwent testing at the foot, calf, thigh, and hand. Normal ranges were calculated for SG density, mean sweat rate per SG, and total sweat volume. Neuropathy subjects demonstrated reduced sweating, and values differed between body sites.
Interpretation
The described method precisely measures the total and individual sweat output of hundreds of SGs in response to a standard, axon reflex-mediated stimulus, and quantifies alterations in axon reflex sweating seen in peripheral neuropathy.



Similar content being viewed by others
References
Kennedy WR, Navarro X (1989) Sympathetic sudomotor function in diabetic neuropathy. Arch Neurol 46(11):1182–1186
Hoeldtke RD, Bryner KD, Horvath GG, Phares RW, Broy LF, Hobbs GR (2001) Redistribution of sudomotor responses is an early sign of sympathetic dysfunction in type 1 diabetes. Diabetes 50(2):436–443
Mariani LL, Lozeron P, Theaudin M, Mincheva Z, Signate A, Ducot B, Algalarrondo V, Denier C, Adam C, Nicolas G, Samuel D, Slama MS, Lacroix C, Misrahi M, Adams D, French Familial Amyloid Polyneuropathies Network Study G (2015) Genotype-phenotype correlation and course of transthyretin familial amyloid polyneuropathies in France. Ann Neurol 78(6):901–916. https://doi.org/10.1002/ana.24519
Singer W, Spies JM, McArthur J, Low J, Griffin JW, Nickander KK, Gordon V, Low PA (2004) Prospective evaluation of somatic and autonomic small fibers in selected autonomic neuropathies. Neurology 62(4):612–618
Sumner CJ, Sheth S, Griffin JW, Cornblath DR, Polydefkis M (2003) The spectrum of neuropathy in diabetes and impaired glucose tolerance. Neurology 60(1):108–111
Abraham A, Alabdali M, Alsulaiman A, Breiner A, Barnett C, Katzberg HD, Lovblom LE, Perkins BA, Bril V (2016) Laser doppler flare imaging and quantitative thermal thresholds testing performance in small and mixed fiber neuropathies. PLoS One 11(11):e0165731. https://doi.org/10.1371/journal.pone.0165731
Siepmann T, Gibbons CH, Illigens BM, Lafo JA, Brown CM, Freeman R (2012) Quantitative pilomotor axon reflex test: a novel test of pilomotor function. Arch Neurol 69(11):1488–1492. https://doi.org/10.1001/archneurol.2012.1092
Thaisetthawatkul P, Fernandes Filho JA, Herrmann DN (2013) Contribution of QSART to the diagnosis of small fiber neuropathy. Muscle Nerve 48(6):883–888. https://doi.org/10.1002/mus.23891
Sharma S, Venkitaraman R, Vas PR, Rayman G (2015) Assessment of chemotherapy-induced peripheral neuropathy using the LDIFLARE technique: a novel technique to detect neural small fiber dysfunction. Brain Behav 5(7):e00354. https://doi.org/10.1002/brb3.354
Kubasch ML, Kubasch AS, Torres Pacheco J, Buchmann SJ, Illigens BM, Barlinn K, Siepmann T (2017) Laser Doppler assessment of vasomotor axon reflex responsiveness to evaluate neurovascular function. Front Neurol 8:370. https://doi.org/10.3389/fneur.2017.00370
Illigens BM, Siepmann T, Roofeh J, Gibbons CH (2013) Laser doppler imaging in the detection of peripheral neuropathy. Auton Neurosci Basic Clin 177(2):286–290. https://doi.org/10.1016/j.autneu.2013.06.006
Low VA, Sandroni P, Fealey RD, Low PA (2006) Detection of small-fiber neuropathy by sudomotor testing. Muscle Nerve 34(1):57–61. https://doi.org/10.1002/mus.20551
Low PA, Denq JC, Opfer-Gehrking TL, Dyck PJ, O’Brien PC, Slezak JM (1997) Effect of age and gender on sudomotor and cardiovagal function and blood pressure response to tilt in normal subjects. Muscle Nerve 20(12):1561–1568
Gibbons CH, Illigens BM, Centi J, Freeman R (2008) QDIRT: quantitative direct and indirect test of sudomotor function. Neurology 70(24):2299–2304. https://doi.org/10.1212/01.wnl.0000314646.49565.c0
Gibbons CH, Illigens BM, Wang N, Freeman R (2010) Quantification of sudomotor innervation: a comparison of three methods. Muscle Nerve 42(1):112–119. https://doi.org/10.1002/mus.21626
Low PA, Caskey PE, Tuck RR, Fealey RD, Dyck PJ (1983) Quantitative sudomotor axon reflex test in normal and neuropathic subjects. Ann Neurol 14(5):573–580. https://doi.org/10.1002/ana.410140513
Low PA, Vernino S, Suarez G (2003) Autonomic dysfunction in peripheral nerve disease. Muscle Nerve 27(6):646–661. https://doi.org/10.1002/mus.10333
Low PA (1993) Composite autonomic scoring scale for laboratory quantification of generalized autonomic failure. Mayo Clin Proc 68(8):748–752
Kennedy WR, Selim MM, Wendelschaefer-Crabbe G, Blaes AH, Suter F, Nolano M, Provitera V (2013) A device to quantify sweat in single sweat glands to diagnose neuropathy. J Med Devices 7(3):2
Provitera V, Nolano M, Caporaso G, Stancanelli A, Santoro L, Kennedy WR (2010) Evaluation of sudomotor function in diabetes using the dynamic sweat test. Neurology 74(1):50–56. https://doi.org/10.1212/WNL.0b013e3181c7da4b
Loavenbruck AJ, Hodges JS, Provitera V, Nolano M, Wendelshafer-Crabb G, Kennedy WR (2017) A device to measure secretion of individual sweat glands for diagnosis of peripheral neuropathy. J Peripher Nerv Syst 22(2):139–148. https://doi.org/10.1111/jns.12212
Lindsay SL, Holmes S, Corbett AD, Harker M, Bovell DL (2008) Innervation and receptor profiles of the human apocrine (epitrichial) sweat gland: routes for intervention in bromhidrosis. Br J Dermatol 159(3):653–660. https://doi.org/10.1111/j.1365-2133.2008.08740.x
Low PA, Opfer-Gehrking TL, Kihara M (1992) In vivo studies on receptor pharmacology of the human eccrine sweat gland. Clin Auton Res 2(1):29–34
Schulz IJ (1969) Micropuncture studies of the sweat formation in cystic fibrosis patients. J Clin Invest 48(8):1470–1477. https://doi.org/10.1172/JCI106113
Sato K, Nishiyama A, Kobayashi M (1979) Mechanical properties and functions of the myoepithelium in the eccrine sweat gland. Am J Physiol 237(3):C177–C184. https://doi.org/10.1152/ajpcell.1979.237.3.C177
Randall WC, Calman C (1954) The surface tension of human sweat; its determination and its significance. J Invest Dermatol 23(2):113–118
Sato K (1980) Pharmacological responsiveness of the myoepithelium of the isolated human axillary apocrine sweat gland. Br J Dermatol 103(3):235–243
Sato K (1973) Sweat induction from an isolated eccrine sweat gland. Am J Physiol 225(5):1147–1152. https://doi.org/10.1152/ajplegacy.1973.225.5.1147
Loavenbruck A, Wendelschaefer-Crabbe G, Sandroni P, Kennedy WR (2014) Quantification of sweat gland volume and innervation in neuropathy: correlation with thermoregulatory sweat testing. Muscle Nerve 50(4):528–534. https://doi.org/10.1002/mus.24185
Waller LA, Sarkka A, Olsbo V, Myllymaki M, Panoutsopoulou IG, Kennedy WR, Wendelschafer-Crabb G (2011) Second-order spatial analysis of epidermal nerve fibers. Stat Med 30(23):2827–2841. https://doi.org/10.1002/sim.4315
Myllymaki M, Panoutsopoulou IG, Sarkka A (2012) Analysis of spatial structure of epidermal nerve entry point patterns based on replicated data. J Microsc 247(3):228–239. https://doi.org/10.1111/j.1365-2818.2012.03636.x
Kennedy WR, Nolano M, Wendelschafer-Crabb G, Johnson TL, Tamura E (1999) A skin blister method to study epidermal nerves in peripheral nerve disease. Muscle Nerve 22(3):360–371
Schlereth T, Brosda N, Birklein F (2005) Somatotopic arrangement of sudomotor axon reflex sweating in humans. Auton Neurosci Basic Clin 123(1–2):76–81. https://doi.org/10.1016/j.autneu.2005.08.004
Schlereth T, Brosda N, Birklein F (2005) Spreading of sudomotor axon reflexes in human skin. Neurology 64(8):1417–1421. https://doi.org/10.1212/01.WNL.0000158473.60148.FE
Schmidt R, Weidner C, Schmelz M (2011) Time course of acetylcholine-induced activation of sympathetic efferents matches axon reflex sweating in humans. J Peripher Nerv Syst 16(1):30–36. https://doi.org/10.1111/j.1529-8027.2011.00320.x
Schmelz M, Schmidt R, Bickel A, Torebjork HE, Handwerker HO (1998) Innervation territories of single sympathetic C fibers in human skin. J Neurophysiol 79(4):1653–1660
Gibbons CH, Illigens BM, Wang N, Freeman R (2009) Quantification of sweat gland innervation: a clinical-pathologic correlation. Neurology 72(17):1479–1486. https://doi.org/10.1212/WNL.0b013e3181a2e8b8
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Rights and permissions
About this article
Cite this article
Loavenbruck, A., Sit, N., Provitera, V. et al. High-resolution axon reflex sweat testing for diagnosis of neuropathy. Clin Auton Res 29, 55–62 (2019). https://doi.org/10.1007/s10286-018-0546-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10286-018-0546-7