Abstract
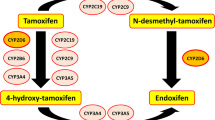
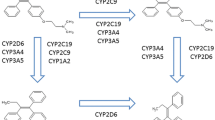
Cytochrome P450 2D6 (CYP2D6) is a key enzyme in the transformation of the Tamoxifen (TAM) in its most active two derivatives, a 4-hydroxy-TAM and Endoxifen. The hypothesis that a genetic polymorphism of CYP2D6 and/or concomitant absorption of these enzyme-inhibitory drugs can modify both the frequency and intensity of hot flushes and the antineoplastic therapeutic efficacy is the object of dissenting conclusions in the available literature. CYP2D6 genotyping is not the object of official recommendation at present before prescription of TAM while a common recommendation of Afssaps and INCa contraindicates the co-prescription of the paroxetine and the fluoxetine with the TAM.
Résumé
Le cytochrome P450 2D6 (CYP2D6) est un enzyme clé de la transformation du tamoxifène (TAM) en ses deux dérivés les plus actifs, le 4-hydroxy-TAM et l’endoxifène. L’hypothèse qu’un polymorphisme génétique du CYP2D6 et/ou la prise concomitante de médicaments inhibiteurs de cet enzyme puissent modifier sous TAM à la fois la fréquence et l’intensité des bouffées de chaleur (BdC) d’une part, et l’efficacité thérapeutique antinéoplasique d’autre part, fait l’objet de conclusions discordantes dans la littérature disponible. Il n’y a pas actuellement de recommandation officielle au génotypage du CYP2D6 avant prescription de TAM alors qu’une recommandation commune de l’Afssaps et de l’INCa contreindique la coprescription de la paroxétine et de la fluoxétine avec le TAM.
Similar content being viewed by others
Références
Ahern TP, Pedersen L, Cronin-Fenton DP, et al. (2009) No increase in breast cancer recurrence with concurrent use of tamoxifen and some CYP2D6-inhibiting medications. Cancer Epidemiol Biomarkers Prev 18: OF1–OF3
Azoulay L, Dell’Aniello S, Huiart L, et al. (2011) Concurrent use of tamoxifen with CYP2D6 inhibitors and the risk of breast cancer recurrence. Breast Cancer Res Treat 126: 695–703
Borges S, Desta Z, Li L, et al. (2006) Quantitative effect of CYP2D6 genotype and inhibitors on tamoxifen metabolism: implication for optimization of breast cancer treatment. Clin Pharmacol Ther 80: 61–74
Boutet G (2012) Traitement des bouffées de chaleur après cancer du sein. Gynecol Obstet Fertil 40: 241–254
Brauch H, Mürdter TE, Eichelbaum M, et al. (2009) Pharmacogenomics of tamoxifen therapy. Clin Chem 55: 1770–1782
Burstein HJ, Prestrud AA, Seidenfeld J, et al. (2010) American Society of Clinical Oncology clinical practice guideline: update on adjuvant endocrine therapy for women with hormone receptor-positive breast cancer. J Clin Oncol 28: 3784–3796
Cuzick J, Forbes JF, Sestak I, et al. (2007) Long-term results of tamoxifen prophylaxis for breast cancer: 96-month follow-up of the randomized IBIS-I Trial. J Natl Cancer Inst 99: 272–282
Cuzick J, Sestac I, Cella D et al. (2008) Treatment-emergent endocrine symptoms and the risk of breast cancer recurrence: a retrospective analysis of the ATAC trial. Lancet Oncol 12: 1143–1148
Early Breast Cancer Trialists’ Collaborative group (EBCTCG) (2011) Relevance of breast cancer hormone receptors and other factors to the efficacy of adjuvant tamoxifen: patient-level meta-analysis of randomised trials. Lancet 378: 771–784
Fisher B, Costantino JP, Wickerham DL, et al. (1998) Tamoxifen for prevention of breast cancer: report of the national surgical adjuvant breast and bowel project P-1 study. J Natl Cancer Inst 90: 1371–1388
Fleeman N, Martin-Saborido C, Payne K, et al. (2011) The clinical effectiveness and cost-effectiveness of genotyping for CYP2D6 for the management of women with breast cancer treated with tamoxifen: a systematic review. Health Technol Assess 15: 1–102
Goetz MP, Rae JM, Suman VJ, et al. (2005) Pharmacogenetics of tamoxifen biotransformation is associated with clinical outcomes of efficacy and hot flasches. J Clin Oncol 23: 9312–9318
Goetz MP, Knox SK, Suman VJ, et al. (2007) The impact of cytochrome P450 2D6 metabolism in women receiving adjuvant tamoxifen. Breast Cancer Res Treat 101: 113–121
Goetz MP, Schaid DJ, Wickerham D, et al. (2011) Evaluation of CYP2D6 and efficacy of tamoxifen and raloxifene in women treated for breast cancer chemoprevention: results from the NSABP P1 and P2 clinical trials. Clin Cancer Res 17: 6944–6951
Henry NL, Rae JM, Li L, et al. (2009) Association between CYP2D6 genotype and tamoxifen-induced hot flashes in a prospective cohort. Breast Cancer Res Treat 117: 571–575
Irvin WJ, Walko CM, Weck KE, et al. (2011) Genotype-guided tamoxifen dosing increases active metabolite exposure in women with reduced CYP2D6 metabolism: a multicenter study. J Clin Oncol 29: 3232–3239
Jin Y, Desta Z, Stearns V, et al. (2005) CYP2D6 genotype, antidepressant use, and tamoxifen metabolism during adjuvant breast cancer treatment. J Natl Cancer Inst 97: 30–39
Kelly CM, Juurlink DN, Gomes T, et al. (2010) Selective serotonin reuptake inhibitors and breast cancer mortality in women receiving tamoxifen: a population based cohort study. BMJ 340:c693
Kelly CM, Pritchard KI (2012) CYP2D6 genotype as a marker for benefit of adjuvant tamoxifen in postmenopausal women: lessons learned. J Natl Cancer Inst 104: 427–428
Lash T, Lien EA, Sorensen HT, et al. (2009) Genotype-guided tamoxifen therapy: time to pause for reflection? Lancet Oncol 10: 825–833
Loprinzi CL, Sloan J, Stearns V, et al. (2009) Newer antidepressants and Gabapentin for hot flashes: an individual patient pooled analysis. J Clin Oncol 27: 2831–2837
Madlensky L, Flatt SW, Natarajan L, et al. (2008) Hot flashes are associated with CYP2D6 genotype in breast cancer survivors taking tamoxifen. 31st Annual San Antonio Breast Cancer Symposium, San Antonio, abstract no 6045
Madlensky L, Natarajan L, Tchu S, et al. (2011) Tamoxifen metabolite concentrations, CYP2D6 genotype, and breast cancer outcomes. Clin Pharmacol Ther 89: 718–724
Mortimer JE, Flatt SW, Parker BA, et al. (2008) Tamoxifen, hot flashes and recurrence in breast cancer. Breast Cancer Rest Treat 108: 421–426
Rae JM, Sikora MJ, Henry NL, et al. (2009) Cytochrome P450 2D6 activity predicts discontinuation of tamoxifen therapy in breast cancer patients. Pharmacogenomics J 9: 258–264
Rae JM (2011) Personalized tamoxifen: what is the best way forward? J Clin Oncol 29: 3206–3208
Rae JM, Drury S, Hayes DF, et al. (2012) CYP2D6 and UGT2B7 genotype and risk of recurrence in tamoxifen-treated breast cancer patients. J Natl Cancer Inst 104: 452–460
Regan MM, Leyland-Jones B, Bouzyk M, et al. (2012) CYP2D6 genotype and tamoxifen response in postmenopausal women with endocrine-responsive breast cancer: the Breast International Group 1-98 trial. J Natl Cancer Inst 104: 441–451
Sideras K, Ingle JN, Ames MM, et al. (2010) Coprescription of tamoxifen and medications that inhibit CYP2D6. J Clin Oncol 28: 2768–2776
Singh MS, Francis PA, Michael M (2011) Tamoxifen, cytochrome P450 genes and breast cancer clinical outcomes. Breast 20: 111–118
Stearns V, Johnson MD, Rae JM, et al. (2003) Active tamoxifen metabolite plasma concentrations after coadministration of tamoxifen and the selective serotonin reuptake inhibitor paroxetine. J Natl Cancer Instit 95: 1758–1764
Thompson AM, Johnson A, Quinlan P, et al. (2011) Comprehensive CYP2D6 genotype and adherence affect outcome in breast cancer patients treated with tamoxifen monotherapy. Breast Cancer Res Treat 125: 279–287
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Boutet, G. Bouffées de chaleur, tamoxifène et cytochrome P450 2D6 . Oncologie 14, 357–364 (2012). https://doi.org/10.1007/s10269-012-2170-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10269-012-2170-9