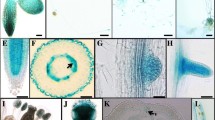
Abstract
This manuscript reports the production of specific polyclonal antibodies for PsPIN1, a putative auxin efflux carrier in Alaska pea (Pisum sativum L.) plants, and the cellular immunolocalization of PsPIN1. When pea seeds were set with the seed axis horizontal to the upper surface of a rockwool block, and allowed to germinate and grow for 3 days in the dark, the epicotyl grew upward. On the other hand, the application of 2,3,5-triiodobenzoic acid (TIBA) inhibited graviresponse. In the subapical epicotyl regions, PsPIN1 has been found to localize in the basal side of the plasma membrane of cells in endodermal tissues. Asymmetric PsPIN1 localization between the proximal and distal sides of the epicotyl was observed, the total amounts of PsPIN1 being more abundant in the proximal side. The asymmetric PsPIN1 distribution between the proximal and distal sides of the epicotyl was well correlated with unequal polar auxin transport as well as asymmetric accumulation of mRNA of PsPIN1 (Ueda et al. in Biol Sci Space 26:32–41, 2012; Ueda et al. in Plant Biol 16(suppl 1):43–49, 2014). In the proximal side of an apical hook, PsPIN1 localized in the basal side of the plasma membrane of cells in endodermal tissues, whereas in the distal side, the abundant distribution of PsPIN1 localized in the basal-lower (endodermal) side of the basal plasma membrane, suggesting possible lateral auxin movement from the distal side to the proximal side in this region. The application of TIBA significantly reduced the amount of PsPIN1 in the proximal side of epicotyls, but little in the distal side. These results suggest that unequal auxin transport in epicotyls during the early growth stage of etiolated pea seedlings is derived from asymmetric PsPIN1 localization in the apical hook and subapical region of epicotyls, and that asymmetric transport between the proximal and distal sides of epicotyls is required for the graviresponse of epicotyls.







Similar content being viewed by others
References
Adamowski M, Friml J (2015) PIN-dependent auxin transport: action, regulation, and evolution. Plant Cell 27:20–32. https://doi.org/10.1105/tpc.114.134874
Bai F, DeMason (2006) Hormone interactions and regulation of Unifoliata. PsPK2, PsPIN1 and LE gene expression in pea (Pisum sativum) shoot tips. Plant Cell Physiol 47:935–948. https://doi.org/10.1093/pcp/pcj066
Bandyopadhyay A, Blakeslee JJ, Lee OR, Mravec J, Sauer M, Titapiwatanakun B, Makam SN, Bouchard R, Geisler M, Martinoia E, Friml J, Peer WA, Murphy AS (2007) Interactions of PIN and PGP auxin transport mechanisms. Biochem Soc Trans 35:137–141. https://doi.org/10.1042/BST0350137
Benjamins R, Malenica N, Luschnig C (2005) Regulating the regulator: the control of auxin transport. Bioessays 27:1246–1255. https://doi.org/10.1002/bies.20322
Berleth T, Sachs T (2001) Plant morphogenesis: long-distance coordination and local pattering. Curr Opin Plant Biol 4:57–62. https://doi.org/10.1016/S1369-5266(00)00136-9
Boutté Y, Crosnier MT, Carraro N, Traas J, Satiat-Jeunemaitre B (2006) The plasma membrane recycling pathway and cell polarity in plants: studies on PIN proteins. J Cell Sci 119:1255–1265. https://doi.org/10.1242/jcs.02847
Carraro N, Forestan C, Canova S, Traas J, Varotto S (2006) ZmPIN1a and ZmPIN1b encode two novel putative candidates for polar auxin transport and plant architecture determination of maize. Plant Physiol 142:254–264. https://doi.org/10.1104/pp.106.080119
Chawla R, DeMason DA (2004) Molecular expression of PsPIN1, a putative auxin efflux carrier gene from pea (Pisum sativum L.). Plant Growth Regul 44:1–14
DeMason DA, Chawla R (2009) Raising anti-PINl polyclonal antibodies for pea. Pisum Genet 41:7–12
Dhonukshe P, Grigoriev I, Fischer R, Tominaga M, Robinson DG, Hasek J, Paciorek T, Petrásek J, Seifertová D, Tejos R, Meisel LA, Zazímalová E, Gadella TW Jr, Stierhof YD, Ueda T, Oiwa K, Akhmanova A, Brock R, Spang A, Friml J (2008) Auxin transport inhibitors impair vesicle motility and actin cytoskeleton dynamics in diverse eukaryotes. Proc Natl Acad Sci USA 105:4489–4494. https://doi.org/10.1073/pnas.0711414105
Friml J, Palme K (2002) Polar auxin transport—old question and new concepts? Plant Mol Biol 49:273–282. https://doi.org/10.1023/A:1015248926412
Friml J, Wiśniewska J, Benková E, Mendgen K, Palme K (2002) Lateral relocation of auxin efflux regulator PIN3 mediates tropism in Arabidopsis. Nature 415:806–809. https://doi.org/10.1038/415806a
Friml J, Vieten A, Sauer M, Weijers D, Schwarz H, Hamann T, Offringa R, Jürgens G (2003) Efflux-dependent auxin gradients establish the apical–basal axis of Arabidopsis. Nature 426:147–153. https://doi.org/10.1038/nature02085
Fukaki H, Wysocka-Diller J, Kato T, Fujisawa H, Benfey PN, Tasaka M (1998) Genetic evidence that the endodermis is essential for shoot gravitropism in Arabidopsis thaliana. Plant J 14:425–430. https://doi.org/10.1046/j.1365-313X.1998.00137.x
Gälweiler L, Guan C, Müller A, Wisman E, Mendgen K, Yephremov A, Palme K (1998) Regulation of polar auxin transport by AtPIN1 in Arabidopsis vascular tissue. Science 282:2226–2230. https://doi.org/10.1126/science.282.5397.2226
Geldner N, Friml J, Stierhof YD, Jürgens G, Palme K (2001) Auxin transport inhibitors block PIN1 cycling and vesicle trafficking. Nature 413:425–428. https://doi.org/10.1038/35096571
Hoshino T, Hitotsubashi R, Miyamoto K, Tanimoto E, Ueda J (2005) Isolation of PsPIN2 and PsAUX1 from etiolated pea epicotyls and their expression on a three-dimensional clinostat. Adv Space Res 36:1284–1294. https://doi.org/10.1016/j.asr.2005.03.121
Hoshino T, Miyamoto K, Ueda J (2006) Requirement for the gravity-controlled transport of auxin for a negative gravitropic response of epicotyls in the early growth stage of etiolated pea seedlings. Plant Cell Physiol 47:1496–1508. https://doi.org/10.1093/pcp/pcl015
Hoshino T, Miyamoto K, Ueda J (2007) Gravity-controlled asymmetrical transport of auxin regulates a gravitropic response in the early growth stage of etiolated pea (Pisum sativum) epicotyls: studies using simulated microgravity conditions on a three-dimensional clinostat and using an agravitropic mutant, ageotropum. J Plant Res 120:619–628. https://doi.org/10.1007/s10265-007-0103-2
Kim JY, Henrichs S, Bailly A, Vincenzetti V, Sovero V, Mancuso S, Pollmann S, Kim D, Geisler M, Nam HG (2010) Identification of an ABCB/P-glycoprotein-specific inhibitor of auxin transport by chemical genomics. J Biol Chem 285:23307–23315. https://doi.org/10.1074/jbc.M110.105981
Kojo KH, Yasuhara H, Hasezawa S (2014) Time-sequential observation of spindle and phragmoplast orientation in BY-2 cells with altered cortical actin microfilament patterning. Plant Signal Behav 9:e29579. https://doi.org/10.4161/psb.29579
Kramer EM, Bennett MJ (2006) Auxin transport: a field in flux. Trends Plant Sci 11:382–386. https://doi.org/10.1016/j.tplants.2006.06.002
Michniewicz M, Brewer PB, Friml J (2007) Polar auxin transport and asymmetric auxin distribution. Arabidopsis Book 5:e0108. https://doi.org/10.1199/tab.0108
Miyamoto K, Hoshino T, Yamashita M, Ueda J (2005) Automorphosis of etiolated pea seedlings in space is simulated by a three-dimensional clinostat and the application of inhibitors of auxin polar transport. Physiol Plant 123:467–474. https://doi.org/10.1111/j.1399-3054.2005.00472.x
Miyamoto K, Uheda E, Oka M, Ueda J (2011) Auxin polar transport and automorphosis in plants. Biol Sci Space 25:57–68. https://doi.org/10.2187/bss.25.57
Miyamoto K, Yamasaki T, Uheda E, Ueda J (2014) Analysis of apical hook formation in Alaska pea with a 3-D clinostat and agravitropic mutant ageotropum. Front Plant Sci 5:137. https://doi.org/10.3389/fpls.2014.00137
Morohashi K, Okamoto M, Yamazaki C, Fujii N, Miyazawa Y, Kamada M, Kasahara H, Osada I, Shimazu T, Fusejima Y, Higashibata A, Yamazaki T, Ishioka N, Kobayashi A, Takahashi H (2017) Gravitropism interferes with hydrotropism via counteracting auxin dynamics in cucumber roots: clinorotation and spaceflight experiments. New Phytol 215:1476–1489. https://doi.org/10.1111/nph.14689
Muday GK, Murphy AS (2002) An emerging model of auxin transport regulation. Plant Cell 14:293–299. https://doi.org/10.1105/tpc.140230
Nishimura T, Nakano H, Hayashi K, Niwa C, Koshiba T (2009) Differential downward stream of auxin synthesized at the tip has a key role in gravitropic curvature via TIR1/AFBs-mediated auxin signaling pathways. Plant Cell Physiol 50:1874–1885. https://doi.org/10.1093/pcp/pcp129
Oka M, Miyamoto K, Okada K, Ueda J (1999) Auxin polar transport and flower formation in Arabidopsis thaliana transformed with indoleacetamide hydrolase (iaaH) gene. Plant Cell Physiol 40:231–237. https://doi.org/10.1093/oxfordjournals.pcp.a029532
Okada K, Shimura Y (1994) Genetic analyses of signalling in flower development using Arabidopsis. Plant Mol Biol 26:1357–1377. https://doi.org/10.1007/BF00016480
Okada K, Ueda J, Komaki MK, Bell CJ, Shimura Y (1991) Requirement of auxin polar transport system in early stage of Arabidopsis floral bud formation. Plant Cell 3:677–684. https://doi.org/10.1105/tpc.3.7.677
Petrásek J, Elčkner M, Morris DA, Zazímalová E (2002) Auxin efflux carrier activity and auxin accumulation regulate cell division and polarity in tobacco cells. Planta 216:302–308. https://doi.org/10.1007/s00425-002-0845-y
Robert HS, Friml J (2009) Auxin and other signals on the move in plants. Nat Chem Biol 5:325–332. https://doi.org/10.1038/nchembio.170
Tanaka H, Kitakura S, Rakusova H, Uemura T, Feraru MI, De Ricke R, Robert S, Kakimoto T, Friml J (2013) Cell polarity and patterning by PIN trafficking through early endosomal compartments in Arabidopsis thaliana. PLOS Genet 9:e1003540. https://doi.org/10.1371/journal.pgen.1003540
Ueda J, Miyamoto K, Yuda T, Hoshino T, Fujii S, Mukai C, Kamigaichi S, Aizawa S, Yoshizaki I, Shimazu T, Fukui K (1999) Growth and development, and auxin polar transport in higher plants under microgravity conditions in space: BRIC-AUX on STS-95 space experiment. J Plant Res 112:487–492. https://doi.org/10.1007/PL00013904
Ueda J, Miyamoto K, Uheda E, Oka M (2011) Auxin transport and graviresponse in plants: relevance to ABC proteins. Biol Sci Space 25:69–75. https://doi.org/10.2187/bss.25.69
Ueda J, Tada T, Hoshino T, Miyamoto K, Uheda E, Oka M (2012) Isolation of PsPINs and PsAUX1 cDNAs encoding putative auxin efflux and influx carriers and/or facilitators, respectively from etiolated epicotyls of an agravitropic pea (Pisum sativum L.) mutant, ageotropum. Biol Sci Space 26:32–41. https://doi.org/10.2187/bss.26.32
Ueda J, Miyamoto K, Uheda E, Oka M, Yano S, Higashibata A, Ishioka N (2014) Close relationships between polar auxin transport and graviresponse in plants. Plant Biol 16(suppl.1):43–49. https://doi.org/10.1111/plb.12101
Ueda J, Saniewski M, Miyamoto K (2016) Chap. 8, Auxins, one major plant hormone, in soil. In: Szajdak LW (ed) Bioactive compounds in agricultural soils. Springer, Cham, pp 175–206. https://doi.org/10.1007/978-3-319-43107-9_8
Watanabe C, Fujii N, Yanai K, Hotta T, Kim DH, Kamada M, Saito Y, Nishimura T, Koshiba T, Miyazawa Y, Kim KM, Takahashi H (2012) Gravistimulation changes the accumulation pattern of CsPIN1 auxin efflux facilitator in endodermis of the transition zone in cucumber seedlings. Plant Physiol 158:239–251. https://doi.org/10.1104/pp.111.188615
Went FW, Thimann KV (1937) Phytohormones. MacMillan, New York
Yamazaki C, Fujii N, Miyazawa Y, Kamada M, Kasahara H, Osada I, Shimazu T, Fusejima Y, Higashibata A, Yamazaki T, Ishioka N, Takahashi H (2016) The gravity-induced re-localization of auxin efflux carrier CsPIN1 in cucumber seedlings: spaceflight experiments for immunohistochemical microscopy. NPJ Microgravity 2:16030. https://doi.org/10.1038/npjmgrav.2016.30
Yoneyama M, Kitayama T, Taniura H, Yoneda Y (2003) Immersion fixation with Carnoy solution for conventional immunohistochemical detection of particular N-methyl-d-aspartate receptor subunits in murine hippocampus. J Neurosci Res 73:416–426. https://doi.org/10.1002/jnr.10622
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Kamada, M., Miyamoto, K., Oka, M. et al. Regulation of asymmetric polar auxin transport by PsPIN1 in endodermal tissues of etiolated Pisum sativum epicotyls: focus on immunohistochemical analyses. J Plant Res 131, 681–692 (2018). https://doi.org/10.1007/s10265-018-1031-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10265-018-1031-z