Abstract
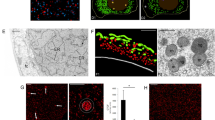
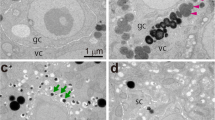
The processes involved in sexual reproduction have been diversified during plant evolution. Whereas charales, bryophytes, pteridophytes, and some gymnosperms utilize motile sperm as male gametes, in other gymnosperms and angiosperms the immotile sperm cells are delivered to the egg cells through elongated pollen tubes. During formation of the motile sperms, cells undergo a dynamic morphological transformation including drastic changes in shape and the generation of locomotor architecture. The molecular mechanism involved in this process remains mostly unknown. Membrane trafficking fulfills the exchange of various proteins and lipids among single membrane-bound organelles in eukaryotic cells, contributing to various biological functions. RAB GTPases and SNARE proteins are evolutionarily conserved key machineries of membrane trafficking mechanisms, which regulate tethering and fusion of the transport vesicles to target membranes. Our observation of fluorescently tagged plasma membrane-resident SNARE proteins demonstrated that these proteins relocalize to spherical structures during the late stages in spermiogenesis. Similar changes in subcellular localization were also observed for other fluorescently tagged SNARE proteins and a RAB GTPase, which acts on other organelles including the Golgi apparatus and endosomes. Notably, a vacuolar SNARE, MpVAMP71, was localized on the membrane of the spherical structures. Electron microscopic analysis revealed that there are many degradation-related structures such as multi-vesicular bodies, autophagosomes, and autophagic bodies containing organelles. Our results indicate that the cell-autonomous degradation pathway plays a crucial role in the removal of membrane components and the cytoplasm during spermiogenesis of Marchantia polymorpha. This process differs substantially from mammalian spermatogenesis in which phagocytic removal of excess cytoplasm involves neighboring cells.



Similar content being viewed by others
References
Carothers ZB, Kreitner GL (1968) Studies of spermatogenesis in the Hepaticae. II. Blepharoplast structure in the spermatid of Marchantia. J Cell Biol 36:603–616
Chiyoda S, Ishizaki K, Kataoka H, Yamato KT, Kohchi T (2008) Direct transformation of the liverwort Marchantia polymorpha L. by particle bombardment using immature thalli developing from spores. Plant Cell Rep 27:1467–1473. doi:10.1007/s00299-008-0570-5
Doelling JH, Walker JM, Friedman EM, Thompson AR, Vierstra RD (2002) The APG8/12-activating enzyme APG7 is required for proper nutrient recycling and senescence in Arabidopsis thaliana. J Biol Chem 277:33105–33114. doi:10.1074/jbc.M204630200
Fujimoto M, Ueda T (2012) Conserved and plant-unique mechanisms regulating plant post-Golgi traffic. Front Plant Sci 3:197. doi:10.3389/fpls.2012.00197
Graham LE, McBride GE (1979) The occurrence and phylogenetic significance of a multilayered structure in Coleochaete spermatozoids. Am J Botany 66:887–894
Griesbeck O, Baird GS, Campbell RE, Zacharias DA, Tsien RY (2001) Reducing the environmental sensitivity of yellow fluorescent protein. Mechanism and applications. J Biol Chem 276:29188–29194. doi:10.1074/jbc.M102815200
Hanamata S, Kurusu T, Kuchitsu K (2014) Roles of autophagy in male reproductive development in plants. Front Plant Sci 5:457. doi:10.3389/fpls.2014.00457
Hanaoka H et al (2002) Leaf senescence and starvation-induced chlorosis are accelerated by the disruption of an Arabidopsis autophagy gene. Plant Physiol 129:1181–1193. doi:10.1104/pp.011024
Hatsugai N et al. (2009) A novel membrane fusion-mediated plant immunity against bacterial pathogens. Genes Dev 23:2496–2506. doi:10.1101/gad.1825209
Henne WM, Buchkovich NJ, Emr SD (2011) The ESCRT pathway. Dev Cell 21:77–91. doi:10.1016/j.devcel.2011.05.015
Higo A et al. (2016) Transcriptional framework of male gametogenesis in the liverwort Marchantia polymorpha L. Plant Cell Physiol 57:325–338. doi:10.1093/pcp/pcw005
Ishizaki K, Chiyoda S, Yamato KT, Kohchi T (2008) Agrobacterium-mediated transformation of the haploid liverwort Marchantia polymorpha L., an emerging model for plant biology. Plant Cell Physiol 49:1084–1091. doi:10.1093/pcp/pcn085
Ishizaki K et al (2015) Development of gateway binary vector series with four different selection markers for the liverwort Marchantia polymorpha. PloS One 10:e0138876. doi:10.1371/journal.pone.0138876
Ishizaki K, Nishihama R, Yamato KT, Kohchi T (2016) Molecular genetic tools and techniques for Marchantia polymorpha research. Plant Cell Physiol 57:262–270. doi:10.1093/pcp/pcv097
Kanazawa T et al. (2016) SNARE molecules in Marchantia polymorpha: unique and conserved features of the membrane fusion machinery. Plant Cell Physiol 57:307–324. doi:10.1093/pcp/pcv076
Kubota A, Ishizaki K, Hosaka M, Kohchi T (2013) Efficient agrobacterium-mediated transformation of the liverwort Marchantia polymorpha using regenerating thalli. Biosci Biotechnol Biochem 77:167–172. doi:10.1271/bbb.120700
Kurusu T et al (2014) OsATG7 is required for autophagy-dependent lipid metabolism in rice postmeiotic anther development. Autophagy 10:878–888. doi:10.4161/auto.28279
Lai Z, Wang F, Zheng Z, Fan B, Chen Z (2011) A critical role of autophagy in plant resistance to necrotrophic fungal pathogens. Plant J 66:953–968. doi:10.1111/j.1365-313X.2011.04553.x
Li Y, Wang FH, Knox RB (1989) Ultrastructural analysis of the flagellar apparatus in sperm cells of ginkgo-biloba. Protoplasma 149:57–63. doi:10.1007/Bf01623983
Liu Y, Xiong Y, Bassham DC (2014) Autophagy is required for tolerance of drought and salt stress in plants. Autophagy 5:954–963. doi:10.4161/auto.5.7.9290
Mizushima N, Komatsu M (2011) Autophagy: renovation of cells and tissues. Cell 147:728–741. doi:10.1016/j.cell.2011.10.026
Nakatogawa H, Suzuki K, Kamada Y, Ohsumi Y (2009) Dynamics and diversity in autophagy mechanisms: lessons from yeast. Nat Rev Mol Cell Biol 10:458–467. doi:10.1038/nrm2708
O’Donnell L, Nicholls PK, O’Bryan MK, McLachlan RI, Stanton PG (2011) Spermiation: The process of sperm release. Spermatogenesis 1:14–35. doi:10.4161/spmg.1.1.14525
Renzaglia KS, Duckett JG (1987) Spermatogenesis in Blasia pusilla: from young antheridium through mature spermatozoid. Bryologist 90:419. doi:10.2307/3243109
Renzaglia KS, Garbary DJ (2001) Motile gametes of land plants: diversity, development, and evolution. Crit Rev Plant Sci 20:107–213. doi:10.1080/20013591099209
Saito C, Ueda T (2009) Chap. 4: functions of RAB and SNARE proteins in plant life. Int Rev Cell Mol Biol 274:183–233. doi:10.1016/S1937-6448(08)02004-2
Sanderfoot A (2007) Increases in the number of SNARE genes parallels the rise of multicellularity among the green plants. Plant Physiol 144:6–17. doi:10.1104/pp.106.092973
Shaner NC, Steinbach PA, Tsien RY (2005) A guide to choosing fluorescent proteins. Nat Methods 2:905–909. doi:10.1038/nmeth819
Shimamura M (2016) Marchantia polymorpha: taxonomy, phylogeny and morphology of a model system. Plant Cell Physiol 57:230–256. doi:10.1093/pcp/pcv192
Ueda K (1979) Denshikenbikyou de Mita Syokubutsu no Kouzou. Baishukan, Tokyo, pp 220–237 (in Japanese)
Uemura T (2016) Physiological roles of plant post-golgi transport pathways in membrane trafficking. Plant Cell Physiol doi:10.1093/pcp/pcw149
van der Sluijs P, Zibouche M, van Kerkhof P (2013) Late steps in secretory lysosome exocytosis in cytotoxic lymphocytes. Front Immunol 4:359. doi:10.3389/fimmu.2013.00359
Vaughn KC, Renzaglia KS (2006) Structural and immunocytochemical characterization of the Ginkgo biloba L. sperm motility apparatus. Protoplasma 227:165–173. doi:10.1007/s00709-005-0141-3
Yoshimoto K (2012) Beginning to understand autophagy, an intracellular self-degradation system in plants. Plant Cell Physiol 53:1355–1365. doi:10.1093/pcp/pcs099
Zhen Y, Stenmark H (2015) Cellular functions of Rab GTPases at a glance. J Cell Sci 128:3171–3176. doi:10.1242/jcs.166074
Acknowledgements
We thank Dr. Atsuko Era for sharing unpublished results. This work was supported by Grants-in-Aid for Scientific Research of the Ministry of Education, Culture, Sports, Science, and Technology of Japan and by a Grant-in-Aid for JSPS Fellows (N.M. and T.K.). This research was also supported by JST, PRESTO.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Minamino, N., Kanazawa, T., Nishihama, R. et al. Dynamic reorganization of the endomembrane system during spermatogenesis in Marchantia polymorpha . J Plant Res 130, 433–441 (2017). https://doi.org/10.1007/s10265-017-0909-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10265-017-0909-5