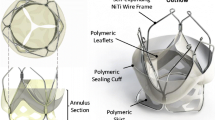
Abstract
It has been proposed that inappropriate positioning of transcatheter aortic valves (TAVs) is associated with procedural complications and decreased device durability. Second-generation TAVs allow for repositioning giving greater control over the final deployment position. However, the impact of positioning on the tissue surrounding these devices needs to be better understood, in particular for the interleaflet triangle in which the conductance system (bundle of His) resides. In this study, we investigate the impact of implantation depth on the frame–tissue interaction for a next-generation repositionable Lotus™ valve. For this purpose, a computational model simulating deployment of the Lotus valve frame into a calcified patient-specific aortic root geometry was generated to predict aortic root stress and frame eccentricity at three different deployment depths. The results of this study predicted that positioning of the Lotus valve had an influence on the stresses in the aortic sinus and frame eccentricity. An analysis of levels of stress arising in the vicinity of the bundle of His, as a function of implantation depth, was conducted, and it was found that, for the specific patient anatomy studied, although the sub-annular position showed reduced peak stress in the aortic sinus, this implantation position showed the highest stress in the area of greatest risks of conductance interference. In contrast, while a supra-annular position increased the peak arterial stress, this implantation position resulted in lower stress in the interleaflet triangle and thus might reduce the risk of conductance interference. These results provide pre-operative information that can inform clinical decision-making regarding TAVI positioning.










Similar content being viewed by others
References
Auricchio F, Taylor RL (1997) Shape-memory alloys: modelling and numerical simulations of the finite-strain superelastic behavior. Comput Methods Appl Mech Eng 143:175–194. https://doi.org/10.1016/S0045-7825(96)01147-4
Auricchio F, Taylor RL, Lubliner J (1997) Shape-memory alloys: macromodelling and numerical simulations of the superelastic behavior. Comput Methods Appl Mech Eng 146:281–312. https://doi.org/10.1016/S0045-7825(96)01232-7
Auricchio F, Conti M, Morganti S, Reali A (2014) Simulation of transcatheter aortic valve implantation: a patient-specific finite element approach. Comput Methods Biomech Biomed Eng 17:1347–1357. https://doi.org/10.1080/10255842.2012.746676
Baan J Jr et al (2010) Factors associated with cardiac conduction disorders and permanent pacemaker implantation after percutaneous aortic valve implantation with the CoreValve prosthesis. Am Heart J 159:497–503. https://doi.org/10.1016/j.ahj.2009.12.009
Block PC (2010) Leaks and the “great ship” TAVI. Catheter Cardiovasc Interv 75:873–874. https://doi.org/10.1002/ccd.22581
Cao C, Ang SC, Vallely MP, Ng M, Adams M, Wilson M (2012) Migration of the transcatheter valve into the left ventricle. Ann Cardiothorac Surg 1:243–244. https://doi.org/10.3978/j.issn.2225-319X.2012.07.11
Capelli C et al (2012) Patient-specific simulations of transcatheter aortic valve stent implantation. Med Biol Eng Compu 50:183–192. https://doi.org/10.1007/s11517-012-0864-1
Charitos EI, Sievers H-H (2013) Anatomy of the aortic root: implications for valve-sparing surgery. Ann Cardiothorac Surg 2:53–56
Colombo A, Latib A (2012) Left bundle branch block after transcatheter aortic valve implantation inconsequential or a clinically important endpoint? J Am Coll Cardiol 60:1753–1755. https://doi.org/10.1016/j.jacc.2012.07.034
Conway C, Sharif F, McGarry JP, McHugh PE (2012) A computational test-bed to assess coronary stent implantation mechanics using a population-specific approach. Cardiovasc Eng Technol 3:374–387. https://doi.org/10.1007/s13239-012-0104-8
Finotello A, Morganti S, Auricchio F (2017) Finite element analysis of TAVI: impact of native aortic root computational modeling strategies on simulation outcomes. Med Eng Phys 47:2–12. https://doi.org/10.1016/j.medengphy.2017.06.045
Fraccaro C et al (2011) Incidence, predictors, and outcome of conduction disorders after transcatheter self-expandable aortic valve implantation. Am J Cardiol 107:747–754. https://doi.org/10.1016/j.amjcard.2010.10.054
Généreux P et al (2013) Paravalvular leak after transcatheter aortic valve replacement the new Achilles’ heel? A comprehensive review of the literature. J Am Coll Cardiol 61:1125–1136. https://doi.org/10.1016/j.jacc.2012.08.1039
Gooley RP, Cameron JD, Meredith IT (2015) Assessment of the geometric interaction between the lotus transcatheter aortic valve prosthesis and the native ventricular aortic interface by 320-multidetector computed tomography. JACC Cardiovasc Interv 8:740–749. https://doi.org/10.1016/j.jcin.2015.03.002
Gunning P (2014) Experimental and computational investigation of the impact of distorted stent expansion on the performance of transcatheter aortic valve replacements. NUI Galway
Gunning PS, Saikrishnan N, McNamara LM, Yoganathan AP (2014a) An in vitro evaluation of the impact of eccentric deployment on transcatheter aortic valve hemodynamics. Ann Biomed Eng 42:1195–1206. https://doi.org/10.1007/s10439-014-1008-6
Gunning PS, Vaughan TJ, McNamara LM (2014b) Simulation of self expanding transcatheter aortic valve in a realistic aortic root: implications of deployment geometry on leaflet deformation. Ann Biomed Eng 42:1989–2001. https://doi.org/10.1007/s10439-014-1051-3
Gunning PS, Saikrishnan N, Yoganathan AP, McNamara LM (2015) Total ellipse of the heart valve: the impact of eccentric stent distortion on the regional dynamic deformation of pericardial tissue leaflets of a transcatheter aortic valve replacement. J R Soc Interface 12:20150737. https://doi.org/10.1098/rsif.2015.0737
Hayashida K et al (2013) Potential mechanism of annulus rupture during transcatheter aortic valve implantation. Catheter Cardiovasc Interv 82:E742–746. https://doi.org/10.1002/ccd.24524
Houthuizen P et al (2012) Left bundle-branch block induced by transcatheter aortic valve implantation increases risk of death. Circulation 126:720–728. https://doi.org/10.1161/circulationaha.112.101055
Hsu M-C, Bazilevs Y (2011) Blood vessel tissue prestress modeling for vascular fluid–structure interaction simulation. Finite Elem Anal Des 47:593–599. https://doi.org/10.1016/j.finel.2010.12.015
Hunter RJ, Liu Y, Lu Y, Wang W, Schilling RJ (2012) Left atrial wall stress distribution and its relationship to electrophysiologic remodeling in persistent atrial fibrillation. Circulation Arrhythm Electrophysiol 5:351
Iung B et al (2003) A prospective survey of patients with valvular heart disease in Europe: the Euro heart survey on valvular heart disease. Eur Heart J 24:1231–1243
Kasel AM, Cassese S, Bleiziffer S, Amaki M, Hahn RT, Kastrati A, Sengupta PP (2013) Standardized imaging for aortic annular sizing implications for transcatheter valve selection. JACC Cardiovasc Imaging 6:249–262. https://doi.org/10.1016/j.jcmg.2012.12.005
Khawaja MZ et al (2011) Permanent pacemaker insertion after core valve transcatheter aortic valve implantation. Circulation 123:951
Loree HM, Grodzinsky AJ, Park SY, Gibson LJ, Lee RT (1994) Static circumferential tangential modulus of human atherosclerotic tissue. J Biomech 27:195–204
Martin C, Pham T, Sun W (2011) Significant differences in the material properties between aged human and porcine aortic tissues. Eur J Cardiothorac Surg 40:28–34. https://doi.org/10.1016/j.ejcts.2010.08.056
Masson J-B et al (2009) Transcatheter aortic valve implantation review of the nature, management, and avoidance of procedural complications. JACC Cardiovasc Interv 2:811–820. https://doi.org/10.1016/j.jcin.2009.07.005
Misfeld M, Sievers H-H (2007) Heart valve macro- and microstructure. Philos Trans R Soc B Biol Sci 362:1421–1436. https://doi.org/10.1098/rstb.2007.2125
Morganti S, Brambilla N, Petronio AS, Reali A, Bedogni F, Auricchio F (2016) Prediction of patient-specific post-operative outcomes of TAVI procedure: the impact of the positioning strategy on valve performance. J Biomech. https://doi.org/10.1016/j.jbiomech.2015.10.048
Mummert J, Sirois E, Sun W (2013) Quantification of biomechanical interaction of transcatheter aortic valve stent deployed in porcine and ovine hearts. Ann Biomed Eng 41:577–586
Ogden RW, Roxburgh DG (1999) A pseudo-elastic model for the Mullins effect in filled rubber. Proc R Soc Lond Ser A Math Phys Eng Sci 455:2861
Pericevic I, Lally C, Toner D, Kelly DJ (2009) The influence of plaque composition on underlying arterial wall stress during stent expansion: the case for lesion-specific stents. Med Eng Phys 31:428–433. https://doi.org/10.1016/j.medengphy.2008.11.005
Piazza N, de Jaegere P, Schultz C, Becker AE, Serruys PW, Anderson RH (2008) Anatomy of the aortic valvar complex and its implications for transcatheter implantation of the aortic valve. Circ Cardiovasc Interv 1:74–81. https://doi.org/10.1161/circinterventions.108.780858
Sirois E, Wang Q, Sun W (2011) Fluid simulation of a transcatheter aortic valve deployment into a patient-specific aortic root. Cardiovasc Eng Technol 2:186–195. https://doi.org/10.1007/s13239-011-0037-7
Sun W, Li K, Sirois E (2010) Simulated elliptical bioprosthetic valve deformation: implications for asymmetric transcatheter valve deployment. J Biomech 43:3085–3090. https://doi.org/10.1016/j.jbiomech.2010.08.010
Tzamtzis S, Viquerat J, Yap J, Mullen MJ, Burriesci G (2013) Numerical analysis of the radial force produced by the Medtronic-CoreValve and Edwards-SAPIEN after transcatheter aortic valve implantation (TAVI). Med Eng Phys 35:125–130. https://doi.org/10.1016/j.medengphy.2012.04.009
Urena M et al (2012) Predictive factors and long-term clinical consequences of persistent left bundle branch block following transcatheter aortic valve implantation with a balloon-expandable valve. J Am Coll Cardiol 60:1743–1752. https://doi.org/10.1016/j.jacc.2012.07.035
Walther T, Blumenstein J, van Linden A, Kempfert J (2012) Contemporary management of aortic stenosis: surgical aortic valve replacement remains the gold standard. Heart 98(Suppl 4):iv23–29. https://doi.org/10.1136/heartjnl-2012-302399
Wang Q, Sirois E, Sun W (2012) Patient-specific modeling of biomechanical interaction in transcatheter aortic valve deployment. J Biomech 45:1965–1971. https://doi.org/10.1016/j.jbiomech.2012.05.008
Wang Q, Kodali S, Primiano C, Sun W (2015) Simulations of transcatheter aortic valve implantation: implications for aortic root rupture. Biomech Model Mechanobiol 14:29–38. https://doi.org/10.1007/s10237-014-0583-7
Zahedmanesh H, John Kelly D, Lally C (2010) Simulation of a balloon expandable stent in a realistic coronary artery—determination of the optimum modelling strategy. J Biomech 43:2126–2132. https://doi.org/10.1016/j.jbiomech.2010.03.050
Acknowledgements
The authors acknowledge funding from the Irish Researcher Council Enterprise Partnership Scheme Postgraduate Scholarship and the Irish Centre for High-End Computing (ICHEC).
Funding
This study was funded by an Irish Research Council Enterprise Partnership Scheme Postgraduate Scholarship 2014 in collaboration with Boston Scientific (EPSPG/2014/120).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Orla M. McGee and Professor Laoise M. McNamara are collaborating with Boston Scientific under an Irish Research Council Enterprise Partnership Scheme Postgraduate Scholarship.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
McGee, O.M., Gunning, P.S., McNamara, A. et al. The impact of implantation depth of the Lotus™ valve on mechanical stress in close proximity to the bundle of His. Biomech Model Mechanobiol 18, 79–88 (2019). https://doi.org/10.1007/s10237-018-1069-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10237-018-1069-9