Abstract
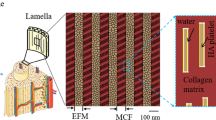
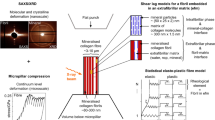
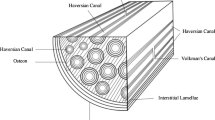
Bone toughness emerges from the interaction of several multiscale toughening mechanisms. Recently, the formation of nanoscale dilatational bands and hence the accumulation of submicron diffuse damage were suggested as an important energy dissipation processes in bone. However, a detailed mechanistic understanding of the effect of this submicron toughening mechanism across multiple scales is lacking. Here, we propose a new three-dimensional ultrastructure volume element model showing the formation of nanoscale dilatational bands based on stress-induced non-collagenous protein denaturation and quantify the total energy released through this mechanism in the vicinity of a propagating crack. Under tensile deformation, large hydrostatic stress develops at the nanoscale as a result of local confinement. This tensile hydrostatic stress supports the denaturation of non-collagenous proteins at organic–inorganic interfaces, which leads to energy dissipation. Our model provides new fundamental understanding of the mechanism of dilatational bands formation and its contribution to bone toughness.









Similar content being viewed by others
References
Almer JD, Stock SR (2005) Internal strains and stresses measured in cortical bone via high-energy X-ray diffraction. J Struct Biol 152:14–27
Almer JD, Stock SR (2007) Micromechanical response of mineral and collagen phases in bone. J Struct Biol 157:365–370. https://doi.org/10.1016/j.jsb.2006.09.001
Alexander B, Daulton TL, Genin GM et al (2012) The nanometre-scale physiology of bone: steric modelling and scanning transmission electron microscopy of collagen–mineral structure. J R Soc Interface 9:1774–1786. https://doi.org/10.1098/rsif.2011.0880
Bonar LC, Lees S, Mook HA (1985) Neutron diffraction studies of collagen in fully mineralized bone. J Mol Biol 181:265–270
Boskey AL, Gadaleta S, Gundberg C et al (1998) Fourier transform infrared microspectroscopic analysis of bones of osteocalcin-deficient mice provides insight into the function of osteocalcin. Bone 23:187–196. https://doi.org/10.1016/S8756-3282(98)00092-1
Burger C, Zhou H-W, Wang H et al (2008) Lateral packing of mineral crystals in bone collagen fibrils. Biophys J 95:1985–1992. https://doi.org/10.1529/biophysj.107.128355
Burr DB, Milgrom C, Fyhrie D et al (1996) In vivo measurement of human tibial strains during vigorous activity. Bone 18:405–410. https://doi.org/10.1016/8756-3282(96)00028-2
Burr DB, Turner CH, Naick P et al (1998) Does microdamage accumulation affect the mechanical properties of bone? J Biomech 31:337–345. https://doi.org/10.1016/S0021-9290(98)00016-5
Ciuchi IV, Olariu CS, Mitoseriu L (2013) Determination of bone mineral volume fraction using impedance analysis and Bruggeman model. Mater Sci Eng B 178:1296–1302. https://doi.org/10.1016/j.mseb.2013.04.001
Cowin SC, Weinbaum S (1998) Strain amplification in the bone mechanosensory system. Am J Med Sci 316:184–188. https://doi.org/10.1016/S0002-9629(15)40399-4
De Buffrénil V, Dabin W, Zylberberg L (2004) Histology and growth of the cetacean petro-tympanic bone complex. J Zool 262:371–381. https://doi.org/10.1017/S0952836903004758
Deshpande AS, Beniash E (2008) Bioinspired synthesis of mineralized collagen fibrils. Cryst Growth Des 8:3084–3090. https://doi.org/10.1021/cg800252f
Ellman R, Spatz J, Cloutier A et al (2013) Partial reductions in mechanical loading yield proportional changes in bone density, bone architecture, and muscle mass. J Bone Miner Res 28:875–885. https://doi.org/10.1002/jbmr.1814
Fantner GE, Hassenkam T, Kindt JH et al (2005) Sacrificial bonds and hidden length dissipate energy as mineralized fibrils separate during bone fracture. Nat Mater 4:nmat1428–616. https://doi.org/10.1038/nmat1428
Fantner GE, Adams J, Turner P et al (2007) Nanoscale ion mediated networks in bone: osteopontin can repeatedly dissipate large amounts of energy. Nano Lett 7:2491–2498. https://doi.org/10.1021/nl0712769
Fratzl P, Paris O, Klaushofer K, LW J (1996) Bone mineralization in an osteogenesis imperfecta mouse model studied by small-angle X-ray scattering. J Clin Invest 97(2):396–402. https://doi.org/10.1172/JCI118428
Grynpas MD, Bonar LC, Glimcher MJ (1984) X-ray diffraction radial distribution function studies on bone mineral and synthetic calcium phosphates. J Mater Sci 19:723–736. https://doi.org/10.1007/BF00540442
Gupta HS, Messmer P, Roschger P et al (2004) Synchrotron diffraction study of deformation mechanisms in mineralized tendon. Phys Rev Lett 93:158101. https://doi.org/10.1103/PhysRevLett.93.158101
Gupta HS, Fratzl P, Kerschnitzki M et al (2006) Evidence for an elementary process in bone plasticity with an activation enthalpy of 1 ev. J R Soc Interface 4:277–282. https://doi.org/10.1098/rsif.2006.0172
Hall BK (2015) Chapter 24—Osteoblast and osteocyte diversity and osteogenesis in vitro. Bones and cartilage, 2nd edn. Academic, San Diego, pp 401–413
Hamed E, Lee Y, Jasiuk I (2010) Multiscale modeling of elastic properties of cortical bone. Acta Mech 213:131–154. https://doi.org/10.1007/s00707-010-0326-5
Hamed E, Jasiuk I, Yoo A et al (2012) Multi-scale modelling of elastic moduli of trabecular bone. J R Soc Interface 9:1654–1673. https://doi.org/10.1098/rsif.2011.0814
Hang F, Barber AH (2011) Nano-mechanical properties of individual mineralized collagen fibrils from bone tissue. J R Soc Interface 8:500–505. https://doi.org/10.1098/rsif.2010.0413
Hang F, Gupta HS, Barber AH (2014) Nanointerfacial strength between non-collagenous protein and collagen fibrils in antler bone. J R Soc Interface 11:20130993
Hauschka PV, Wians FH (1989) Osteocalcin-hydroxyapatite interaction in the extracellular organic matrix of bone. Anat Rec 224:180–188. https://doi.org/10.1002/ar.1092240208
Hodge AJ, Petruska JA (1962) Some recent results on the electron microscopy of tropocollagen structures. In: Breese SS Jr (ed) Proc. of the fifth Internat. Congr. for Electron Microscopy, vol 2. Academic, New York, p QQ-1
Jäger I, Fratzl P (2000) Mineralized collagen fibrils: a mechanical model with a staggered arrangement of mineral particles. Biophys J 79:1737–1746. https://doi.org/10.1016/S0006-3495(00)76426-5
Jaschouz D, Paris O, Roschger P et al (2003) Pole figure analysis of mineral nanoparticle orientation in individual trabecular of human vertebral bone. J Appl Cryst 36:494–498. https://doi.org/10.1107/S0021889803000566
Ji B (2008) A study of the interface strength between protein and mineral in biological materials. J Biomech 41:259–266. https://doi.org/10.1016/j.jbiomech.2007.09.022
Jin H, Lewis JL (2004) Determination of Poisson’s ratio of articular cartilage by indentation using different-sized indenters. J Biomech Eng 126:138–145. https://doi.org/10.1115/1.1688772
Karunaratne A, Esapa CR, Hiller J et al (2012) Significant deterioration in nanomechanical quality occurs through incomplete extrafibrillar mineralization in rachitic bone: Evidence from in-situ synchrotron X-ray scattering and backscattered electron imaging. J Bone Miner Res 27:876–890. https://doi.org/10.1002/jbmr.1495
Kiviranta P, Rieppo J, Korhonen RK et al (2006) Collagen network primarily controls Poisson’s ratio of bovine articular cartilage in compression. J Orthop Res 24:690–699. https://doi.org/10.1002/jor.20107
Koch CF, Johnson S, Kumar D et al (2007) Pulsed laser deposition of hydroxyapatite thin films. Mater Sci Eng C 27:484–494. https://doi.org/10.1016/j.msec.2006.05.025
Landis WJ, Hodgens KJ, Arena J et al (1996) Structural relations between collagen and mineral in bone as determined by high voltage electron microscopic tomography. Microsc Res Tech 33:192–202
Lees S, Prostak K (1988) The locus of mineral crystallites in bone. Connect Tissue Res 18:41–54. https://doi.org/10.3109/03008208809019071
Li X, Shephard MS, Beall MW (2005) 3D anisotropic mesh adaptation by mesh modification. Unstructured Mesh Generation. Comput Methods Appl Mech Eng 194:4915–4950. https://doi.org/10.1016/j.cma.2004.11.019
McNally EA, Schwarcz HP, Botton GA, Arsenault AL (2012) A model for the ultrastructure of bone based on electron microscopy of ion-milled sections. PLoS ONE 7(1):e29258. https://doi.org/10.1371/journal.pone.0029258
Meyers MA, Chen P-Y, Lin AY-M, Seki Y (2008) Biological materials: Structure and mechanical properties. Prog Mater Sci 53:1–206. https://doi.org/10.1016/j.pmatsci.2007.05.002
Mullins LP, Bruzzi MS, mchugh PE (2007) Measurement of the microstructural fracture toughness of cortical bone using indentation fracture. J Biomech 40:3285–3288. https://doi.org/10.1016/j.jbiomech.2007.04.020
Nair AK, Gautieri A, Chang SW, Buehler MJ (2013) Molecular mechanics of mineralized collagen fibrils in bone. Nat Commun 4:1724
Nair AK, Gautieri A, Buehler MJ (2014) Role of intrafibrillar collagen mineralization in defining the compressive properties of nascent bone. Biomacromolecules 15:2494–2500. https://doi.org/10.1021/bm5003416
Nalla RK, Kinney JH, Ritchie RO (2003) Mechanistic fracture criteria for the failure of human cortical bone. Nat Mater 2:164–168. https://doi.org/10.1038/nmat832
Neil Dong X, Edward Guo X (2004) The dependence of transversely isotropic elasticity of human femoral cortical bone on porosity. J Biomech 37:1281–1287. https://doi.org/10.1016/j.jbiomech.2003.12.011
Nikel O, Laurencin D, mccallum SA et al (2013) NMR investigation of the role of osteocalcin and osteopontin at the organic–inorganic interface in bone. Langmuir 29:13873–13882. https://doi.org/10.1021/la403203w
Peroos S, Du Z, de Leeuw NH (2006) A computer modelling study of the uptake, structure and distribution of carbonate defects in hydroxy-apatite. Biomaterials 27:2150–2161. https://doi.org/10.1016/j.biomaterials.2005.09.025
Pidaparti RM, Chandran A, Takano Y, Turner CH (1996) Bone mineral lies mainly outside collagen fibrils: predictions of a composite model for osternal bone. J Biomech 29:909–916
Poundarik AA, Gundberg CM, Vashishth D (2011) Non-collageneous proteins influence bone crystal size and morphology: a SAXS study. In: 2011 IEEE 37th annual northeast bioengineering conference (NEBEC), Troy, pp 1–2. https://doi.org/10.1109/nebc.2011.5778671
Poundarik AA, Diab T, Sroga GE et al (2012) Dilatational band formation in bone. Proc Natl Acad Sci. https://doi.org/10.1073/pnas.1201513109/-/dcsupplemental
Poundarik AA, Wu P-C, Evis Z et al (2015) A direct role of collagen glycation in bone fracture. J Mech Behav Biomed Mater 52:120–130. https://doi.org/10.1016/j.jmbbm.2015.08.012
Rath Bonivtch A, Bonewald LF, Nicolella DP (2007) Tissue strain amplification at the osteocyte lacuna: a microstructural finite element analysis. J Biomech 40:2199–2206. https://doi.org/10.1016/j.jbiomech.2006.10.040
Reznikov N, Shahar R, Weiner S (2014) Bone hierarchical structure in three dimensions. Acta Biomater 10:3815–3826. https://doi.org/10.1016/j.actbio.2014.05.024
Rho J-Y, Kuhn-Spearing L, Zioupos P (1998) Mechanical properties and the hierarchical structure of bone. Med Eng Phys 20:92–102. https://doi.org/10.1016/S1350-4533(98)00007-1
Ritter NM, Farach-Carson MC, Butler WT (1992) Evidence for the formation of a complex between osteopontin and osteocalcin. J Bone Miner Res 7:877–885. https://doi.org/10.1002/jbmr.5650070804
Rubin MA, Jasiuk I, Taylor J et al (2003) TEM analysis of the nanostructure of normal and osteoporotic human trabecular bone. Bone 33:270–282. https://doi.org/10.1016/S8756-3282(03)00194-7
Schaffler MB, Radin EL, Burr DB (1989) Mechanical and morphological effects of strain rate on fatigue of compact bone. Bone 10:207–214. https://doi.org/10.1016/8756-3282(89)90055-0
Schaffler MB, Choi K, Milgrom C (1995) Aging and matrix microdamage accumulation in human compact bone. Bone 17:521–525. https://doi.org/10.1016/8756-3282(95)00370-3
Schwarcz HP, mcnally EA, Botton GA (2014) Dark-field transmission electron microscopy of cortical bone reveals details of extrafibrillar crystals. J Struct Biol 188:240–248. https://doi.org/10.1016/j.jsb.2014.10.005
Seref-Ferlengez Z, Basta-Pljakic J (2014) Structural and mechanical repair of diffuse damage in cortical bone in vivo. J Bone Miner Res 29:2537–2544. https://doi.org/10.1002/jbmr.2309
Sih GC, Paris PC, Irwin GR (1965) On cracks in rectilinearly anisotropic bodies. Int J FractMech 1:189–203. https://doi.org/10.1007/BF00186854
Siegmund T, Allen MR, Burr DB (2008) Failure of mineralized collagen fibrils: modeling the role of collagen cross-linking. J Biomech 41:1427–1435. https://doi.org/10.1016/j.jbiomech.2008.02.017
Sroga GE, Vashishth D (2012) Effects of bone matrix proteins on fracture and fragility in osteoporosis. Curr Osteoporos Rep 10:141–150. https://doi.org/10.1007/s11914-012-0103-6
Stock SR (2015) The mineral–collagen interface in bone. Calcif Tissue Int 97:1–19. https://doi.org/10.1007/s00223-015-9984-6
Stroh AN (1962) Steady state problems in anisotropic elasticity. J Math Phys 41:77–103. https://doi.org/10.1002/sapm196241177
Su X, Sun K, Cui FZ, Landis WJ (2003) Organization of apatite crystals in human woven bone. Bone 32:150–162. https://doi.org/10.1016/S8756-3282(02)00945-6
Taylor D, Hazenberg JG, Lee TC (2007) Living with cracks: damage and repair in human bone. Nat Mater 6:263–268. https://doi.org/10.1038/nmat1866
Termine JD, Kleinman HK, Whitson SW et al (1981) Osteonectin, a bone-specific protein linking mineral to collagen. Cell 26:99–105. https://doi.org/10.1016/0092-8674(81)90037-4
Tresoldi I, Oliva F, Benvenuto M et al (2013) Tendon’s ultrastructure. Muscles Ligaments Tendons J 3(1):2–6. https://doi.org/10.11138/mltj/2013.3.1.002
Vashishth D (2004) Rising crack-growth-resistance behavior in cortical bone: implications for toughness measurements. J Biomech 37:943–946. https://doi.org/10.1016/j.jbiomech.2003.11.003
Vashishth D, Behiri JC, Bonfield W (1997) Crack growth resistance in cortical bone: concept of microcrack toughening. J Biomech 30:763–769. https://doi.org/10.1016/S0021-9290(97)00029-8
Vashishth D, Koontz J, Qiu SJ et al (2000) In vivo diffuse damage in human vertebral trabecular bone. Bone 26:147–152. https://doi.org/10.1016/S8756-3282(99)00253-7
Weiner S, Traub W, Wagner HD (1999) Lamellar bone: structure–function relations. J Struct Biol 126:241–255. https://doi.org/10.1006/jsbi.1999.4107
Yuan F, Stock SR, Haeffner DR et al (2010) A new model to simulate the elastic properties of mineralized collagen fibril. Biomech Model Mechanobiol 10:147–160. https://doi.org/10.1007/s10237-010-0223-9
Zimmermann EA, Gludovatz B, Schaible E et al (2014) Fracture resistance of human cortical bone across multiple length-scales at physiological strain rates. Biomaterials 35:5472–5481. https://doi.org/10.1016/j.biomaterials.2014.03.066
Zioupos P, Currey JD (1998) Changes in the stiffness, strength, and toughness of human cortical bone with age. Bone 22:57–66. https://doi.org/10.1016/S8756-3282(97)00228-7
Acknowledgements
This study was financially supported by the US National Science Foundation (NSF) through Grant CMMI 1363526 and the National Institute of Health (NIH) through Grant AR49635.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, Z., Vashishth, D. & Picu, R.C. Bone toughening through stress-induced non-collagenous protein denaturation. Biomech Model Mechanobiol 17, 1093–1106 (2018). https://doi.org/10.1007/s10237-018-1016-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10237-018-1016-9