Abstract
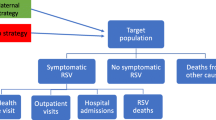
Respiratory syncytial virus (RSV) is a common cause of respiratory infection that is highly prevalent in infants, particularly those with underlying medical conditions. Severe cases of RSV require hospitalisation as well as admission to intensive care and may even result in death. The objective of the study was to measure the net benefits that could arise from an immunisation programme of infants that may well eradicate RSV to a high degree and save the direct and indirect medical care costs from hospitalisation, morbidity and the gain from potential life-time earnings by reducing the probability of mortality. In this context, the majority of existing empirical investigations are based on data from clinical trials, and where relevant facts are not available, a series of strong assumptions is derived from the published literature, whereas in this study, for the first time, the hospital episode statistics database is used to calculate the cost-benefit ratios. The methodology of the analysis adopts a cost-benefit approach to assess the impact of the immunisation and whether it is beneficial to society. The underlying assumptions of the basic model are assessed by adopting a sensitivity analysis. The results show that a number of categories are cost-effective with the use of the passive drug, which means benefits by raising the life expectancy and quality as well as reducing the resource burden on society.


Similar content being viewed by others
References
Bentley, A., Filipovic, I., Gooch, K., Busch, K.: A cost-effectiveness analysis of respiratory syncytial virus (RSV) prophylaxis in infants in the United Kingdom. Health Econ Rev 3, 1–12 (2013)
Committee on infectious diseases: Report of the committee on infectious diseases. Red book, 26th edn, pp. 523–528. American Academy of Pediatrics, Elk Grove (2003)
Food and drug administration. Palivizumab product approval information, clinical review [Online]. Available at http://www.fda.gov/downloads/Drugs/DevelopmentApprovalProcess/HowDrugsareDevelopedandApproved/ApprovalApplications/TherapeuticBiologicApplications/ucm113384.pdf (1998). Accessed 3 Oct 2013
Simpson, S., Burls, A.: A systematic review of the effectiveness and cost-effectiveness of palivizumab (Synagis®) in the prevention of respiratory syncytial virus (RSV) infection in infants at high risk of infection. University of Birmingham, Department of Public Health and Epidemiology (2001)
Department of health. Immunisation against infectious disease. In: green book. s.l.: the stationery office (2006)
Wang, D., et al.: Immunoprophylaxis against respiratory syncytial virus (RSV) with palivizumab in children: a systematic review and economic evaluation. Health Technol. Assess. 12, 1–86 (2008)
Wang, D., Bayliss, S., Meads, C.: Palivizumab for immunoprophylaxis of respiratory syncytial virus (RSV) bronchiolitis in high-risk infants and young children: a systematic review and additional economic modelling of subgroup analyses. Health Technol. Assess. 15, 1–124 (2011)
Nuijten, J.M., Wittenberg, W., Lebmeier, M.: Cost effectiveness of Palivizumab for respiratory syncytial virus Prophylaxis in high risk children: a UK analysis. Pharmaeconomics 25, 55–71 (2007)
Hussey, D.G., Laser, L.M., Reekie, D.W.: The costs and benefits of a vaccination programme for Haemophilus influenzae type B disease. S. Afr. Med. J. 85, 20–25 (1995)
Hospital episode statistics. HES online: understanding the data [Online]. Available at: http://www.hesonline.nhs.uk/Ease/servlet/ContentServer?siteID=1937&categoryID=289 (2012). Accessed 24 Aug 2012
Faddy, M.J., McClean, S.I.: Analysing data on lengths of stay of hospital patients using phase type distributions. Appl. Stochastic Models Bus. Ind. 15, 311–317 (1999)
Department of health. NHS reference costs: financial year 2011–12 [Online]. Available at: https://www.gov.uk/government/publications/nhs-reference-costs-financial-year-2011-to-2012 (2012). Accessed 2 July 2013
Patokos, T.: Lives in the hands of economists: a critical review of the main methodologies used to derive the value of a statistical life. Environ. Econ. 1, 11–20 (2010)
Philips, Z., Bojke, L., Sculpher, M., Claxton, K., Golder, S.: Good practice guidelines for decision-analytic modelling in health technology assessment. Pharmacoeconomics 24(4), 355–371 (2006)
IMPACT study group: Palivizumab, a humanized respiratory syncytial virus monoclonal antibody, reduces hospitalization from respiratory syncytial virus infection in high-risk infants. Pediatrics 102, 531–537 (1998)
NICE DSU decision support unit—NICE methods guide updates [Online]. Available at: https://www.nicedsu.org.uk/NICE-Methods-Guide-updates (2013). Accessed 2 July 2013
PSSRU. Unit costs of health and social care. University of Kent (2012)
Acknowledgments
We wish to acknowledge the grant of £10,000 from the University of Hertfordshire to carry out the study and purchase the HES data set. Also, the writers would like to thank the reviewers for their constructive comments on the content of the paper.
Author information
Authors and Affiliations
Corresponding author
Appendix
Rights and permissions
About this article
Cite this article
Thomas, G. A cost-benefit analysis of the immunisation of children against respiratory syncytial virus (RSV) using the English Hospital Episode Statistics (HES) data set. Eur J Health Econ 19, 177–187 (2018). https://doi.org/10.1007/s10198-014-0662-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10198-014-0662-9