Abstract
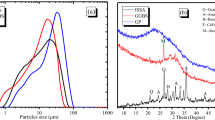
Heavy metals are naturally occurring trace elements present in the environment, including soil, water, and air. However, high heavy metal concentration contamination from waste is a serious environmental problem brought on by industrial activities. The research in this study evaluated different biomineralized CaCO3 forms from individual bacteria species in heavy-metal-contaminated soils and mine tailings. Scanning electron microscope (SEM) images of the crystals were used to characterize the precipitated CaCO3. Generally, urea-hydrolysis bacteria form minerals via a microbiologically induced calcite precipitation (MICP) process. These bacteria produce the urease enzyme, which leads to urea-hydrolysis. These bacteria were isolated from heavy-metal-contaminated soils and characterized for their potential utilization in the S/S process. Optimum conditions for indigenous bacterial growth were 30 °C and a pH range of 7–8; and growth patterns were further affected by the growth medium salinity. SEM and X-ray diffraction (XRD) analyses demonstrated that bioaccumulated heavy metal ions were deposited around the cell envelope as rhombohedral and sphere shaped crystalline carbonate minerals in optimum conditions. In this study, the authors hypothesize that the indigenous bacteria can effectively precipitate heavy metals in soil and mine tailing with the urea-hydrolysis enzyme, and play an important role in heavy metal stabilization.









Similar content being viewed by others
References
Zhuang P, Zou B, Li NY, Li ZA (2009) Heavy metal contamination in soils and food crops around Dabaoshan mine in Guangdong, China: implication for human health. Environ Geochem Health 31:707–715
Olayinka KO, Oyeyiola AO, Odujebe FO, Oboh B (2011) Uptake of potentially toxic metals by vegetable plants grown on contaminated soil and their potential bioavailability using sequential extraction. J Soil Sci Environ 2(8):220–227
Pruvot C, Douay F, Herve F, Waterlot C (2006) Heavy metals in soil, crops and grass as a source of human exposure in the former mining areas. J Soils Sediments 6:215–220
Chen QY, Tayrer M, Hills CD, Yang XM, Carey P (2009) Immobilisation of heavy metal in cement-based solidification/stabilisation: a review. Waste Manag 29:390–403
Jang A, Kim IS (2000) Solidification and stabilization of Pb, Zn, Cd and Cu intailing wastes using cemnet and fly ash. Miner Eng 13(14–15):1659–1662
Alpaslan B, Yukselen MA (2000) Remediation of lead contaminated soils by stabilization/solidification. Water Air Soil Pollut 133:253–263
Cheung KH, Lai HY, Gu J-D (2006) Membrane-associated hexavalent chromium reductase of Bacillus megaterium TKW 3 with induced expression. J Microbiol Biotechnol 16:855–862
DeJong JT, Fritzges MB, Nusslein K (2006) Microbially induced cementation to control sand response to undrained shear. J Geotech Geoenviron Eng 132:1381–1392
Cheung KH, Gu JD (2007) Mechanisms of hexavalent chromium detoxification by bacteria and bioremediation applications. Int Biodeterior Biodegrad 59:8–15
Sarda D, Choonia HS, Sarode DD, Lele SS (2009) Biocalcification by Bacillus pasteurii urease: a novel application. J Ind Microbiol Biotechnol 36:1111–1115
Ramachandran SK, Ramakrishnan V, Bang SS (2001) Remediation of concrete using microorganisms. ACI Mater J 98:3–9
Ivanov V, Chu J (2008) Applications of microorganisms to geotechnical engineering for bioclogging and biocementation of soil in situ. Rev Environ Sci Biotechnol 7:139–153
Mary SR, Williams, Seraphin S (1998) Heavy metal biomineralization in free-living nematodes, Panagrolaimus spp. Mater Sci Eng 6:47–51
Bang SS, Galinat JK, Ramakrishnan V (2001) Calcite precipitation induced by polyurethane-immobilized Bacillus pasteurii. Enzyme Microb Technol 28:404–409
Stocks-Fischer S, Galinat JK, Bang SS (1999) Microbiological precipitation of CaCO3. Soil Biol Biochem 31:1563–1571
Gollapudi UK, Knutson CL, Bang SS, Islam MR (1995) A new method for controlling leaching through permeable channels. Chemosphere 30:695–705
Dick J, De Windt W, De Graef B, Saveyn H, Van der Meeren P, De Belie N, Verstraete W (2006) Bio-deposition of a calcium carbonate layer on degraded limestone by Bacillus species. Biodegradation 17:357–367
Lee JR (2011) A study of stabilization methods using minerals for arsenic contaminated soil near Samkwang mine area at Cheongyang-gun, Chungcheongnam-do. Ph.D. Dissertation, Graduated School Pusan National University
Ahn JS, Kim JY, Chon CM, Moon HS (2003) Mineralogical and chemical characterization of arsenic solid phases in weathered mine tailings and their leaching potential. Econ Environ Geol 36(1):27–38
Hwang BS (2005) Assessment of arsenic and heavy metals leaching behavior in contaminated soils of abandoned metalliferous mines. M.S. Dissertation, Semyung University Graduated School
Weaver RW, Angle TS, Bottomley PS (1994) Methods of soil analysis, Part 2. Microbiological and biochemical properties. Soil Science Society of America, USA
Nindy Tupple Miller (1982) Single derivatization method for routine analysis of bacterial whole-cell fatty acid methyl esters, including hydroxy acids. J Clin Microbiol 16(3):584–586
Lee JY, Koo JK, Kim KS, Dong JI, Park YH, Bae WK, Yang JW, Yeom IT, Yoon SP, Lee JS, Jang YY, Chung JC, Choi SI, Hwang KY, Hwang JS (2001) Soil environmental engineering. Hyangmunsa, Korea
Phae CG, Oh JM (2002) Soil contamination measurement analysis. Sinkwang-munhwasa, Korea
Lee S, Kim TH, Lee JH (2007) Soil test method. Goomibook, Korea
Liu D, Yates MZ (2006) Formation of rod-shaped calcite crystals by microemulsion-based synthesis. Langmuir 22(13):5566–5569
Dhami NK, Reddy MS, Mukherjee A (2014) Synergistic role of bacterial urease and carbonic anhydrase in carbonate mineralization. Appl Biochem Biotechnol 172:2552–2561
Siddique R, Achal V, Reddy M, Mukherjee A (2008) Improvement in the compressive strength of cement mortar by the use of a microorganism—Bacillus megaterium. In: Limbachiya MC, Kew H (eds) Excellence in concrete construction through innovation. Taylor and Francis, London, pp 27–30
Soon NW, Lee LM, Khun TC, Ling HS (2013) Improvements in engineering properties of soils through microbial-induced calcite precipitation. KSCE J Civil Eng 17:718–728
Achal V, Pan X, Zhang D, Fu Q (2012) Bioremediation of Pb-contaminated soil based on microbially induced calcite precipitation. J Microbiol Biotechnol 22(2):244–347
Kang CH, Han SH, Shin YJ, Oh SJ, So JS (2014) Bioremediation of Cd by microbially induced calcite precipitation. Appl Biochem Biotechnol 172:1929–1937
De Yoreo JJ, Vekilov PG (2003) Principles of crystal nucleation and growth. Rev Mineral Geochem 54:57–93
Favre N, Christ ML, Pierre AC (2009) Biocatalytic capture of CO2 with carbonic anhydrase and its transformation to solid carbonate. J Mol Catal B Enzym 60:163–170
Gorospe CM, Han SH, Kim SG, Park JY, Kang CH, Jeong JH, So JS (2013) Effects of different calcium salts on calcium carbonate crystal formation by Sporosarcina pasteurii KCTC 3558. Biotechnol Bioprocess Eng 18:903–908
Tai CY, Chen FB (1998) Polymorphism of CaCO3 precipitated in a constant-composition environment. Am Inst Chem Eng 44:1790–1798
Bäuerlein E (2007) Hand book of biomineralization. Wiley, Weinheim
Bazylinski DA, Frankel RB, Konhauser KO (2007) Modes of biomineralization of magnetite by microbes. Geomicrobiology 24:465–475
Abo-El-Enein SA, Ali AH, Talkhan FN, Abdel-Gawwad HA (2012) Utilization of microbial induced calcite precipitation for sand consolidation and mortar crack remediation. HBRC J 8:185–192
Hammes F, Boon N, de Villiers J, Verstraete W, Siciliano SD (2003) Strain-specific ureolytic microbial calcium carbonate precipitation. Appl Environ Microbiol 69:4901–4909
Park SJ, Park YM, Chun WY, Kim WJ, Ghim SY (2010) Calcite-forming bacteria for compressive strength improvement in mortar. J Microbiol Biotechnol 20:782–788
Kawaguchi T, Decho AW (2002) A laboratory investigation of cyanobacterial extracellular polymeric secretion (EPS) in influencing CaCO3 polymorphism. J Cryst Growth 240:230–235
Dhami NK, Reddy MS, Mukherjee A (2013) Biomineralization of calcium carbonate polymorphs by the bacterial strains isolated from calcareous sites. J Microbiol Biotechnol 23:707–714
Ferrer MR, Quevedo-Sarmiento J, Bejar V, Delgado R, Ramos-Cormenzana A, Rivadeneyra MA (1988) Calcium carbonate formation by Deleya halophila: effect on salt concentration and incubation temperature. Geomicrobiol J 6:49–57
Hammes F, Verstraete W (2002) Key roles of pH and calcium metabolism in microbial carbonate precipitation. Rev Environ Sci Biotechnol 1:3–7
Mitchell AC, Ferris FG (2005) The coprecipitation of Sr into calcite precipitates induced by bacterial ureolysis in artificial groundwater: temperature and kinetics dependence. Geochim Gosmochim Acta 69:4199–4210
Okwadha GDO, Li J (2010) Optimum conditions for microbial carbonate precipitation. Chemosphere 81:1143–1148
Loewenthal RE, Marais GVR (1978) Carbonate chemistry of aquatic systems: theory and application, vol 1. Ann Arbor Science, Ann Arbor
Anne S, Rozen bamuO, Andreazza P, Rouet JL (2010) Evidence of a bacterial carbonate coating on plaster samples subjected to the calcite bioconcept biomineralization techniques. Constr Build Mater 24:1036–1042
Ferris FG, Phoenix V, Fujita Y, Smith RW (2003) Kinetics of calcite precipitation induced by ureolytic bacteria at 10 to 20 °C in artificial groundwater. Geochim Cosmochim Acta 67:1701–1710
Dupraz S, Menez B, Gouze P, Leprovost R, Benezeth P, Pokrovsky OS, Guyot F (2009) Experimental approach of CO2 biomineralization in deep saline aquifers. Chem Geol 265:54–62
Mobley HLT, Island MD, Hausinger RP (1995) Molecular biology of microbial ureases. Microbiol Rev 59:451–580
Braissant O, Caillaume G, Dupraz C, Verrecchia EP (2003) Bacterially induced mineralization of calcium carbonate in terrestrial environments: the role of exopolysaccharides and amino acids. J Sediment Res 73(3):485–490
Acknowledgements
This study was supported by the Korea Ministry of Environment (MOE) as part of the GAIA Project (Geo-Advanced Innovative Action Project; No. 2015000550007).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, JH., Lee, JY. An optimum condition of MICP indigenous bacteria with contaminated wastes of heavy metal. J Mater Cycles Waste Manag 21, 239–247 (2019). https://doi.org/10.1007/s10163-018-0779-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10163-018-0779-5