Abstract
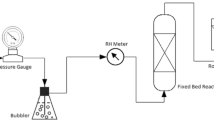
A continuous system (feeding rate >1 kg/h) consisting of thermal dechlorination pre-treatment and catalytic pyrolysis with Fe-restructured clay (Fe-RC) catalyst was developed for feedstock recycling of PVC-containing mixed plastic waste. The vented screw conveyor which was specially designed for continuous dechlorination was able to achieve dechlorination efficiency of over 90 % with a feedstock retention time longer than 35.5 min. The chlorine content of the pyrolytic oil obtained after dechlorination was in the range of 6.08–39.50 ppm, which meet the specification for reclamation pyrolytic oil in Japan. Fe-RC was found to significantly improve the yield of pyrolytic oil (achieved to 83.73 wt%) at the optimized pyrolysis temperature of 450 °C and catalyst dosage of 60 g. With the optimized parameters, Fe-RC showed high selectivity for the C9–C12 and C13–C19 oil fraction, which are the major constituents of kerosene and diesel fuel, demonstrating that this catalyst can be applied in the pyrolysis of mixed plastic wastes for the production of kerosene and diesel fuel. Overall, the continuous process exhibited high stability and consistently high-oil yield upon reaching steady state, indicating its potential up-scaling application in the industry.







Similar content being viewed by others
References
Plastics Europe (2009) The compelling facts about plastics: an analysis of European plastics production, demand and recovery for 2008. Plastics Europe, Brussels
Plastics Europe (2015) Plastics—the Facts 2015: an analysis of European plastics production, demand and waste data. Plastics Europe, Brussels, Belgium. see http://issuu.com/plasticseuropeebook/docs/finalplasticsthefacts2015ebookwebve/1?e=14847134/31230241
NEA (2015) Waste statistics and overall recycling: waste statistics and recycling rate for 2014. National Environmental Agency (NEA), Singapore. see http://www.nea.gov.sg/energy-waste/waste-management/waste-statistics-and-overall-recycling
Anhui Oursun Resource Technology Co., Ltd. see http://www.oursunworld.com/gylc.html. Retrieved 07 April 2016
Plastic2Oil, Inc. see http://www.plastic2oil.com/site/p2o-technology. Retrieved 07 April 2016
Moriya S, Watanabe H, Yaginuma R, Matsumoto T, Nakajima M, Tsukada M, Isshiki N (2000) Studied of recycled fuel oil for diesel engine extracted from waste plastics disposals. Energy Conversion Engineering Conference and Exhibit, 35th Intersociety, vol 1, pp 510–515
Zmuda H (2004) US Patent No. 6,777581. Washington, DC: U.S. Patent and Trademark Office
Mendyka B, Musialik-Piotrowska A, Syczewska K (1992) Effect of chlorine compounds on the deactivation of platinum catalysts. Catal Today 11:597–610
Spivey JJ, Butt JB (1992) Literature review: deactivation of catalysts in the oxidation of volatile organic compounds. Catal Today 11:465–500
Imamura S (1992) Catalytic decomposition of halogenated organic compounds and deactivation of the catalysts. Catal Today 11:547–567
Saito K, Narita H (2001) Studies on the dechlorination and oil-production technology of waste plastics. J Mater Cycles Waste Manag 3:93–98
Fukushima M, Wu B, Ibe H, Wakai K, Sugiyama E, Abe H, Kitagawa K, Tsuruga S, Shimura K, Ono E (2010) Study on dechlorination technology for municipal waste plastics containing polyvinyl chloride and polyethylene terephthalate. J Mater Cycles Waste Manag 12:108–122
Yuan G, Chen D, Yin L, Wang Z, Zhao L, Wang JY (2014) High efficiency chlorine removal from polyvinyl chloride (PVC) pyrolysis with a gas–liquid fluidized bed reactor. Waste Manage 34:1045–1050
Wang JY, Li K, Yang Y, Wee LS, Weerachanchai P, Guoan Y, Junxi L (2015) application of modified pillared clay with transition metals in catalytic pyrolysis (technical disclosure), accorded Singapore provisional application number 10201503425X
Bhaskar T, Uddin MA, Murai K, Kaneko J, Hamano K, Kusaba T, Muto A, Sakata Y (2003) Comparison of thermal degradation products from real municipal waste plastic and model mixed plastics. J Anal Appl Pyrolysis 70:579–587
Ma S, Lu J, Gao J (2002) Study of the low temperature pyrolysis of PVC. Energy Fuels 16:338–342
Japan Industrial Standard (2004) Pyrolytic oil from waste plastics—boiler fuel and diesel generator fuel. JIS TS Z 0025:2004
Manos G, Yusof IY, Papayannakos N, Gangas NH (2001) Catalytic cracking of polyethylene over clay catalysts. Comparison with an ultrastable Y zeolite. Ind Eng Chem Res 40:2220–2225
Serrano DP, Aguado J, Escola JM (2012) Developing advanced catalysts for the conversion of polyolefinic waste plastics into fuels and chemicals. ACS Catal 2:1924–1941
Gobin K, Manos G (2004) Thermogravimetric study of polymer catalytic degradation over microporous materials. Polym Degrad Stab 86:225–231
De Stefanis A, Cafarelli P, Gallese F, Borsella E, Nana A, Perez G (2013) Catalytic pyrolysis of polyethylene: a comparison between pillared and restructured clays. J Anal Appl Pyrolysis 104:479–484
Ahmad I, Khan MI, Khan H, Ishaq M, Tariq R, Gul K, Ahmad W (2015) Influence of metal-oxide-supported bentonites on the pyrolysis behavior of polypropylene and high-density polyethylene. J Appl Polym Sci 132:41221
De León MA, De Los Santos C, Latrónica L, Cesio AM, Volzone C, Castiglioni J, Sergio M (2014) High catalytic activity at low temperature in oxidative dehydrogenation of propane with Cr–Al pillared clay. Chem Eng J 241:336–343
López A, de Marco I, Caballero BM, Laresgoiti MF, Adrados A (2011) Influence of time and temperature on pyrolysis of plastic wastes in a semi-batch reactor. Chem Eng J 173:62–71
Singh RK, Ruj B (2016) Time and temperature depended fuel gas generation from pyrolysis of real world municipal plastic waste. Fuel 174:164–171
Acknowledgments
This research is funded by the National Environment Agency of Singapore under the Environment Technology Research Program (ETRP Grant No.: 1202-109).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Lei, J., Yuan, G., Weerachanchai, P. et al. Investigation on thermal dechlorination and catalytic pyrolysis in a continuous process for liquid fuel recovery from mixed plastic wastes. J Mater Cycles Waste Manag 20, 137–146 (2018). https://doi.org/10.1007/s10163-016-0555-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10163-016-0555-3