Abstract
Background
Immune checkpoint inhibitors (ICPis) are associated with multi-organ immune-related adverse effects. Here, we examined the incidence rate, recovery rate, and risk factors of acute kidney injury complicated with ICPis (ICPi-AKI) and evaluted the association between ICPi-AKI and mortality in Japanese patients.
Methods
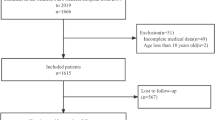
We analyzed 152 consecutive patients receiving ICPis between 2015 and 2019. A logistic regression analysis was performed to identify risk factors for ICPi-AKI incidence and Cox regression analysis was performed to evaluate the association between ICPi-AKI and mortality.
Results
The mean patient age was 67 ± 10 years, with the median baseline serum creatinine level of 0.78 mg/dL. Twenty-seven patients (18%) developed ICPi-AKI, and 19 (73%) of them recovered. Pembrolizumab use and liver diseases were significant risk factors for the ICPi-AKI incidence. During the follow-up, 85 patients (59%) died, 17 patients (63%) with ICPi-AKI and 68 (54%) patients without ICPi-AKI, respectively. The ICPi-AKI incidence was not independently associated with mortality (adjusted hazard ratio, 0.85; 95% confidence intervals, 0.46–1.61).
Conclusions
Our finding suggest that pembrolizumab use and liver diseases are associated with a higher risk of ICPi-AKI development, but ICPi-AKI did not affect mortality. Future multi-center studies are needed to develop optimal management and prevention strategies for this complication in patients receiving ICPis.


Similar content being viewed by others
References
Sury K, Parezella MA, Shirali AC. Cardiorenal complications of immune checkpoint inhibitors. Nat Rev Nephrol. 2018;14:571–88.
Cortazar FB, Marrone KA, Troxell ML, Ralto KM, Hoenig MP, Brahmer JR, et al. Clinicopathological features of acute kidney injury associated with immune checkpoint inhibitors. Kidney Int. 2016;90:638–47.
Shirali AC, Perazella MA, Gettinger S. Association of acute interstitial nephritis with programmed cell death 1 inhibitor therapy in lung cancer patients. Am J Kidney Dis. 2016;68:287–91.
Mamlouk O, Selament U, Machado S, Abdelrahim M, Glass WF, Tchakarov A, et al. Nephrotoxicity of immune checkpoint inhibitors beyond tubulointerstitial nephritis: Single-center experience. J Immunother Cancer. 2019;7:2.
Seethapathy H, Zhao S, Chute DF, Zubiri L, Oppong Y, Strohbehn I, et al. The incidence, causes, and risk factors of acute kidney injury in patients receiving immune checkpoint inhibitors. Clin J Am Soc Nephrol. 2019;14:1692–700.
Cortarzar FB, Kibbelaar ZA, Glezerman IG, Budayyeh A, Mamlouk O, Motwani SS, et al. Clinical features and outcomes of immune checkpoint inhibitor-associated AKI: A multicenter study. J Am Soc Nephrol. 2020;31:435–46.
Sawhney S, Marks A, Fluck N, Levin A, Prescott G, Black C. Intermediate and long-term outcomes of survivors of acute kidney injury episodes: a large population-based cohort study. Am J Kidney Dis. 2017;69:18–28.
Fujii T, Uchino S, Doi K, Sato T, Kawamura T, JAKID study group. Diagnosis, management, and prognosis of patients with acute kidney injury in Japanese intensive care units: the JAKID study. J Crit Care. 2018;47:185–91.
Tabei A, Watanabe M, Ikeuchi H, Nakasatomi M, Sakairi T, Kaneko Y, et al. The analysis of renal infiltrating cells in acute tubulointerstitial nephritis induced by anti-PD-1 antibodies: a case report and review of literature. Intern Med. 2018;57:3135–9.
The STROBE checklist. http://www.plosmedicine.org/. Accessed 6 Mar 2020
Kellum JA, Lameire N, Aspelin P, Barsoum RS, Burdmann EA, Goldstein SL, et al. Kidney disease: Improving Global Outcomes (KDIGO) acute kidney injury work group. KDIGO clinical practice guideline from acute kidney injury. Kidney Int Suppl. 2012;2:1–138.
Sndararajan V, Henderson T, Perry C, Muggivan A, Quan H, Ghali WA. New ICD-10 version of the Charlson comorbidity index predicted in-hospital mortality. J Clin Epidemiol. 2004;57:1288–94.
Goldman L, Schafer AI. Chronic viral and autoimmune hepatitis. In: Goldman-Cecil Medicine. 25th ed. Philadelphia: Elsevier Saunders; 2014. p. 993–1006.
Matsuo S, Imai E, Horio M, Yasuda Y, Tomita K, Nitta K, et al. Collaborators developing the Japanese equation for estimated GFR Revised equations for estimated GFR from serum creatinine in Japan. Am J Kidney Dis. 2009;53:982–92.
Maan R, Al Marzooqi SH, Klair JS, Karkada J, Cerocchi O, Kowgier M, et al. The frequency of acute kidney injury in patients with chronic hepatitis C virus infection treated with sofosbuvir-based regimens. Aliment Pharmacol Ther. 2017;46:46–55.
Brown PR, Sadiq O, Weick A, Lenhart A, Elbatta M, Fernandez C, et al. Acute kidney injury in patients undergoing chronic hepatitis C virus treatment with Ledipasvir/Sofosbuvir. Hepatol Commun. 2018;24:1172–8.
Gibson A, Gaulkner L, Lichtenfels M, Ogese M, Park BK, Naisbitt DJ. The effect of inhibitory signals on the priming of drug hapten-specific T cells that express distinct Vβ receptors. J Immunol. 2017;199:1223–7.
Wesson DE, Buysse JM, Bushinsky DA. Mechanisms of metabolic acidosis-induced kidney injury in chronic kidney disease. J Am Soc Nephrol. 2020. https://doi.org/10.1681/ASN.2019070677 (Accessed March 6, 2020).
Herbst RS, Baas P, Kim DW, Felip E, Pérez-Gracia JL, Han JY, et al. Pembrolizumab versus docetaxel for previously treated, PD-L1-positive, advanced non-small-cell lung cancer (KEYNOTE-010): a randomized controlled trial. Lancet. 2016;387:1540–50.
Reck M, Rodríguez-Abreu D, Robinson AG, Hui R, Csőszi T, Fűlőp A, et al. Pembrolizumab versus chemotherapy for PD-L1-positive non-small-cell lung cancer. N Engl J Med. 2016;375:1823–33.
Bellmunt J, Vaughn WDJ, Lee FJL, Fong L, Vogelzang NJ, Climent MA, et al. Pembrolizumab as second-line therapy for advanced urothelial carcinoma. N Engl J Med. 2017;376:1015–26.
Rizo-Topete LM, Rosner MH, Ronco C. Acute kidney injury risk assessment and nephrology rapid response team. Blood Purif. 2017;43:82–8.
Koyner JL, Carey KA, Edelson DP, Churpek MM. The development of a machine learning inpatient acute kidney injury prediction model. Crit Care Med. 2018;46:1070–7.
Mendu ML, Ciociolo GR Jr, Mclaughlin SR, Graham DA, Ghazinouri R, Parmar S, et al. A decision-making algorithm for initiation and discontinuation of RRT in severe AKI. Clin J Am Soc Nephrol. 2017;12:228–36.
Park S, Baek SH, Ahn S, Lee KH, Hwang H, Ryu J, et al. Impact of electronic acute kidney injury (AKI) alerts with automated nephrologist consultation on detection and severity of AKI: a quality improvement study. Am J Kidney Dis. 2018;71:9–19.
Al-Jaghbeer M, Dealmeida D, Bilderback A, Ambrosino R, Kellum JA. Clinical decision support for in-hospital AKI. J Am Soc Nephrol. 2018;29:654–60.
Brahmer JR, Lacchetti C, Schneider BJ, Atkins MB, Brassil KJ, Caterino JM, et al. Management of immune-related adverse events in patients treated with immune checkpoint inhibitor therapy: American Society of Clinical Oncology clinical practice guideline. J Clin Oncol. 2018;36:1714–68.
González E, Gutiérrez E, Galeano C, Chevia C, de Sequera P, Bernis C, et al. Early steroid treatment improves the recovery of renal function in patients with drug-induced acute interstitial nephritis. Kidney Int. 2008;73:940–6.
Fernandez-Juarez G, Perez JV, Caravaca-Fontán F, Quintana L, Shabaka A, Rodriguez E, et al. Duration of treatment with corticosteroids and recovery of kidney function in acute interstitial nephritis. Clin J Am Soc Nephrol. 2018;13:1851–8.
Acknowledgments
We would like to thank Editage (www.editage.jp) for English language editing.
Author information
Authors and Affiliations
Contributions
Conceptualization: YS and HT; Methodology: YS, SW, KA, and TM; Formal analysis and investigation: YS, YO, and SW; Writing: original draft preparation: YS; Writing: review and editing: HT; Supervision: HT.
Corresponding author
Ethics declarations
Conflict of interest
The authors have declared that no conflict of interest exists.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee at which the studies were conducted (IRB approval number 2-019096-00) and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Shimamura, Y., Watanabe, S., Maeda, T. et al. Incidence and risk factors of acute kidney injury, and its effect on mortality among Japanese patients receiving immune check point inhibitors: a single-center observational study. Clin Exp Nephrol 25, 479–487 (2021). https://doi.org/10.1007/s10157-020-02008-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10157-020-02008-1