Abstract
Background
To retard progression of chronic kidney disease (CKD) and reduce end-stage kidney disease, it is important to detect acute kidney function worsening on CKD (AW-CKD) immediately and bring back their kidney functions to baselines by appropriate treatment. However, in general outpatient practice, it is difficult to detect the change in the slope of estimated glomerular filtration rate (eGFR).
Methods
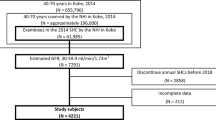
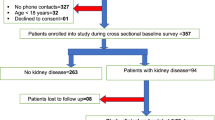
We made automatic calculation system for the rate of eGFR decline (ΔeGFR), and retrospectively observed the situation of AW-CKD among outpatients, who had visited all clinical departments of Steel Memorial Hirohata Hospital between May and August 2016, using the system. The patients with ΔeGFR over 20 mL/min/1.73 m2/year were classified into “Detected cases”, who were immediately detected AW-CKD by the attending physicians, and “Not detected cases”. For each stratum of ΔeGFR, subsequent eGFR recovery rates between two groups were compared.
Results
Among 6719 outpatients, 865 had CKD stages G3–5 and of which 196 had ΔeGFR over 20 mL/min/1.73 m2/year. We revealed that, in cases of ΔeGFR over 30 mL/min/1.73 m2/year, eGFR recovery rates in “Detected cases” were significantly higher than those in “Not detected cases” (103.2 vs 43.9%, p < 0.001). There were no differences in the clinical backgrounds except kidney function between two groups.
Conclusion
In general outpatient practice, a substantial number of AW-CKD was latent. It is expected to improve kidney prognoses of outpatients having CKD through immediately detecting the patients, whose ΔeGFR over 30 mL/min/1.73 m2/year using the system and alerting the attending physicians on the electronic medical record.







Similar content being viewed by others
References
Matsuo S, Imai E, Horio M, et al. Revised equations for estimated GFR from serum creatinine in Japan. Am J Kidney Dis. 2009;53(6):982–92.
Kidney Disease Improving Global Outcomes (KDIGO) Acute Kidney Injury Work Group. KDIGO clinical practice guideline for acute kidney injury. Kidney Int. 2012;Suppl 2:1–138.
Imai E, Horio M, Yamagata K, et al. Slower decline of glomerular filtration rate in the Japanese general population: a longitudinal 10-year follow-up study. Hypertens Res. 2008;31(3):433–41.
Eriksen BO, Ingebretsen OC. The progression of chronic kidney disease: a 10-year population-based study of the effects of gender and age. Kidney Int. 2006;69:375–82.
Molitch ME, DeFronzo RA, Franz MJ, et al. Diabetic nephropathy. Diabetes Care. 2003;26(Suppl 1):S94–S98.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have declared that no conflict of interest exists.
Human and animal rights
All procedures performed in the present study involving human participants were in accordance with the ethical standards of the Institutional Review Board of Steel Memorial Hirohata Hospital (IRB Approval No. 2924) and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. This article does not contain any studies with animals performed by any of the authors.
Informed consent
Since the present study is a retrospective observational study using only past clinical data, it does not involve invasion or intervention. Therefore, document or verbal consent from each study subject is not required. In accordance with “Ethical guidelines for medical and health research involving human subjects (the Ministry of Education, Culture, Sports, Science and Technology/the Ministry of Health, Labour and Welfare in Japan, revised in 2017)”, by posting documents at conspicuous places in the hospital, we notified study subjects of study content so that they could easily understand it, and guaranteed opportunities for them to refuse it (opt out). If there was an offer that they would not want to be a subject of the study, we quickly excluded the subject’s data from the analysis and did not use their clinical information in this study.
About this article
Cite this article
Ito, J., Kita, H. & Fujisawa, T. Significance of early detection of acute kidney function worsening among outpatients having CKD using automatic calculation system for the rate of eGFR decline. Clin Exp Nephrol 22, 898–905 (2018). https://doi.org/10.1007/s10157-018-1541-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10157-018-1541-y