Abstract
Background
Arterial hypertrophy and interstitial fibrosis are important characteristics in kidneys of angiotensinogen-knockout (Atg −/−) mice. In these mice, which exhibit polyuria and hypotension, sympathetic nerve signaling is estimated to be compensatorily hyperactive. Furthermore, transforming growth factor (TGF)-β1 is overexpressed in mice kidneys. To determine whether sympathetic nerve signaling and TGF-β1 exacerbate arterial hypertrophy and interstitial fibrosis, intervention studies of such signaling are required.
Methods
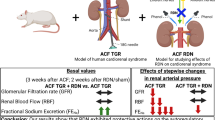
We performed renal denervation and administered the α2-adrenergic receptor (AR) antagonist, atipamezole, to Atg −/− mice. A renin inhibitor, aliskiren, which was preliminarily confirmed to reduce TGF-β1 gene expression in kidneys of the mice, was additionally administered to assess the effect on the arterial hypertrophy and interstitial fibrosis.
Results
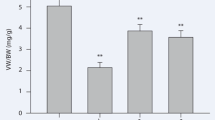
Norepinephrine content in kidneys of Atg −/− mice was three times higher than in kidneys of wild-type mice. Interventions by renal denervation and atipamezole resulted in amelioration of the histological findings. Overexpression of TGF-β1 gene in kidneys of Atg −/− mice was altered in a manner linked to the histological findings. Surprisingly, aliskiren reduced α2-AR gene expression, interstitial fibrosis, and arterial hypertrophy in kidneys of Atg −/− mice, which lack renin substrate.
Conclusions
Alpha2-AR signaling is one of the causes of persistent renal arterial hypertrophy in Atg −/− mice. Aliskiren also angiotensinogen-independently reduces the extent of renal arterial hypertrophy, partly thorough downregulation of α2-ARs. Although renal arterial hypertrophy in Atg −/− mice appears to be of multifactorial origin, TGF-β1 may play a key role in the persistence of such hypertrophy.





Similar content being viewed by others
References
Tanimoto K, Sugiyama F, Goto Y, Ishida J, Takimoto E, Yagami K, et al. Angiotensinogen-deficient mice with hypotension. J Biol Chem. 1994;269(50):31334–7.
Kihara M, Umemura S, Sumida Y, Yokoyama N, Yabana M, Nyui N, et al. Genetic deficiency of angiotensinogen produces an impaired urine concentrating ability in mice. Kidney Int. 1998;53(3):548–55.
Niimura F, Labosky PA, Kakuchi J, Okubo S, Yoshida H, Oikawa T, et al. Gene targeting in mice reveals a requirement for angiotensin in the development and maintenance of kidney morphology and growth factor regulation. J Clin Invest. 1995;96(6):2947–54.
Kim J, Padanilam BJ. Renal nerves drive interstitial fibrogenesis in obstructive nephropathy. J Am Soc Nephrol.
Zheng H, Liu X, Rao US, Patel KP. Increased renal ENaC subunits and sodium retention in rats with chronic heart failure. Am J Physiol Renal Physiol. 2011;300(3):F641–F9.
Westermann D, Riad A, Lettau O, Roks A, Savvatis K, Becher PM, et al. Renin inhibition improves cardiac function and remodeling after myocardial infarction independent of blood pressure. Hypertension. 2008;52(6):1068–75.
Inokuchi S, Kimura K, Sugaya T, Inokuchi K, Murakami K, Sakai T. Hyperplastic vascular smooth muscle cells of the intrarenal arteries in angiotensin II type 1a receptor null mutant mice. Kidney Int. 2001;60(2):722–31.
Racasan S, Hahnel B, van der Giezen DM, Blezer EL, Goldschmeding R, Braam B, et al. Temporary losartan or captopril in young SHR induces malignant hypertension despite initial normotension. Kidney Int. 2004;65(2):575–81.
Hashimoto T, Kihara M, Imai N, Yoshida S, Shimoyamada H, Yasuzaki H, et al. Requirement of apelin–apelin receptor system for oxidative stress-linked atherosclerosis. Am J Pathol. 2007;171(5):1705–12.
Oliver JA. Receptor-mediated actions of renin and prorenin. Kidney Int. 2006;69(1):13–5.
Feldman DL, Jin L, Xuan H, Contrepas A, Zhou Y, Webb RL, et al. Effects of aliskiren on blood pressure, albuminuria, and (pro)renin receptor expression in diabetic TG(mRen-2)27 rats. Hypertension. 2008;52(1):130–6.
Border WA, Noble NA. TGF-beta in kidney fibrosis: a target for gene therapy. Kidney Int. 1997;51(5):1388–96.
Sato M, Muragaki Y, Saika S, Roberts AB, Ooshima A. Targeted disruption of TGF-beta1/Smad3 signaling protects against renal tubulointerstitial fibrosis induced by unilateral ureteral obstruction. J Clin Invest.
Guan Q, Li S, Gao S, Chen H, Nguan CY, Du C. Reduction of chronic rejection of renal allografts by anti-transforming growth factor-beta antibody therapy in a rat model. Am J Physiol Renal Physiol.
Blobe GC, Schiemann WP, Lodish HF. Role of transforming growth factor beta in human disease. N Engl J Med. 2000;342(18):1350–8.
Suzuki H, Okada K, Abe M, Maruyama N, Yoshida Y, Baba S, et al. Aliskiren reduces home blood pressure and albuminuria in patients with hypertensive nephrosclerosis. Clin Exp Nephrol. 2013;17(3):386–95.
Acknowledgements
We acknowledge Ms. Hiroko Morinaga and Ms. Emi Maeda, Department of Medical Science and Cardiorenal Medicine, Yokohama City University Graduate School of Medicine and School of Medicine for technical assistance. S.Y. and S.U. received research funding from Novartis Pharmaceuticals.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare no conflicts of interest.
Ethical statement
Experiments were conducted under the guidelines for animal experiments set by the Animal Experiment Committee of Yokohama City University School of Medicine (Approval number F-A-14-042).
Informed consent
In this study, informed consent was not required because any human sample was not used.
Electronic supplementary material
Below is the link to the electronic supplementary material.
About this article
Cite this article
Nakamori, H., Yoshida, Si., Ishiguro, H. et al. Arterial wall hypertrophy is ameliorated by α2-adrenergic receptor antagonist or aliskiren in kidneys of angiotensinogen-knockout mice. Clin Exp Nephrol 22, 773–781 (2018). https://doi.org/10.1007/s10157-017-1520-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10157-017-1520-8