Abstract
Background
Glomerular hypertension aggravates glomerular sclerosis by inducing growth factors, e.g., transforming growth factor-β (TGF-β) to mesangial matrix expansion. Smads are intracellular proteins that transmit signals from TGF-β to nucleus, and Smads are also negatively regulated by inhibitory Smads (I-Smads), Smad6 and Smad7. However, little is known about the role of I-Smads in glomerular hypertension. We studied I-Smad expression in cultured mesangial cells subjected to mechanical stretch as an in vitro model of glomerular hypertension.
Methods
Rat mesangial cells were cultured under cyclic mechanical stretch conditions using the Flexercell Strain Unit. Phosphorylated Smad1 and Smad2 were determined by Western blots. The expression of Smad6 and Smad7 mRNAs was determined by Northern blots. Stretch-mediated I-Smad mRNAs of cells pre-treated with MAPK-ERK kinase inhibitor, U0126, were also determined. Localization of phospho-Smad1, Smad6 and Smad7 proteins in the glomerulus of Dahl salt-sensitive rats was determined by immunohistochemistry.
Results
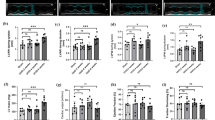
Stretch stress increased phospho-Smad1 levels, and significantly decreased Smad6 mRNA to 32 % of control, and increased Smad7 mRNA to 136 % of control. U0126 significantly attenuated stretch-mediated decreases in Smad6 mRNA, but had no effect on stretch-mediated increases in Smad7 mRNA. Phospho-Smad1, Smad6 and Smad7 proteins were localized in podocytes and mesangial cells of Dahl rats.
Conclusion
Mechanical stretch increases phospho-Smad1 levels and down-regulates Smad6 mRNA expression in mesangial cells. Stretch-mediated down-regulation of Smad6 is partially involved in ERK1/2 activation. These results indicate that glomerular hypertension might augment Smad1 signaling with concomitant attenuation of Smad6-mediated negative feedback.





Similar content being viewed by others
References
Riser BL, Cortes P, Zhao X, Bernstein J, Dumler F, Narins RG. Intraglomerular pressure and mesangial stretching stimulate extracellular matrix formation in the rat. J Clin Invest. 1992;90:1932–43.
Hori Y, Katoh T, Hirakata M, Joki N, Kaname S, Fukagawa M, et al. Anti-latent TGF-β binding protein-1 antibody or synthetic oligopeptides inhibit extracellular matrix expression induced by stretch in cultured rat mesangial cells. Kidney Int. 1998;53:1616–25.
Ishida T, Haneda M, Maeda S, Koya D, Kikkawa R. Stretch-induced overproduction of fibronectin in mesangial cells is mediated by the activation of mitogen-activated protein kinase. Diabetes. 1999;48:595–602.
Moustakas A, Heldin CH. The regulation of TGFβ signal transduction. Development. 2009;136:3699–714.
Chen YG, Hata A, Lo RS, Wotton D, Shi Y, Pavletich N, et al. Determinants of specificity in TGF-β signal transduction. Genes Dev. 1998;12:2144–52.
Hanyu A, Ishidou Y, Ebisawa T, Shimanuki T, Imamura T, Miyazono K. The N domain of smad7 is essential for specific inhibition of transforming growth factor-β signaling. J Cell Biol. 2001;155:1017–27.
Pannu J, Nakerakanti S, Smith E, ten Dijke P, Trojanowska M. Transforming growth factor-β receptor type I-dependent fibrogenic gene program is mediated via activation of smad1 and ERK1/2 pathways. J Biol Chem. 2007;282:10405–13.
Mima A, Abe H, Nagai K, Arai H, Matsubara T, Araki M, et al. Activation of Src mediates PDGF-induced Smad1 phosphorylation and contributes to the progression of glomerulosclerosis in glomerulonephritis. PLoS ONE. 2011;6:e17929.
Nakerakanti SS, Bujor AM, Trojanowska M. Ccn2 is required for the TGF-β induced activation of Smad1-ERK1/2 signaling network. PLoS ONE. 2011;6:e21911.
Abe H, Matsubara T, Arai H, Doi T. Role of Smad1 in diabetic nephropathy: molecular mechanisms and implications as a diagnostic marker. Histol Histopathol. 2011;26:531–41.
Schiffer M, Schiffer LE, Gupta A, Shaw AS, Roberts IS, Mundel P, et al. Inhibitory Smads and tgf-beta signaling in glomerular cells. J Am Soc Nephrol. 2002;13:2657–66.
Riser BL, Cortes P, Heilig C, Grondin J, Ladson-Wofford S, Patterson D, et al. Cyclic stretching force selectively up-regulates transforming growth factor-beta isoforms in cultured rat mesangial cells. Am J Pathol. 1996;148:1915–23.
Yasuda T, Kondo S, Homma T, Harris RC. Regulation of extracellular matrix by mechanical stress in rat glomerular mesangial cells. J Clin Invest. 1996;98:1991–2000.
Kido S, Kuriwaka-Kido R, Umino-Miyatani Y, Endo I, Inoue D, Taniguchi H, et al. Mechanical stress activates Smad pathway through PKCδ to enhance interleukin-11 gene transcription in osteoblasts. PLoS One. 2010;5:e13090.
Abe H, Matsubara T, Iehara N, Nagai K, Takahashi T, Arai H, et al. Type IV collagen is transcriptionally regulated by Smad1 under advanced glycation end product (AGE) stimulation. J Biol Chem. 2004;279:14201–6.
Tsunobuchi H, Ishisaki A, Imamura T. Expressions of inhibitory Smads, Smad6 and Smad7, are differentially regulated by TPA in human lung fibroblast cells. Biochem Biophys Res Commun. 2004;316:712–9.
Matsubara T, Abe H, Arai H, Nagai K, Mima A, Kanamori H, et al. Expression of Smad1 is directly associated with mesangial matrix expansion in rat diabetic nephropathy. Lab Invest. 2006;86:357–68.
Mima A, Arai H, Matsubara T, Abe H, Nagai K, Tamura Y, et al. Urinary Smad1 is a novel marker to predict later onset of mesangial matrix expansion in diabetic nephropathy. Diabetes. 2008;57:1712–22.
Acknowledgments
This work was supported by a research grant 11671042 from the Ministry of Education and Science of Japan. We thank Dr. K. Miyazono for generous donations of cDNA clones, and we thank Hiroko Ohashi and Atsuko Hashimoto for technical assistance.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Hayashi, Y., Katoh, T., Asano, K. et al. Mechanical stretch down-regulates expression of the Smad6 gene in cultured rat mesangial cells. Clin Exp Nephrol 16, 690–696 (2012). https://doi.org/10.1007/s10157-012-0630-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10157-012-0630-6