Abstract
Background
We recently reported that nephrin, a major slit-diaphragm protein, is phosphorylated at Y1204 and Y1228 in normal human glomeruli and that phosphorylation decreased significantly in minimal-change nephrosis. These results indicate that phosphorylation of nephrin is important for maintenance of normal podocyte morphology and function. On the other hand, phosphorylation of nephrin was reportedly increased in certain animal models of glomerular injury.
Methods
We performed immunofluorescent and immunoelectron staining of phosphorylated nephrin in human kidney biopsy specimens of membranous nephropathy (MN) to investigate whether phosphorylation of nephrin was altered in human MN and whether it correlated with MN staging.
Results
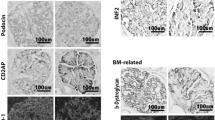
Although aberrant localization of phosphorylated nephrin was detected using immunoelectron microscopy in stage I MN, a decrease in the immunofluorescent intensity of phosphorylated nephrin was not observed in stage I, and only a slight decrease was seen in stages II, III, and IV compared with controls. No significant correlation between nephrin phosphorylation and proteinuria was observed.
Conclusion
Nephrin phosphorylation was not significantly decreased in the early stage of MN.



Similar content being viewed by others
References
Kestila M, Lenkkeri U, Mannikko M, Lamerdin J, McCready P, Putaala H, et al. Positionally cloned gene for a novel glomerular protein––nephrin––is mutated in congenital nephrotic syndrome. Mol Cell. 1998;1:575–82.
Khoshnoodi J, Sigmundsson K, Ofverstedt LG, Skoglund U, Obrink B, Wartiovaara J, et al. Nephrin promotes cell-cell adhesion through homophilic interactions. Am J Pathol. 2003;163:2337–46.
Jones N, Blasutig IM, Eremina V, Ruston JM, Bladt F, Li H, et al. Nck adaptor proteins link nephrin to the actin cytoskeleton of kidney podocytes. Nature. 2006;440:818–23.
Verma R, Kovari I, Soofi A, Nihalani D, Patrie K, Holzman LB. Nephrin ectodomain engagement results in Src kinase activation, nephrin phosphorylation, Nck recruitment, and actin polymerization. J Clin Invest. 2006;116:1346–59.
Li H, Zhu J, Aoudjit L, Latreille M, Kawachi H, Larose L, et al. Rat nephrin modulates cell morphology via the adaptor protein Nck. Biochem Biophys Res Commun. 2006;349:310–6.
Lu W, Katz S, Gupta R, Mayer BJ. Activation of Pak by membrane localization mediated by an SH3 domain from the adaptor protein Nck. Curr Biol. 1997;7:85–94.
Rohatgi R, Nollau P, Ho HY, Kirschner MW, Mayer BJ. Nck and phosphatidylinositol 4,5-bisphosphate synergistically activate actin polymerization through the N-WASP-Arp2/3 pathway. J Biol Chem. 2001;276:26448–52.
Verma R, Wharram B, Kovari I, Kunkel R, Nihalani D, Wary KK, et al. Fyn binds to and phosphorylates the kidney slit diaphragm component nephrin. J Biol Chem. 2003;278:20716–23.
Uchida K, Suzuki K, Iwamoto M, Kawachi H, Ohno M, Horita S, et al. Decreased tyrosine phosphorylation of nephrin in rat and human nephrosis. Kidney Int. 2008;73:926–32.
Li H, Lemay S, Aoudjit L, Kawachi H, Takano T. SRC-family kinase Fyn phosphorylates the cytoplasmic domain of nephrin and modulates its interaction with podocin. J Am Soc Nephrol. 2004;15:3006–15.
Churg J, Ehrenreich T. Membranous nephropathy. Perspect Nephrol Hypertens. 1973;1(Pt 1):443–8.
Sohara E, Rai T, Yang SS, Uchida K, Nitta K, Horita S, et al. Pathogenesis and treatment of autosomal-dominant nephrogenic diabetes insipidus caused by an aquaporin 2 mutation. Proc Natl Acad Sci USA. 2006;103:14217–22.
Yang SS, Morimoto T, Rai T, Chiga M, Sohara E, Ohno M, et al. Molecular pathogenesis of pseudohypoaldosteronism type II: generation and analysis of a Wnk4(D561A/+) knockin mouse model. Cell Metab. 2007;5:331–44.
Acknowledgments
We thank Ms. K. Sasaki for technical assistance.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Ohashi, T., Uchida, K., Asamiya, Y. et al. Phosphorylation status of nephrin in human membranous nephropathy. Clin Exp Nephrol 14, 51–55 (2010). https://doi.org/10.1007/s10157-009-0241-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10157-009-0241-z