Abstract
Background
In the present guidelines for the management of metastatic castration-resistant prostate cancer (mCRPC), it is unclear who benefits most from androgen receptor axis-targeted agents (ARATs) or docetaxel as the first-line treatment.
Methods
We conducted a retrospective study to explore new treatment-specific biomarkers in mCRPC. A total of 211 patients with mCRPC who received either ARAT or docetaxel as first-line treatment were included. Patients were compared for radiographic progression and prostate-specific antigen (PSA) progression. Multivariable Cox regression models were used to assess the association between pretreatment biomarkers and risk of events. The statistical interaction between biomarkers and clinical outcomes was also evaluated.
Results
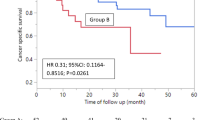
Of all analyzed biomarkers, multivariable Cox regression models identified MPV [≤ median (9.7 fL)] as an independent prognostic factor of radiographic progression [hazard ratio (HR), 2.35; 95% confidence interval (CI), 1.15–4.80; P = 0.019] and PSA progression (HR, 1.96; 95% CI, 1.01–3.95; P = 0.048) in patients treated with ARAT, whereas such associations were not observed in those treated with docetaxel. Interaction analyses showed that those initially treated with docetaxel have lower risk of radiographic progression (HR, 0.33; 95% CI, 0.13–0.79; P = 0.014) and PSA progression (HR, 0.48; 95% CI, 0.23–0.98; P = 0.044) than ARAT when MPV was small.
Conclusions
The present study identified pretreatment MPV as a significant treatment-specific prognostic factor of PSA and radiographic progression in patients with mCRPC who received first-line treatment. Furthermore, our results suggested that those with small MPV may better be treated initially with docetaxel than ARAT.


Similar content being viewed by others
References
Bray F, Ferlay J, Soerjomataram I et al (2018) Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 68:394–424. https://doi.org/10.3322/caac.21492
Cornford P, Bellmunt J, Bolla M et al (2017) EAU-ESTRO-SIOG guidelines on prostate cancer. Part II: treatment of relapsing, metastatic, and castration-resistant prostate cancer. Eur Urol 71:630–642. https://doi.org/10.1016/j.eururo.2016.08.002
Cookson MS, Roth BJ, Dahm P et al (2013) Castration-resistant prostate cancer: AUA Guideline. J Urol 190:429–438. https://doi.org/10.1016/j.juro.2013.05.005
Armstrong AJ, Lin P, Higano CS et al (2018) Development and validation of a prognostic model for overall survival in chemotherapy-naïve men with metastatic castration-resistant prostate cancer. Ann Oncol Off J Eur Soc Med Oncol 29:2200–2207. https://doi.org/10.1093/annonc/mdy406
Chi KN, Kheoh T, Ryan CJ et al (2016) A prognostic index model for predicting overall survival in patients with metastatic castration-resistant prostate cancer treated with abiraterone acetate after docetaxel. Ann Oncol 27:454–460. https://doi.org/10.1093/annonc/mdv594
Fukuokaya W, Kimura T, Onuma H et al (2019) Red cell distribution width predicts prostate-specific antigen response and survival of patients with castration-resistant prostate cancer treated with androgen receptor axis-targeted agents. Clin Genitourin Cancer 17:223–230. https://doi.org/10.1016/j.clgc.2019.04.010
Armstrong AJ, Halabi S, Luo J et al (2019) prospective multicenter validation of androgen receptor splice variant 7 and hormone therapy resistance in high-risk castration-resistant prostate cancer: the prophecy study. J Clin Oncol 37:1120–1129. https://doi.org/10.1200/JCO.18.01731
Templeton AJ, Pezaro C, Omlin A et al (2014) Simple prognostic score for metastatic castration-resistant prostate cancer with incorporation of neutrophil-to-lymphocyte ratio. Cancer 120:3346–3352. https://doi.org/10.1002/cncr.28890
Scher HI, Graf RP, Schreiber NA et al (2018) Assessment of the validity of nuclear-localized androgen receptor splice variant 7 in circulating tumor cells as a predictive biomarker for castration-resistant prostate cancer. JAMA Oncol 4:1179–1186. https://doi.org/10.1001/jamaoncol.2018.1621
Antonarakis ES, Lu C, Wang H et al (2014) AR-V7 and resistance to enzalutamide and abiraterone in prostate cancer. N Engl J Med 371:1028–1038. https://doi.org/10.1056/NEJMoa1315815
Scher HI, Lu D, Schreiber NA et al (2016) Association of AR-V7 on circulating tumor cells as a treatment-specific biomarker with outcomes and survival in castration-resistant prostate cancer. JAMA Oncol 2:1441–1449. https://doi.org/10.1001/jamaoncol.2016.1828
Tjon-Kon-Fat L-A, Lundholm M, Schröder M et al (2018) Platelets harbor prostate cancer biomarkers and the ability to predict therapeutic response to abiraterone in castration resistant patients. Prostate 78:48–53. https://doi.org/10.1002/pros.23443
Wang X, Cui M-M, Xu Y et al (2017) Decreased mean platelet volume predicts poor prognosis in invasive bladder cancer. Oncotarget 8:68115–68122
Chang J, Lin G, Ye M et al (2019) Decreased mean platelet volume predicts poor prognosis in metastatic colorectal cancer patients treated with first-line chemotherapy: results from mCRC biomarker study. BMC Cancer 19:15. https://doi.org/10.1186/s12885-018-5252-2
Gunduz S, Mutlu H, Uysal M et al (2014) Elucidating the correlation between treatment with tyrosine kinase inhibitors and mean platelet volume in patients with metastatic renal cell cancer. Oncol Lett 8:2249–2252. https://doi.org/10.3892/ol.2014.2495
Cao W, Yao X, Cen D et al (2019) Prognostic role of pretreatment thrombocytosis on survival in patients with cervical cancer: a systematic review and meta-analysis. World J Surg Oncol 17:132. https://doi.org/10.1186/s12957-019-1676-7
Rachidi S, Kaur M, Lautenschlaeger T, Li Z (2019) Platelet count correlates with stage and predicts survival in melanoma. Platelets 00:1–5. https://doi.org/10.1080/09537104.2019.1572879
Scher HI, Halabi S, Tannock I et al (2008) Design and end points of clinical trials for patients with progressive prostate cancer and castrate levels of testosterone: recommendations of the prostate cancer clinical trials working group. J Clin Oncol 26:1148–1159. https://doi.org/10.1200/JCO.2007.12.4487
Sweeney CJ, Chen Y-H, Carducci M et al (2015) Chemohormonal Therapy in Metastatic Hormone-Sensitive Prostate Cancer. N Engl J Med 373:737–746. https://doi.org/10.1056/NEJMoa1503747
Evans TC, Jehle D (1991) The red blood cell distribution width. J Emerg Med 9(Suppl 1):71–74. https://doi.org/10.1016/0736-4679(91)90592-4
Scher HI, Morris MJ, Stadler WM et al (2016) Trial design and objectives for castration-resistant prostate cancer: updated recommendations from the prostate cancer clinical trials working group 3. J Clin Oncol 34:1402–1418. https://doi.org/10.1200/JCO.2015.64.2702
Soloway MS, Hardeman SW, Hickey D et al (1988) Stratification of patients with metastatic prostate cancer based on extent of disease on initial bone scan. Cancer 61:195–202. https://doi.org/10.1002/1097-0142(19880101)61:1<195:aid-cncr2820610133>3.0.co;2-y
Haemmerle M, Stone RL, Menter DG et al (2018) The platelet lifeline to cancer: challenges and opportunities. Cancer Cell 33:965–983. https://doi.org/10.1016/j.ccell.2018.03.002
Karpatkin S, Pearlstein E, Ambrogio C, Coller BS (1988) Role of adhesive proteins in platelet tumor interaction in vitro and metastasis formation in vivo. J Clin Invest 81:1012–1019. https://doi.org/10.1172/JCI113411
Jain S, Harris J, Ware J (2010) Platelets: linking hemostasis and cancer. Arterioscler Thromb Vasc Biol 30:2362–2367. https://doi.org/10.1161/ATVBAHA.110.207514
Gay LJ, Felding-Habermann B (2011) Contribution of platelets to tumour metastasis. Nat Rev Cancer 11:123–134. https://doi.org/10.1038/nrc3004
Yun Z-Y, Zhang X, Liu Y-S et al (2017) Lower mean platelet volume predicts poor prognosis in renal cell carcinoma. Sci Rep 7:6700. https://doi.org/10.1038/s41598-017-07168-x
Yun Z-Y, Zhang X, Liu Z-P et al (2017) Association of decreased mean platelet volume with renal cell carcinoma. Int J Clin Oncol 22:1076–1080. https://doi.org/10.1007/s10147-017-1158-2
Gasparyan AY, Ayvazyan L, Mikhailidis DP, Kitas GD (2011) Mean platelet volume: a link between thrombosis and inflammation? Curr Pharm Des 17:47–58. https://doi.org/10.2174/138161211795049804
Korniluk A, Koper-Lenkiewicz OM, Kamińska J et al (2019) Mean platelet volume (mpv): new perspectives for an old marker in the course and prognosis of inflammatory conditions. Mediators Inflamm 2019:9213074. https://doi.org/10.1155/2019/9213074
Astle WJ, Elding H, Jiang T et al (2016) The allelic landscape of human blood cell trait variation and links to common complex disease. Cell 167:1415–1429.e19. https://doi.org/10.1016/j.cell.2016.10.042
Wolf SS, Patchev VK, Obendorf M (2007) A novel variant of the putative demethylase gene, s-JMJD1C, is a coactivator of the AR. Arch Biochem Biophys 460:56–66. https://doi.org/10.1016/j.abb.2007.01.017
Jin G, Sun J, Kim S-T et al (2012) Genome-wide association study identifies a new locus JMJD1C at 10q21 that may influence serum androgen levels in men. Hum Mol Genet 21:5222–5228. https://doi.org/10.1093/hmg/dds361
Ryan CJ, Molina A, Li J et al (2013) Serum androgens as prognostic biomarkers in castration-resistant prostate cancer: results from an analysis of a randomized phase III trial. J Clin Oncol 31:2791–2798. https://doi.org/10.1200/JCO.2012.45.4595
Armstrong AJ, Halabi S, de Wit R et al (2009) The relationship of body mass index and serum testosterone with disease outcomes in men with castration-resistant metastatic prostate cancer. Prostate Cancer Prostatic Dis 12:88–93. https://doi.org/10.1038/pcan.2008.36
Shiota M, Kashiwagi E, Murakami T et al (2019) Serum testosterone level as possible predictive marker in androgen receptor axis-targeting agents and taxane chemotherapies for castration-resistant prostate cancer. Urol Oncol 37:180.e19–180.e24. https://doi.org/10.1016/j.urolonc.2018.10.020
Zaslavsky AB, Gloeckner-Kalousek A, Adams M et al (2015) Platelet-synthesized testosterone in men with prostate cancer induces androgen receptor signaling. Neoplasia 17:490–496. https://doi.org/10.1016/j.neo.2015.05.003
Oudard S, Fizazi K, Sengeløv L et al (2017) Cabazitaxel versus docetaxel as first-line therapy for patients with metastatic castration-resistant prostate cancer: a randomized phase III Trial-FIRSTANA. J Clin Oncol 35:3189–3197. https://doi.org/10.1200/JCO.2016.72.1068
Eisenberger M, Hardy-Bessard A-C, Kim CS et al (2017) Phase III Study comparing a reduced dose of Cabazitaxel (20 mg/m(2)) and the currently approved dose (25 mg/m(2)) in Postdocetaxel patients with metastatic castration-resistant prostate cancer-PROSELICA. J Clin Oncol 35:3198–3206. https://doi.org/10.1200/JCO.2016.72.1076
James ND, de Bono JS, Spears MR et al (2017) Abiraterone for prostate cancer not previously treated with hormone therapy. N Engl J Med 377:338–351. https://doi.org/10.1056/NEJMoa1702900
Beer TM, Armstrong AJ, Rathkopf DE et al (2014) Enzalutamide in metastatic prostate cancer before chemotherapy. N Engl J Med 371:424–433. https://doi.org/10.1056/NEJMoa1405095
Funding
Not applicable.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest directly relevant to the content of this article.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained in the form of opt-out on the website. Those who rejected were excluded.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
About this article
Cite this article
Fukuokaya, W., Kimura, T., Urabe, F. et al. Blood platelet volume predicts treatment-specific outcomes of metastatic castration-resistant prostate cancer. Int J Clin Oncol 25, 1695–1703 (2020). https://doi.org/10.1007/s10147-020-01712-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10147-020-01712-y