Abstract
Background
Taxane/platinum (TP)-based combination chemotherapy is standard for the treatment of metastatic or recurrent cervical cancer. The aim of this study was to investigate the efficacy of postoperative TP therapy in early stage cervical cancer.
Methods
A retrospective review of patients with FIGO IB–IIB stage cervical cancer who were treated with radical hysterectomy and displayed surgical-pathological risk factors was performed. 122 patients were identified between 2003 and 2012. Survival was analyzed by Kaplan–Meier method and compared by the log-rank test. The Cox proportional hazards model was used to investigate predictors of survival.
Results
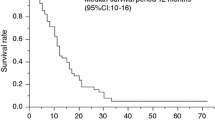
The median follow-up period was 82.4 months. The postoperative adjuvant therapy was TP in 82 (67.2%) patients, other chemotherapies in 10 (8.2%), radiotherapy (RT) in 25 (20.5%), and no further therapy (NFT) in 5 (4.1%). Survival was analyzed using 4 subgroups according to the postoperative adjuvant therapy. The estimated 5-year overall survival was 95.1% in the TP group, 90.0% in the other chemotherapy group, 78.9% in the RT group, and 100% in the NFT group. No significant difference of survival was observed in the subgroups. However, when analyzing only patients who displayed high-risk factors, non-TP adjuvant therapy (including RT and other chemotherapies) was independently associated with shorter survival on multivariate analysis. In the TP group, multivariate analysis revealed that a positive surgical margin was a significant predictor of shorter survival.
Conclusions
Postoperative TP is effective in patients with surgically treated early stage cervical cancer. In these populations, a positive surgical margin could be associated with poor prognosis.



Similar content being viewed by others
References
Organization WH World Health Organization. http://www.who.int/cancer/prevention/diagnosis-screening/cervical-cancer/en/. Accessed 24 May 2017
Cancer Research UK. http://www.cancerresearchuk.org/health-professional/cancer-statistics/statistics-by-cancer-type/cervical-cancer. Accessed 28 May 2017
National Cancer Institute. Surveillance, Epidemiology, and End Results Program. https://seer.cancer.gov/statfacts/html/cervix.html. Accessed 25 May 2017
National Comprehensive Cancer Network Clinical Practice Guidelines in Oncology, Cervical Cancer. https://www.nccn.org/professionals/physician_gls/f_guidelines.asp#site. Accessed 30 May 2017
Peters WA 3rd, Liu PY, Barrett RJ 2nd et al (2000) Concurrent chemotherapy and pelvic radiation therapy compared with pelvic radiation therapy alone as adjuvant therapy after radical surgery in high-risk early-stage cancer of the cervix. J Clin Oncol 18(8):1606–1613
Delgado G, Bundy B, Zaino R et al (1990) Prospective surgical-pathological study of disease-free interval in patients with stage IB squamous cell carcinoma of the cervix: a Gynecologic Oncology Group study. Gynecol Oncol 38(3):352–357
Estape RE, Angioli R, Madrigal M et al (1998) Close vaginal margins as a prognostic factor after radical hysterectomy. Gynecol Oncol 68(3):229–232
Sedlis A, Bundy BN, Rotman MZ et al (1999) A randomized trial of pelvic radiation therapy versus no further therapy in selected patients with stage IB carcinoma of the cervix after radical hysterectomy and pelvic lymphadenectomy: a Gynecologic Oncology Group Study. Gynecol Oncol 73(2):177–183
Monk BJ, Wang J, Im S et al (2005) Rethinking the use of radiation and chemotherapy after radical hysterectomy: a clinical-pathologic analysis of a Gynecologic Oncology Group/Southwest Oncology Group/Radiation Therapy Oncology Group trial. Gynecol Oncol 96(3):721–728
Fuller AF Jr, Elliott N, Kosloff C et al (1989) Determinants of increased risk for recurrence in patients undergoing radical hysterectomy for stage IB and IIA carcinoma of the cervix. Gynecol Oncol 33(1):34–39
Soisson AP, Soper JT, Clarke-Pearson DL et al (1990) Adjuvant radiotherapy following radical hysterectomy for patients with stage IB and IIA cervical cancer. Gynecol Oncol 37(3):390–395
Stock RG, Chen AS, Flickinger JC et al (1995) Node-positive cervical cancer: impact of pelvic irradiation and patterns of failure. Int J Radiat Oncol Biol Phys 31(1):31–36
Sevin BU, Lu Y, Bloch DA et al (1996) Surgically defined prognostic parameters in patients with early cervical carcinoma. A multivariate survival tree analysis. Cancer 78(7):1438–1446
Snijders-Keilholz A, Hellebrekers BW, Zwinderman AH et al (1999) Adjuvant radiotherapy following radical hysterectomy for patients with early-stage cervical carcinoma (1984–1996). Radiother Oncol 51(2):161–167
Marchiole P, Buenerd A, Benchaib M et al (2005) Clinical significance of lympho vascular space involvement and lymph node micrometastases in early-stage cervical cancer: a retrospective case-control surgico-pathological study. Gynecol Oncol 97(3):727–732
Van de Putte G, Lie AK, Vach W et al (2005) Risk grouping in stage IB squamous cell cervical carcinoma. Gynecol Oncol 99(1):106–112
Chernofsky MR, Felix JC, Muderspach LI et al (2006) Influence of quantity of lymph vascular space invasion on time to recurrence in women with early-stage squamous cancer of the cervix. Gynecol Oncol 100(2):288–293
Ebina Y, Yaegashi N, Katabuchi H et al (2015) Japan Society of Gynecologic Oncology guidelines 2011 for the treatment of uterine cervical cancer. Int J Clin Oncol 20(2):240–248
Colombo N, Carinelli S, Colombo A et al (2012) Cervical cancer: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol 23(7):vii27–vii32
Beckmann MW, Mallmann P, Uterus Commission of the Gynecological Oncology Working G (2009) Interdisciplinary S2k guideline on the diagnosis and treatment of cervical carcinoma. J Cancer Res Clin Oncol 135(9):1197–1206
Landoni F, Maneo A, Colombo A et al (1997) Randomised study of radical surgery versus radiotherapy for stage Ib-IIa cervical cancer. Lancet 350(9077):535–540
Ryu SY, Park SI, Nam BH et al (2011) Is adjuvant chemoradiotherapy overtreatment in cervical cancer patients with intermediate risk factors? Int J Radiat Oncol Biol Phys 79(3):794–799
Mabuchi S, Okazawa M, Isohashi F et al (2011) Radical hysterectomy with adjuvant radiotherapy versus definitive radiotherapy alone for FIGO stage IIB cervical cancer. Gynecol Oncol 123(2):241–247
Undurraga M, Loubeyre P, Dubuisson JB et al (2010) Early-stage cervical cancer: is surgery better than radiotherapy? Expert Rev Anticancer Ther 10(3):451–460
Tewari KS, Sill MW, Long HJ 3rd et al (2014) Improved survival with bevacizumab in advanced cervical cancer. N Engl J Med 370(8):734–743
Monk BJ, Sill MW, McMeekin DS et al (2009) Phase III trial of four cisplatin-containing doublet combinations in stage IVB, recurrent, or persistent cervical carcinoma: a Gynecologic Oncology Group study. J Clin Oncol 27(28):4649–4655
Kitagawa R, Katsumata N, Shibata T et al (2015) Paclitaxel plus carboplatin versus paclitaxel plus cisplatin in metastatic or recurrent cervical cancer: the open-label randomized phase III trial JCOG0505. J Clin Oncol 33(19):2129–2135
Takekuma M, Kasamatsu Y, Kado N et al (2016) Adjuvant chemotherapy versus concurrent chemoradiotherapy for high-risk cervical cancer after radical hysterectomy and systematic lymphadenectomy. Int J Clin Oncol 21(4):741–747
Lee KB, Lee JM, Ki KD et al (2008) Comparison of adjuvant chemotherapy and radiation in patients with intermediate risk factors after radical surgery in FIGO stage IB-IIA cervical cancer. Int J Gynecol Cancer 18(5):1027–1031
Hosaka M, Watari H, Kato T et al (2012) Clinical efficacy of paclitaxel/cisplatin as an adjuvant chemotherapy for patients with cervical cancer who underwent radical hysterectomy and systematic lymphadenectomy. J Surg Oncol 105(6):612–616
Li S, Hu T, Chen Y et al (2013) Adjuvant chemotherapy, a valuable alternative option in selected patients with cervical cancer. PLoS One 8(9):e73837
Takeshima N, Umayahara K, Fujiwara K et al (2006) Treatment results of adjuvant chemotherapy after radical hysterectomy for intermediate- and high-risk stage IB-IIA cervical cancer. Gynecol Oncol 103(2):618–622
Hosaka M, Watari H, Takeda M et al (2008) Treatment of cervical cancer with adjuvant chemotherapy versus adjuvant radiotherapy after radical hysterectomy and systematic lymphadenectomy. J Obstet Gynaecol Res 34(4):552–556
Jung PS, Kim DY, Lee SW et al (2015) Clinical role of adjuvant chemotherapy after radical hysterectomy for FIGO stage IB-IIA cervical cancer: comparison with adjuvant RT/CCRT using inverse-probability-of-treatment weighting. PLoS One 10(7):e0132298
Querleu D, Morrow CP (2008) Classification of radical hysterectomy. Lancet Oncol 9(3):297–303
Matsuo K, Shimada M, Aoki Y et al (2017) Comparison of adjuvant therapy for node-positive clinical stage IB-IIB cervical cancer: systemic chemotherapy versus pelvic irradiation. Int J Cancer 141(5):1042–1051
Li L, Song X, Liu R et al (2016) Chemotherapy versus radiotherapy for FIGO stages IB1 and IIA1 cervical carcinoma patients with postoperative isolated deep stromal invasion: a retrospective study. BMC Cancer 16:403
Duenas-Gonzalez A, Lopez-Graniel C, Gonzalez-Enciso A et al (2003) A phase II study of multimodality treatment for locally advanced cervical cancer: neoadjuvant carboplatin and paclitaxel followed by radical hysterectomy and adjuvant cisplatin chemoradiation. Ann Oncol 14(8):1278–1284
Park DC, Kim JH, Lew YO et al (2004) Phase II trial of neoadjuvant paclitaxel and cisplatin in uterine cervical cancer. Gynecol Oncol 92(1):59–63
Tanioka M, Yamaguchi S, Shimada M et al (2017) Cisplatin with dose-dense paclitaxel before and after radical hysterectomy for locally advanced cervical cancer: a prospective multicenter phase II trial with a dose-finding study. Med Oncol 34(8):134
Okazawa M, Mabuchi S, Isohashi F et al (2013) Impact of the addition of concurrent chemotherapy to pelvic radiotherapy in surgically treated stage IB1-IIB cervical cancer patients with intermediate-risk or high-risk factors: a 13-year experience. Int J Gynecol Cancer 23(3):567–575
Mabuchi S, Okazawa M, Isohashi F et al (2011) Postoperative whole pelvic radiotherapy plus concurrent chemotherapy versus extended-field irradiation for early-stage cervical cancer patients with multiple pelvic lymph node metastases. Gynecol Oncol 120(1):94–100
Sakuragi N (2007) Up-to-date management of lymph node metastasis and the role of tailored lymphadenectomy in cervical cancer. Int J Clin Oncol 12(3):165–175
Pieterse QD, Kenter GG, Gaarenstroom KN et al (2007) The number of pelvic lymph nodes in the quality control and prognosis of radical hysterectomy for the treatment of cervical cancer. Eur J Surg Oncol 33(2):216–221
Mabuchi S, Isohashi F, Yokoi T et al (2016) A phase II study of postoperative concurrent carboplatin and paclitaxel combined with intensity-modulated pelvic radiotherapy followed by consolidation chemotherapy in surgically treated cervical cancer patients with positive pelvic lymph nodes. Gynecol Oncol 141(2):240–246
Rotman M, Sedlis A, Piedmonte MR et al (2006) A phase III randomized trial of postoperative pelvic irradiation in Stage IB cervical carcinoma with poor prognostic features: follow-up of a gynecologic oncology group study. Int J Radiat Oncol Biol Phys 65(1):169–176
NCT01101451. https://clinicaltrials.gov/ct2/show/NCT01101451. Accessed 11 June 2017
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
10147_2018_1249_MOESM1_ESM.pdf
Supplemental Figure 1. Study selection schema. RT, radiotherapy; PALN, para-aortic lymph node. Supplemental Figure 2. Survival curves in 122 patients who were treated with radical hysterectomy and displayed surgical-pathological risk factors. A, overall survival (OS). B, recurrence-free survival (RFS). The estimated 5-year OS and RFS rates were 91.7% and 87.5%, respectively (PDF 73 kb)
About this article
Cite this article
Okazawa-Sakai, M., Yokoyama, T., Fujimoto, E. et al. Long-term outcomes of postoperative taxane/platinum chemotherapy for early stage cervical cancer: a retrospective study. Int J Clin Oncol 23, 715–725 (2018). https://doi.org/10.1007/s10147-018-1249-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10147-018-1249-8