Abstract
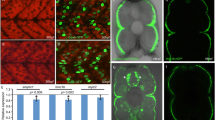
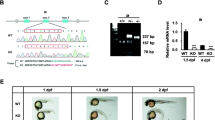
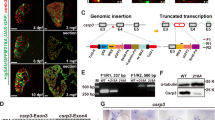
Zebrafish embryonic slow muscle cells, with their superficial localization and clear sarcomere organization, provide a useful model system for genetic analysis of muscle cell differentiation and sarcomere assembly. To develop a quick assay for testing CRISPR-mediated gene editing in slow muscles of zebrafish embryos, we targeted a red fluorescence protein (RFP) reporter gene specifically expressed in slow muscles of myomesin-3-RFP (Myom3-RFP) zebrafish embryos. We demonstrated that microinjection of RFP-sgRNA with Cas9 protein or Cas9 mRNA resulted in a mosaic pattern in loss of RFP expression in slow muscle fibers of the injected zebrafish embryos. To uncover gene functions in sarcomere organization, we targeted two endogenous genes, slow myosin heavy chain-1 (smyhc1) and heat shock protein 90 α1 (hsp90α1), which are specifically expressed in zebrafish muscle cells. We demonstrated that injection of Cas9 protein or mRNA with respective sgRNAs targeted to smyhc1 or hsp90a1 resulted in a mosaic pattern of myosin thick filament disruption in slow myofibers of the injected zebrafish embryos. Moreover, Myom3-RFP expression and M-line localization were also abolished in these defective myofibers. Given that zebrafish embryonic slow muscles are a rapid in vivo system for testing genome editing and uncovering gene functions in muscle cell differentiation, we investigated whether microinjection of Natronobacterium gregoryi Argonaute (NgAgo) system could induce genetic mutations and muscle defects in zebrafish embryos. Single-strand guide DNAs targeted to RFP, Smyhc1, or Hsp90α1 were injected with NgAgo mRNA into Myom3-RFP zebrafish embryos. Myom3-RFP expression was analyzed in the injected embryos. The results showed that, in contrast to the CRISPR/Cas9 system, injection of the NgAgo-gDNA system did not affect Myom3-RFP expression and sarcomere organization in myofibers of the injected embryos. Sequence analysis failed to detect genetic mutations at the target genes. Together, our studies demonstrate that zebrafish embryonic slow muscle is a rapid model for testing gene editing technologies in vivo and uncovering gene functions in muscle cell differentiation.






Similar content being viewed by others
References
Abdelrahman H, ElHady M, Alcivar-Warren A, Allen S, Al-Tobasei R, Bao L, Beck B, Blackburn H, Bosworth B, Buchanan J, Chappell J, Daniels W, Dong S, Dunham R, Durland E, Elaswad A, Gomez-Chiarri M, Gosh K, Guo X, Hackett P, Hanson T, Hedgecock D, Howard T, Holland L, Jackson M, Jin Y, Khalil K, Kocher T, Leeds T, Li N, Lindsey L, Liu S, Liu Z, Martin K, Novriadi R, Odin R, Palti Y, Peatman E, Proestou D, Qin G, Reading B, Rexroad C, Roberts S, Salem M, Severin A, Shi H, Shoemaker C, Stiles S, Tan S, Tang KF, Thongda W, Tiersch T, Tomasso J, Prabowo WT, Vallejo R, van der Steen H, Vo K, Waldbieser G, Wang H, Wang X, Xiang J, Yang Y, Yant R, Yuan Z, Zeng Q, Zhou T (2017) Aquaculture genomics, genetics and breeding in the United States: current status, challenges, and priorities for future research. BMC Genomics 18(1):191
Berger J, Currie PD (2012) Zebrafish models flex their muscles to shed light on muscular dystrophies. Dis Model Mech 5(6):726–732
Bernick EP, Zhang PJ, Du S (2010) Knockdown and overexpression of Unc-45b result in defective myofibril organization in skeletal muscles of zebrafish embryos. BMC Cell Biol 11(1):70
Bohmert K, Camus I, Bellini C, Bouchez D, Caboche M, Benning C (1998) AGO1 defines a novel locus of Arabidopsis controlling leaf development. EMBO J 17(1):170–180
Burgess S, Cheng L, Gu F, Huang J, Huang Z, Lin S, Li J, Li W, Qin W, Sun Y, Songyang Z, Wei W, Wu Q, Wang H, Wang X, Xiong JW, Xi J, Yang H, Zhou B, Zhang B (2016) Questions about NgAgo. Protein Cell 7(12):913–915
Cho SW, Kim S, Kim JM, Kim JS (2013) Targeted genome engineering in human cells with the Cas9 RNA-guided endonuclease. Nat Biotechnol 31(3):230–232
Clark KJ, Balciunas D, Pogoda HM, Ding Y, Westcot SE, Bedell VM, Greenwood TM, Urban MD, Skuster KJ, Petzold AM, Ni J, Nielsen AL, Patowary A, Scaria V, Sivasubbu S, Xu X, Hammerschmidt M, Ekker SC (2011) In vivo protein trapping produces a functional expression codex of the vertebrate proteome. Nat Methods 8(6):506–515
Codina M, Li J, Gutiérrez J, Kao JP, Du SJ (2010) Hsp90alpha1 function results in different effects on myofibril organization in skeletal muscles of zebrafish embryos. PLoS One 5(1):e8416
Currie PD, Ingham PW (1996) Induction of a specific muscle cell type by a hedgehog-like protein in zebrafish. Nature 382(6590):452–455
Currie PD, Ingham PW (1998) The generation and interpretation of positional information within the vertebrate myotome. Mech Dev 73(1):3–21
Dehler CE, Boudinot P, Martin SA, Collet B (2016) Development of an efficient genome editing method by CRISPR/Cas9 in a fish cell line. Mar Biotechnol (NY) 18(4):449–452
Devoto SH, Melançon E, Eisen JS, Westerfield M (1996) Identification of separate slow and fast muscle precursor cells in vivo, prior to somite formation. Development 122(11):3371–3380
Du SJ, Devoto SH, Westerfield M, Moon RT (1997) Positive and negative regulation of muscle cell identity by members of the hedgehog and TGF-beta gene families. J Cell Biol 139(1):145–156
Du SJ, Li H, Bian Y, Zhong Y (2008) Heat-shock protein 90alpha1 is required for organized myofibril assembly in skeletal muscles of zebrafish embryos. Proc Natl Acad Sci U S A 105(2):554–559
Etard C, Behra M, Fischer N, Hutcheson D, Geisler R, Strähle U (2007) The UCS factor Steif/Unc-45b interacts with the heat shock protein Hsp90a during myofibrillogenesis. Dev Biol 308(1):133–143
Ferrante MI, Kiff RM, Goulding DA, Stemple DL (2011) Troponin T is essential for sarcomere assembly in zebrafish skeletal muscle. J Cell Sci 124(Pt 4):565–577
Gagnon JA, Valen E, Thyme SB, Huang P, Ahkmetova L, Pauli A, Montague TG, Zimmerman S, Richter C, Schier F (2014).Efficient Mutagenesis by Cas9 Protein-Mediated Oligonucleotide Insertion and Large-Scale Assessment of Single-Guide RNAs. PLoS One 9(5):e98186
Gao J, Li J, Li BJ, Yagil E, Zhang J, SJ D (2014) Expression and functional characterization of Smyd1a in myofibril organization of skeletal muscles. PLoS One 9(1):e86808
Gao F, Shen XZ, Jiang F, Wu Y, Han C (2016) DNA-guided genome editing using the Natronobacterium gregoryi Argonaute. Nat Biotechnol 34(7):768–773
Goody MF, Carter EV, Kilroy EA, Maves L, Henry CA (2017) “Muscling” throughout life: integrating studies of muscle development, homeostasis, and disease in zebrafish. Curr Top Dev Biol 124:197–234
Gurevich D, Siegel A, Currie PD (2015) Skeletal myogenesis in the zebrafish and its implications for muscle disease modelling. Results Probl Cell Differ 56:49–76
Gurung R, Ono Y, Baxendale S, Lee SL, Moore S, Calvert M, Ingham PW (2017) A zebrafish model for a human myopathy associated with mutation of the unconventional myosin MYO18B. Genetics 205(2):725–735
Hwang PM, Sykes BD (2015) Targeting the sarcomere to correct muscle function. Nat Rev Drug Discov 14(5):313–328
Hwang WY, Fu Y, Reyon D, Maeder ML, Tsai SQ, Sander JD, Peterson RT, Yeh JR, Joung JK (2013) Efficient genome editing in zebrafish using a CRISPR-Cas system. Nat Biotechnol 31(3):227–229
Hawkins TA, Haramis AP, Etard C, Prodromou C, Vaughan CK, Ashworth R, Ray S, Behra M, Holder N, Talbot WS, Pearl LH, Strähle U, Wilson SW (2008) The ATPase-dependent chaperoning activity of Hsp90a regulates thick filament formation and integration during skeletal muscle myofibrillogenesis. Development 135(6):1147–1156
Hegge JW, Swarts DC, van der Oost J (2018) Prokaryotic Argonaute proteins: novel genome-editing tools? Nat Rev Microbiol 16(1):5–11
Hinits Y, Hughes SM (2007) Mef2s are required for thick filament formation in nascent muscle fibres. Development 134(13):2511–2519
Höck J, Meister G (2008) The Argonaute protein family. Genome Biol 9(2):210
Irion U, Krauss J, Nüsslein-Volhard C (2014) Precise and efficient genome editing in zebrafish using the CRISPR/Cas9 system. Development 141(24):4827–4830
Jackson HE, Ingham PW (2013) Control of muscle fibre-type diversity during embryonic development: the zebrafish paradigm. Mech Dev 130(9-10):447–457
Jao LE, Wente SR, Chen W (2013) Efficient multiplex biallelic zebrafish genome editing using a CRISPR nuclease system. Proc Natl Acad Sci U S A 110(34):13904–13909
Jiang W, Bikard D, Cox D, Zhang F, Marraffini LA (2013) RNA-guided editing of bacterial genomes using CRISPR-Cas systems. Nat Biotechnol 31(3):233–239
Just S, Meder B, Berger IM, Etard C, Trano N, Patzel E, Hassel D, Marquart S, Dahme T, Vogel B, Fishman MC, Katus HA, Strähle U, Rottbauer W (2011) The myosin-interacting protein SMYD1 is essential for sarcomere organization. J Cell Sci 124(Pt 18):3127–3136
Kang X, He W, Huang Y, Yu Q, Chen Y, Gao X, Sun X, Fan Y (2016) Introducing precise genetic modifications into human 3PN embryos by CRISPR/Cas-mediated genome editing. J Assist Reprod Genet 33(5):581–588
Ketting RF (2011) The many faces of RNAi. Dev Cell 20(2):148–161
Khin NC, Lowe JL, Jensen LM, Burgio G (2017). No evidence for genome editing in mouse zygotes and K293T human cell line using the DNA-guided Natronobacterium gregoryi Argonaute (NgAgo). PLoS One 12(6):e0178768
Kim S, Kim D, Cho SW, Kim J, Kim JS (2014) Highly efficient RNA-guided genome editing in human cells via delivery of purified Cas9 ribonucleoproteins. Genome Res 24(6):1012–1019
Kotani H, Taimatsu K, Ohga R, Ota S, Kawahara A (2015) Efficient multiple genome modifications induced by the crRNAs, tracrRNA and Cas9 protein complex in zebrafish. PLoS One 10(5):e0128319
Laing NG (ed) (2008) The sarcomere and skeletal muscle disease. Advances in experimental medicine and biology, vol 642. Springer-Verlag New York, New York
Lee SH, Turchiano G, Ata H, Nowsheen S, Romito M, Lou Z, Ryu SM, Ekker SC, Cathomen T, Kim JS (2016) Failure to detect DNA-guided genome editing using Natronobacterium gregoryi Argonaute. Nat Biotechnol 35(1):17–18
Li H, Zhong Y, Wang Z, Gao J, Xu J, Chu W, Zhang J, Fang S, Du SJ (2013) Smyd1b is required for skeletal and cardiac muscle function in zebrafish. Mol Biol Cell 24(22):3511–3521
Li M, Hromowyk KJ, Amacher SL, Currie PD (2017) Muscular dystrophy modeling in zebrafish. Methods Cell Biol 138:347–380
Liang X, Potter J, Kumar S, Zou Y, Quintanilla R, Sridharan M, Carte J, Chen W, Roark N, Ranganathan S, Ravinder N, Chesnut JD (2015) Rapid and highly efficient cell engineering via Cas9 protein transfection. J Biotechnol 208:44–53
Lieschke GJ, Currie PD (2007) Animal models of human disease: zebrafish swim into view. Nat Rev Genet 8(5):353–367
Mali P, Yang L, Esvelt KM, Aach J, Guell M, DiCarlo JE, Norville JE, Church GM (2013) RNA-guided human genome engineering via Cas9. Science 339(6121):823–826
Myhre JL, Hills JA, Prill K, Wohlgemuth SL, Pilgrim DB (2014) The titin A-band rod domain is dispensable for initial thick filament assembly in zebrafish. Dev Biol 387(1):93–108
Ota S, Hisano Y, Ikawa Y, Kawahara A (2014) Multiple genome modifications by the CRISPR/Cas9 system in zebrafish. Genes Cells 19(7):555–564
Prill K, Windsor Reid P, Wohlgemuth SL, Pilgrim DB (2015) Still heart encodes a structural HMT, SMYD1b, with chaperone-like function during fast muscle sarcomere assembly. PLoS One 10(11):e0142528
Qi J, Dong Z, Shi Y, Wang X, Qin Y, Wang Y, Liu D (2016) NgAgo-based fabp11a gene knockdown causes eye developmental defects in zebrafish. Cell Res 26(12):1349–1352
Sanger JW, Chowrashi P, Shaner NC, Spalthoff S, Wang J, Freeman NL, Sanger JM (2002) Myofibrillogenesis in skeletal muscle cells. Clin Orthop Relat Res 403(Suppl):S153–S162
Sanger JW, Wang J, Holloway B, Du A, Sanger JM (2009) Myofibrillogenesis in skeletal muscle cells in zebrafish. Cell Motil Cytoskeleton 66(8):556–566
Sanger JW, Wang J, Fan Y, White J, Sanger JM (2010) Assembly and dynamics of myofibrils. J Biomed Biotechnol 2010(858606):1–8
Swarts DC, Jore MM, Westra ER, Zhu Y, Janssen JH, Snijders AP, Wang Y, Patel DJ, Berenguer J, Brouns SJJ, van der Oost J (2014a) DNA-guided DNA interference by a prokaryotic Argonaute. Nature 507(7491):258–261
Swarts DC, Makarova K, Wang Y, Nakanishi K, Ketting RF, Koonin EV, Patel DJ, van der Oost J (2014b) The evolutionary journey of Argonaute proteins. Nat Struct Mol Biol 21(9):743–753
Swarts DC, Hegge JW, Hinojo I, Shiimori M, Ellis MA, Dumrongkulraksa J, Terns RM, Terns MP, van der Oost J (2015) Argonaute of the archaeon Pyrococcus furiosus is a DNA-guided nuclease that targets cognate DNA. Nucleic Acids Res 43(10):5120–5129
Tan X, Rotllant J, Li H, De Deyne P, SJ D (2006) SmyD1, a histone methyltransferase, is required for myofibril organization and muscle contraction in zebrafish embryos. Proc Natl Acad Sci U S A 103(8):2713–2718
Willkomm S, Oellig CA, Zander A, Restle T, Keegan R, Grohmann D, Schneider S (2017) Structural and mechanistic insights into an archaeal DNA-guided Argonaute protein. Nat Microbiol 2:17035
Xu J, Gao J, Li J, Xue L, Clark KJ, Ekker SC, Du SJ (2012) Functional analysis of slow myosin heavy chain 1 and myomesin-3 in sarcomere organization in zebrafish embryonic slow muscles. J Genet Genomics 39(2):69–80
Zuris JA, Thompson DB, Shu Y, Guilinger JP, Bessen JL, Hu JH, Maeder ML, Joung JK, Chen ZY, Liu DR (2015) Cationic lipid-mediated delivery of proteins enables efficient protein-based genome editing in vitro and in vivo. Nat Biotechnol 33(1):73–80
Acknowledgments
This research was supported by a seed fund from the University of Maryland Baltimore and Chinese National Science Foundation (31230076). MC and YFS were supported by China Scholarship Council.
Author information
Authors and Affiliations
Contributions
Conceived and designed the experiments: MC and SD. Performed the experiments: MC, YFS, and SD. Analyzed the data: MC, JZ, ZT, and SD. Wrote the paper: MC, JZ, and SD.
Corresponding authors
Ethics declarations
All animal studies were carried out according to the guideline for the Care and Use of Laboratory Animals of the National Institutes of Health. The protocol was approved by the Institutional Animal Care and Use Committee of the University of Maryland (Permit Number 0516005).
Rights and permissions
About this article
Cite this article
Cai, M., Si, Y., Zhang, J. et al. Zebrafish Embryonic Slow Muscle Is a Rapid System for Genetic Analysis of Sarcomere Organization by CRISPR/Cas9, but Not NgAgo. Mar Biotechnol 20, 168–181 (2018). https://doi.org/10.1007/s10126-018-9794-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10126-018-9794-8