Abstract
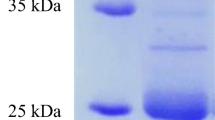
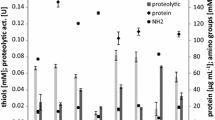
Black chicken feathers generated in large amount from poultry and slaughter houses are highly recalcitrant to microbial degradation due to their tough structural nature. A novel keratinolytic bacterium that possessed high affinity for black feather was isolated from chicken manure and identified as Pseudochrobactrum sp. IY-BUK1. Keratinase and feather soluble protein were effectively produced by the free living cells of the bacterium in media containing only black feathers and a mixture of equal amount of black-, brown- and white-coloured feathers. Complete degradation of 5 g/L of black feathers was completed in 3 days following optimisation of physico-chemical conditions. However, the bacterium selectively completed the degradation of black feather in a medium containing mixture of feathers in 144 h leaving behind approximately 33% and 45% of brown and white feathers in the medium respectively. Gellan gum-immobilised cells of strain IY-BUK1 enhanced the keratinase production by about 150% and were used repeatedly for ten cycles to degrade 5 g/L of black feather in a semi continuous fermentation of 18 h per cycle with enhanced and stable production of soluble protein. The study demonstrated the potential use of Pseudochrobactrum sp. IY-BUK1 not only in biodegradation of highly recalcitrant black feathers, but also in producing keratinase enzymes and valuable soluble proteins for possible industrial usage.







Similar content being viewed by others
References
Adinarayana K, Jyothi B, Ellaiah P (2005) Production of alkaline protease with immobilized cells of Bacillus subtilis PE-11 in various matrices by entrapment technique. AAPS PharmSciTech 6:E391–E397. https://doi.org/10.1208/pt060348
Ahmad S, Shamaan N, Arif N (2012) Enhanced phenol degradation by immobilized Acinetobacter sp. strain AQ5NOL 1. World J Microbiol Biotechnol 28:347–352
Barrowclough G, Sibley F (1980) Feather pigmentation and abrasion: test of a hypothesis. Auk 881–883
Bhange K, Chaturvedi V, Bhatt R (2016) Feather degradation potential of Stenotrophomonas maltophilia KB13 and feather protein hydrolysate (FPH) mediated reduction of hexavalent chromium. 3 Biotech 6:42. https://doi.org/10.1007/s13205-016-0370-5
Bhari R, Kaur M, Singh RS, Pandey A, Larroche C (2018) Bioconversion of chicken feathers by Bacillus aerius NSMk2: a potential approach in poultry waste management. Bioresour Technol Rep 3:224–230. https://doi.org/10.1016/J.BITEB.2018.07.015
Brenner DJ, Krieg N, Garrity G, Staley J (2005) The proteobacteria, part B: the gammaproteobacteria. In: Bergey’s Manual of Systematic Bacteriology, vol 2. Springer, p 323e376
Burtt EH Jr, Schroeder MR, Smith LA, Sroka JE, McGraw KJ (2011) Colourful parrot feathers resist bacterial degradation. Biol Lett 7:214–216. https://doi.org/10.1098/rsbl.2010.0716
Butler M (2004) Are melanized feather barbs stronger? J Exp Biol 207:285–293. https://doi.org/10.1242/jeb.00746
Călin M, Constantinescu-Aruxandei D, Alexandrescu E, Răut I, Doni MB, Arsene ML, Oancea F, Jecu L, Lazăr V (2017) Degradation of keratin substrates by keratinolytic fungi. Electron J Biotechnol 28:101–112. https://doi.org/10.1016/J.EJBT.2017.05.007
Cao ZJ, Zhang Q, Wei D-K, Chen L, Wang J, Zhang XQ, Zhou MH (2009) Characterization of a novel Stenotrophomonas isolate with high keratinase activity and purification of the enzyme. J Ind Microbiol Biotechnol 36:181–188. https://doi.org/10.1007/s10295-008-0469-8
Cappuccino J.G. Sherman N (1996) Microbiology-A laboratory manual (Vol. 9). Benjamin Cummings Science Publishing, Carlifonia. p 471
Chaturvedi V, Bhange K, Bhatt R, Verma P (2014) Production of kertinases using chicken feathers as substrate by a novel multifunctional strain of Pseudomonas stutzeri and its dehairing application. Biocatal Agric Biotechnol 3:167–174. https://doi.org/10.1016/j.bcab.2013.08.005
Chaudhari PN, Chaudhari BL, Chincholkar SB (2013) Iron containing keratinolytic metallo-protease produced by Chryseobacterium gleum. Process Biochem 48:144–151. https://doi.org/10.1016/j.procbio.2012.11.009
Corrêa APF, Daroit DJ, Brandelli A (2010) Characterization of a keratinase produced by Bacillus sp. P7 isolated from an Amazonian environment. Int Biodeterior Biodegradation 64:1–6. https://doi.org/10.1016/j.ibiod.2009.06.015
Dahloum L, Yakubu A, Halbouche M (2018) Effects of housing system and plumage colour on egg quality characteristics of indigenous naked-neck chickens. Livest Res Rural Dev 30:206 Retrieved from http://www.lrrd.org/lrrd30/12/abdul30206.html. Accessed 11 Apr 2019
de Oliveira CT, Pellenz L, Pereira JQ, Brandelli A, Daroit DJ (2016) Screening of bacteria for protease production and feather degradation. Waste Biomass Valoriz 7:447–453. https://doi.org/10.1007/s12649-015-9464-2
Ferrareze P, Correa A, Brandelli A (2016) Purification and characterization of a keratinolytic protease produced by probiotic Bacillus subtilis. Biocatal Agric Biotechnol 7:102–109
Gessesse A, Hatti-Kaul R, Gashe BA, Mattiasson B (2003) Novel alkaline proteases from alkaliphilic bacteria grown on chicken feather. Enzym Microb Technol 32:519–524. https://doi.org/10.1016/S0141-0229(02)00324-1
Ghaffar I, Imtiaz A, Hussain A, Javid A, Jabeen F, Akmal M, Qazi JI (2018) Microbial production and industrial applications of keratinases: an overview. Int Microbiol 21:163–174. https://doi.org/10.1007/s10123-018-0022-1
Goldstein G, Flory K, Browne B, Majid S (2004) Bacterial degradation of black and white feathers. Auk 121:656–659
Grande JM, Negro JJ, Torres MJ (2004) The evolution of bird plumage colouration: a role for feather-degradation bacteria? Ardeola 51:375–383
Gunderson A, Frame A (2008) Resistance of melanized feathers to bacterial degradation: is it really so black and white? J Avian Biol 39:539–545
Gurav RG, Tang J, Jadhav JP (2016) Sulfitolytic and keratinolytic potential of Chryseobacterium sp. RBT revealed hydrolysis of melanin containing feathers. 3 Biotech 6:145. https://doi.org/10.1007/s13205-016-0464-0
Jacob S, Colmas L, Parthuisot N, Heeb P (2014) Do feather-degrading bacteria actually degrade feather colour? No significant effects of plumage microbiome modifications on feather colouration in wild great tits. Naturwissenschaften 101:929–938. https://doi.org/10.1007/s00114-014-1234-7
Jeong J-H, Jeon Y-D, Lee OM, Kim JD, Lee NR, Park GT, Son HJ (2010a) Characterization of a multifunctional feather-degrading Bacillus subtilis isolated from forest soil. Biodegradation 21:1029–1040. https://doi.org/10.1007/s10532-010-9363-y
Jeong J-H, Oh D-J, Hwang D-Y, Kim H-S, Park K-H, Lee C-Y, Son H-J (2010b) Keratinolytic enzyme-mediated biodegradation of recalcitrant feather by a newly isolated Xanthomonas sp. P5. Polym Degrad Stab 95:1969–1977
Joshi S, Tejashwini M, Revati N (2007) Isolation, identification and characterization of a feather degrading bacterium. Int J Poult Sci 6:689–693
Justyn NM, Peteya JA, D’Alba L, Shawkey MD (2017) Preferential attachment and colonization of the keratinolytic bacterium Bacillus licheniformis on black- and white-striped feathers. Auk 134:466–473. https://doi.org/10.1642/AUK-16-245.1
Korkmaz H, Hür H, Di S (2004) Characterization of alkaline keratinase of Bacillus licheniformis strain HK-1 from poultry waste. Ann Microbiol 54:201–211
Kshetri P, Roy SS, Sharma SK, Singh TS, Ansari MA, Prakash N, Ngachan SV (2017) Transforming chicken feather waste into feather protein hydrolysate using a newly isolated multifaceted keratinolytic bacterium Chryseobacterium sediminis RCM-SSR-7. Waste Biomass Valoriz 10:1–11. https://doi.org/10.1007/s12649-017-0037-4
Kumar EV, Vijay M, Srijana K, Chaitanya Y, Reddy HK, Reddy G (2011a) Biodegradation of poultry feathers by a novel bacterial isolate Bacillus altitudinis GVC11. Indian J Biotechnol 10:502–507
Kumar E, Srijana M, Kumar K (2011b) A novel serine alkaline protease from Bacillus altitudinis GVC11 and its application as a dehairing agent. Bioprocess Biosyst Eng 34:403–409
Łaba W, Żarowska B, Chorążyk D, Pudło A, Piegza M, Kancelista A, Kopeć W (2018) New keratinolytic bacteria in valorization of chicken feather waste. AMB Express 8:9. https://doi.org/10.1186/s13568-018-0538-y
Lasekan A, Abu F, Hashim D (2013) Potential of chicken by-products as sources of useful biological resources. Waste Manag 33:552–565. https://doi.org/10.1016/j.wasman.2012.08.001
Lowry OC, Rosebrough N (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265
Lv LX, Sim MH, Li YD, Min J, Feng WH, Guan WJ, Li YQ (2010) Production, characterization and application of a keratinase from Chryseobacterium L99 sp. nov. Process Biochem 45:1236–1244. https://doi.org/10.1016/j.procbio.2010.03.011
Mariyammal A, Ezhilarasu A, Karthy ES, Menaga D (2018) Purification and characterization of novel extracellular keratinase enzyme from poultry feather waste. Int J Curr Res Life Sci 7:1018–1024
McGraw K (2006) Mechanics of uncommon colors: pterins, porphyrins, and psittacofulvins. Bird Color 1:354–398
Moslemy P, Neufeld RJ, Millette D, Guiot SR (2003) Transport of gellan gum microbeads through sand: an experimental evaluation for encapsulated cell bioaugmentation. J Environ Manag 69:249–259. https://doi.org/10.1016/j.jenvman.2003.09.003
Nagarajan S, Eswaran P, Masilamani RP, Natarajan H (2017) Chicken feather compost to promote the plant growth activity by using keratinolytic bacteria. Waste Biomass Valoriz 9:531–538. https://doi.org/10.1007/s12649-017-0004-0
Ningthoujam SD, Tamreihao K, Mukherjee S, et al (2018) Keratinaceous wastes and their valorization through keratinolytic microorganisms in keratin. IntechOpen
Nwogu S (2017) Local farmers produce 30% of Nigeria’s poultry demand. Punch Newspaper, Published September 1, 2017. https://punchng.com/local-farmers-produce-30-of-nigerias-poultry-demand/. Accessed on 3/4/2019
Okoroma EA, Garelick H, Abiola OO, Purchase D (2012) Identification and characterisation of a Bacillus licheniformis strain with profound keratinase activity for degradation of melanised feather. Int Biodeterior Biodegradation 74:54–60. https://doi.org/10.1016/j.ibiod.2012.07.013
Peng Z, Zhang J, Du G, Chen J (2019) Keratin waste recycling based on microbial degradation: mechanisms and prospects. ACS Sustain Chem Eng 7:9727–9736. https://doi.org/10.1021/acssuschemeng.9b01527
Prakash P, Jayalakshmi SK, Sreeramulu K (2010) Production of keratinase by free and immobilized cells of Bacillus halodurans strain PPKS-2: partial characterization and its application in feather degradation and dehairing of the goat skin. Appl Biochem Biotechnol 160:1909–1920. https://doi.org/10.1007/s12010-009-8702-0
Ramarosandratana A, Harvengt L, Bouvet A, Calvayrac R, Pâques M (2001) Effects of carbohydrate source, polyethylene glycol and gellan gum concentration on embryonal-suspensor mass (ESM) proliferation and maturation of maritime pine somatic embryos. Vitr Cell Dev Biol Plant 37:29–34. https://doi.org/10.1007/s11627-001-0006-1
Ramnani P, Gupta R (2004) Optimization of medium composition for keratinase production on feather by Bacillus licheniformis RG1 using statistical methods involving response surface methodology. Biotechnol Appl Biochem 40:191–196. https://doi.org/10.1042/BA20030228
Ramnani P, Singh R, Gupta R (2005) Keratinolytic potential of Bacillus licheniformis RG1: structural and biochemical mechanism of feather degradation. Can J Microbiol 51:191–196. https://doi.org/10.1139/w04-123
Riffel A, Brandelli A (2006) Keratinolytic bacteria isolated from feather waste. Braz J Microbiol 37:395–399. https://doi.org/10.1590/S1517-83822006000300036
Sahoo DK, Das A, Thatoi H, Mondal KC, Mohapatra PKD (2012) Keratinase production and biodegradation of whole chicken feather keratin by a newly isolated bacterium under submerged fermentation. Appl Biochem Biotechnol 167:1040–1051. https://doi.org/10.1007/s12010-011-9527-1
Shrinivas D, Kumar R, Naik G (2012) Enhanced production of alkaline thermostable keratinolytic protease from calcium alginate immobilized cells of thermoalkalophilic Bacillus halodurans JB 99. J Ind Microbiol Biotechnol 39:93–98
Sun W, Griffiths MW (2000) Survival of bifidobacteria in yogurt and simulated gastric juice following immobilization in gellan–xanthan beads. Int J Food Microbiol 61:17–25. https://doi.org/10.1016/S0168-1605(00)00327-5
Tatineni R, Doddapaneni K, Potumarthi R, Mangamoori L (2007) Optimization of keratinase production and enzyme activity using response surface methodology with Streptomyces sp. 7. Appl Biochem Biotechnol 141:187–201. https://doi.org/10.1007/BF02729061
Tiwary E, Gupta R (2010) Medium optimization for a novel 58 kDa dimeric keratinase from Bacillus licheniformis ER-15: biochemical characterization and application in feather degradation and dehairing of hides. Bioresour Technol 101:6103–6110. https://doi.org/10.1016/j.biortech.2010.02.090
Tuna A, Okumuş Y, Çelebi H, Seyhan AT (2015) Thermochemical conversion of poultry chicken feather fibers of different colors into microporous fibers. J Anal Appl Pyrolysis 115:112–124. https://doi.org/10.1016/j.jaap.2015.07.008
Vuillemard J-C, Terré S, Benoit S, Amiot J (1988) Protease production by immobilized growing cells of Serratia marcescens and Myxococcus xanthus in calcium alginate gel beads. Appl Microbiol Biotechnol 27:423–431. https://doi.org/10.1007/BF00451607
Wang T, Liang C, Sun Y, Gao W, Luo X, Gao Q, Li R, Fu S, Xu H, He T, Yuan H (2018) Strategical isolation of efficient chicken feather–degrading bacterial strains from tea plantation soil sample. Int Microbiol 22:227–237. https://doi.org/10.1007/s10123-018-00042-4
Yusuf I, Shukor MY, Yee PL et al (2014) Biodegradation of chicken feather wastes in submerged fermentation containing high concentrations of heavy metals by Bacillus sp. khayat. J Environ Bioremed Toxicol 2:38–41
Yusuf I, Ahmad S, Phang L, Syed M (2016) Keratinase production and biodegradation of polluted secondary chicken feather wastes by a newly isolated multi heavy metal tolerant bacterium-Alcaligenes sp. J Environ Manag 183:182–195
Yusuf I, Aqlima S, Phang LY, Yasid NA, Shukor MY (2019) Effective production of keratinase by gellan gum-immobilised Alcaligenes sp . AQ05-001 using heavy metal-free and polluted feather wastes as substrates. 3 Biotech 9:04–12. https://doi.org/10.1007/s13205-018-1555-x
Zhang RX, Gong JS, Su C, Zhang DD, Tian H, Dou WF, Li H, Shi JS, Xu ZH (2016) Biochemical characterization of a novel surfactant-stable serine keratinase with no collagenase activity from Brevibacillus parabrevis CGMCC 10798. Int J Biol Macromol 93:843–851. https://doi.org/10.1016/j.ijbiomac.2016.09.063
Acknowledgements
The authors wish to acknowledge the heads of Microbiology, Biochemistry and Centre for Biotechnology research for providing the facilities to carry out the work.
Funding
The project was partially supported financially by Bayero University Kano, Nigeria.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yusuf, I., Garba, L., Shehu, M.A. et al. Selective biodegradation of recalcitrant black chicken feathers by a newly isolated thermotolerant bacterium Pseudochrobactrum sp. IY-BUK1 for enhanced production of keratinase and protein-rich hydrolysates. Int Microbiol 23, 189–200 (2020). https://doi.org/10.1007/s10123-019-00090-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10123-019-00090-4