Abstract
This article addresses energy flows in the coffee agro-ecosystems of Costa Rica within the context of the socio-ecological transition, between 1935 and 2010, accounting for the shift from traditional to modern tropical agriculture. Estimating indicators of energy efficiency in crop management makes it possible to analyze the changing productive rationality of growers by studying end uses of all biomass appropriated from coffee agro-ecosystems. Coffee land and labor productivity, as expected, multiplied (by factors of 2.01 and 1.56, respectively). However, considering total biomass produced in coffee agro-ecosystems, productivity did not display such a significant increase. In contrast, all other energy efficiency indicators declined. Final energy return on investment (FEROI) fell from 1.02 in 1935 to 0.51 in 2005. External final EROI fell even farther, from 18.90 down to 1.86. The socio-ecological transition brought about the loss of multifunctionality in the final use of products derived from coffee agro-ecosystems, as new products replaced traditional ones.
Similar content being viewed by others
Introduction
The study of energy flows in farming, understood in a broad sense, represents an effective and versatile way to describe the major changes that have taken place in agrarian systems during the transition from traditional to modern management approaches, as well as to analyze the environmental problems generated in this process. Scholars have used a variety of methods, from traditional energy balances (Leach 1976) to energy return on investment (EROI) (Tello et al. 2016) and life cycle assessment (LCA) (Haas et al. 2000) to analyze energy flows in farming. One aim, from the perspective of environmental and energy sustainability, has been to compare the energy efficiency of traditional and modern systems (Cleveland 1995; Guzmán et al. 2017) or organic vs. conventional management (e.g., Smith et al. 2015). Another approach, from the social sciences and humanities, uses energy flows to study the material foundations and dynamics by which traditional societies were sustained, endeavoring to overcome perspectives that focus exclusively on cultural or monetary concerns. Such studies have assessed hunter-gatherer societies (Rappaport 1969), the collapse of civilizations (Tainter 1990), and the subsistence of peasant farming societies (Infante-Amate 2014).
This article evaluates energy flows in Costa Rica’s coffee agro-ecosystems within the context of the socio-ecological transition (SET) and the “Green Revolution,” between 1935 and 2010, spanning both of the aforementioned perspectives within a tropical agricultural system. It estimates indicators about the energy processes of coffee production systems, evaluating its evolving energy efficiency. There is some published research about energy balances in coffee production in Nicaragua (Cuadra and Rydberg 2006), Brazil (Giannetti et al. 2011a, b; Turco et al. 2012; Flauzino et al. 2014; Muner et al. 2015), and Costa Rica (Marozzi et al. 2004; Mora-Delgado et al. 2006). However, these studies all refer to the present day and address the plantation scale, generally comparing the efficiency of organic vs. conventional management and excluding the crucial shade layer (SL) in coffee agro-forestry systems. The scale of this article is national, offering data for all coffee agro-ecosystems in Costa Rica with a long-term perspective. It also includes the shade layer. In addition to energy efficiency indicators, the analysis reconstructs indicators of both land and labor productivity to draw comparisons between them. It is the first such analysis of a tropical crop at the national scale from a long-term perspective and including the SL. The analysis is not only ecological but also socio-economic, discriminating the end uses of all final products obtained and not only the commercial ones, with a view to understanding better the growers’ productive rationality, the changes in management, and the changes in the functionality of the products and by-products of an agro-forestry crop during the socio-ecological transition (SET).
Materials, methods, and case study
Methods
Most studies of energy flows in agrarian systems aim simply to quantify the energy (direct and indirect) invested in an agro-ecosystem and to relate it with the energy produced by that agro-ecosystem. Divergent methods of determining study boundaries, conversion coefficients, and units of input and output mean that the results reported in the literature are incompatible, generating confusion that makes it hard to draw comparisons (Murphy et al. 2011; Aguilera et al. 2015).
Nevertheless, this analysis builds on recent publications that overcome such limitations by employing standardized protocols for the study of EROIs of agricultural systems. When estimating output, we used Guzmán et al. (2014), occasionally complemented by Rodríguez and Zambrano (2010), Benavides (1998), and Salazar (1985). When estimating input, we used Aguilera et al. (2015), which updates the embodied energy factors and adds an historical dimension to the calculations. With regard to boundaries, Tello et al. (2016) offer complex analysis about the combination of input and output flows and especially a nuanced set of possible indicators of energy efficiency. In this case, we have adapted their proposal to fit a national-level crop scale. Infante-Amate (2014) distinguishes between the different end uses in the output products, making it possible to characterize marketable and subsistence production to better understand how traditional societies met their needs. Based on these studies, this article estimates the energy flows detailed in Fig. 1a, summarized as follows: net primary production (NPP) is divided into final production (FP), which is the proportion of production that reaches society and, as a consequence, has a socio-economic use, plus biomass reused (BR), which is the part of the biomass produced that recirculates within the coffee agro-ecosystem. The part of FP that goes to other agro-ecosystems or to livestock outside the coffee agro-ecosystem is considered FP in this case, and not BR, since it is not used within the coffee plantation. Our NPP estimation is an aboveground estimation that does not include accumulated biomass (Guzmán and González de Molina 2015). Therefore, NPP equals to land produce or NPPh, as defined in Tello et al. (2016), plus that part of the herbaceous layer or the shade layer not harvested. In agro-forestry systems, recycling of the shade layer plays a key role in the circulation of energy, whether the leaves fall naturally or through human action. Finally, society provides external inputs (EI), which are those inputs that are not recycled. EI plus BR constitutes total inputs consumed (TIC).
In the case of coffee, NPP can be divided into three different layers: the coffee layer (or coffee plant) (CL), comprising the bean, leaves, and wood; the herbaceous layer (HL), comprising the small plants and grass; and the shade layer (SL), made up of various species of trees that perform a crucial ecological and socio-economical role in the production of many tropical agrarian systems, including coffee (Perfecto et al. 1996; Beer et al. 1997). This article reconstructs the production of the CL and the HL in annual series, between 1935 and 2010 (Fig. 1c). For the years 1935 and 2005 only, in an effort to characterize two clearly different management approaches—modern and traditional—it is possible to reliably reconstruct the production of the SL, bearing in mind that it integrates dozens of species with different productions and surface compositions each year (Muschler 1999). Accordingly, although this analysis reconstructs EI annually, only for 1935 and 2005 does it estimate TIC, since shade layer calculations include the recycled biomass of the forest. Figure 1b explains the different types of associations between input and output flows and the resulting EROIs, which are estimated for each historical period.
Sources
The first comprehensive Costa Rican coffee census that allows a full energy analysis was published in 1935 (IDC 1935). Full series of bean production and land area from similar official censuses continue through present, including supplementary information from FAO (2016). Taken together, they allow an estimation of the production of by-products (Rodríguez and Zambrano 2010), prunings (Romjin and Wildernik 1981), and the herbaceous layer (Romero 2006). Shade layer estimation is more complex, but we have estimated the shaded land area at two time points, representing traditional and modern systems, based on the dominant tree species. Inga (Inga), Poró (Erythrina poeppigiana), Musaceae (Musaceae), and timber-yielding species (chiefly the Eucalyptus) have always provided canopy covering to around 90% of the total shaded area (DGEC 1953; ICAFÉ various years, 2007); a literature review—chiefly derived from CATIE, the Tropical Agronomical Centre for Research and Teaching—provides the biomass production of each tree species, as well as their recycling on the farm and their final uses. In the case of the Poró tree, of particular note are the works of Montenegro (2005), Romero (2006), and Merlo (2007). In the case of the Inga tree, Jiménez and Martínez (1979) and Salazar and Palm (1987) are the most important. With regard to Musaceae, Farfán-Valencia (2005) provides a complete summary. For timber-yielding species, there is a complete study available (Detlefsen and Somarriba 2012) along with the frequently used estimations of Beer et al. (1997).
Fertilizers are the most significant inputs, usually reconstructed here from official statistics (CAFETICO 1992; ICAFE various years). Otherwise, they come from studies describing total regional fertilizer use connected to other sources of information about the share of fertilizers used in coffee plantations (ICAITI 1967; OAS 1970; López and Picado 2012; FAO 2016). Due to lack of specific information about pesticides, we reconstructed total pesticide use (Ramírez 2009; FAO 2016) from studies that provide the percentage of pesticides added per crop at the national level (Maltby 1980; Hilje et al. 1987). Information about labor and machinery comes from Picado (2000) and Renjifo (1992).
Costa Rica in the global coffee system
Coffee is one of the most widely consumed drinks in the world and one of the most important commodities in global commerce (Grigg 2002). Up to the eighteenth century, its production and consumption was confined to very specific parts of Africa and Asia (Clarence-Smith and Topik 2003). Since then, fired by the first global distribution, its production spreads to other parts of the world, and its consumption became increasingly popular, chiefly in the Americas and Europe, where it is now more popular than tea (Grigg 2002). Their coffee consumption (availability) is 1.2 kg of green beans per person per year (FAO 2016), equivalent to 2500 million cups per day (Dicum and Luttinger 1999).
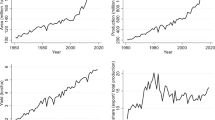
Figure 2a shows that the geographical expansion of coffee worldwide is confined to tropical climates. Currently, Brazil (2.78 teragrams, Tg) and Vietnam (1.16 Tg) account for over half of global production. Figure 2b also shows the main coffee-producing areas in the world: besides Brazil and Vietnam, Colombia, Ethiopia, and Central America. Together they accounted for 55–75% of global production in the last half century. In this period, production almost doubled, from 4.40 Tg in 1961–1965 to 8.38 Tg in 2008–2012. Central America encompasses five coffee-producing countries: Costa Rica, Honduras, Guatemala, El Salvador, and Nicaragua, which have accounted for a tenth of global coffee production over the past half century. The trend has been one of continued growth, with production figures rising from 341 Gg (gigagrams) to 792 Gg (Fig. 2c). Although its participation in the global total is not as striking as Brazil or Vietnam, Central America is a paradigmatic example of a coffee-producing region. Among the main producing areas in the world, Central America is the only one where coffee represents the countries’ primary crop,Footnote 1 and its percentage of cultivated area is the highest in the world, reaching an average of 30% (FAO 2016)
Coffee growing contextual indicators in Costa Rica and the world (Infante-Amate and Picado 2016)
Costa Rica was the region’s biggest coffee-producing country in the late nineteenth century and 20th century. It also led a process of industrial intensification from the 1950s onwards, at unprecedented levels on a global scale. In just three decades, Costa Rica followed an accelerated path of transition that altered the management and functionality of coffee agro-ecosystems; by the 1980s, it was the most productive country (land productivity) in the world (Renjifo 1992:34). However, Costa Rican coffee intensification stagnated since the 1990s, due to several factors: a drive to produce higher quality coffee, production regulations, abandonment of productive areas in the Central Valley due to urbanization, aging plantations, and, above all, falling coffee prices (Deugd 2003; Castro et al. 2004). Costa Rican coffee has not moved towards a later stage of hyper-intensification, as occurred in Vietnam, Brazil, and Colombia, characterized by total mechanization of management and harvesting and with unprecedented levels of production never before seen in history (e.g., Agergaard et al. 2009).
Energy flow analysis
Intensifying agro-ecosystems
Figure 3a, b details the energy flows (direct and embodied) in Costa Rican coffee production between 1935 and 2010. Of particular note is fertilization, which accounted for 63 to 90% of total energy requirements. Although prior to 1950, organic fertilizers represented 30 to 50% of fertilizer energy, today this figure stands at just 10%. Chemical fertilization was present to only a limited extent in 1935; just 5.1% of coffee plantations fertilized using external inputs, of which just 2.2% were chemical fertilizers. Today, these percentages have risen to 90 and 89, respectively.
Human labor was initially the second most important item; between 1935 and 1955, it accounted for between 20 to 35% of energy inputs, with a downward trend that reached on 5 to 10% in recent years. The mechanization of coffee production is still very limited in Costa Rica today. Farmers use only small machinery for manual applications of treatments, fertilizers, and weed management. For that reason, the growth of this input has been more limited. Costa Rica has not moved towards hyper-intensive methods as in countries such as Vietnam (Agergaard et al. 2009).
Since the 1960s, the second most important energy input has been pesticides, with an average consumption of around 12%. The use of pesticides was first documented in 1916 with the use of inorganic mixtures (Hilje et al. 1987); however, it was not until the 1950s that their usage became significant. Nonetheless, coffee has never been a crop treated with high doses of chemicals compared to other crops. In the 1950s, their usage must have been limited (Hilje et al. 1987:76). Between 1970 and 2000, different estimations indicate that coffee consumed only between 5 and 10% of all pesticides in the country, in spite of the fact that coffee was its most prominent crop. By way of example, in 1993, bananas consumed 57% of total pesticides whereas coffee, grown over a larger area, consumed just 7% (Santos et al. 1997:18). Today, over half of coffee plantations still manage weeds manually (ICAFE 2007).
External energy inputs to coffee plantations increased by a factor of 13 between 1935 and 2010. The trajectory of growth has been almost continuous, with the exception of a brief period in 1988–1993, and recent disruptions in the twenty-first century. There was also a switch to more productive varieties—chiefly Caturra—with a higher harvest index; these new varieties produce more bean than traditional ones in relation to total biomass, but also require more inputs, which indirectly alters energy flows (Infante-Amate and Picado 2016).
These changes led to a formidable increase in production (Fig. 3c, d). Between 1935 and 2010, coffee bean production increased from 117 Gg (263.8 TJ) to 500 Gg (1131.5 TJ), peaking in 1989 at 948 Gg (2144.6 TJ). Prior to 1955, production remained stable under traditional management, but in just 30 years, between 1955 and 1985, it underwent a major expansion then sustained until 2000, which marked a turning point towards crisis. The trend for bean production was similar to that for inputs from the perspective of energy, as shown in Fig. 3aa, c, following a pattern of rapid growth between 1955 and 1985, subsequent stagnation and final decline into the current crisis. However, the rate of growth in inputs was even greater, as we will see below.
The literature describes the change in production brought about by the SET in terms of the production of commercial beans—undoubtedly the most important part in monetary terms—but has neglected the production of total biomass derived from pruning or shade trees, which plays a decisive role in farming subsistence (Infante-Amate 2014) and crucial ecosystem functions (Smil 1999). Analyzing the rest of the biomass reveals that its increase has been much less than the increase in coffee beans. This could partly be explained with reference to the harvest index, which increased from 0.44 in 1935 to 0.64 in 2010 (comparing bean production with that of the total coffee plant biomass). If instead we compare the percentage of green coffee to the total aboveground NPP measured in terms of dry matter, we see that it increased from 4.6 to 10.9%. Thus, while bean productivity increased dramatically, total biomass productivity remained relatively stable.
The shade factor
Coffee is usually managed as an agro-forestry system in which the coffee plant grows among dozens of different species of trees that play a fundamental role in the agro-ecosystem’s energy cycle by producing and recycling biomass (e.g., Muschler 1999). During the socio-ecological transition, coffee agro-forestry systems have lost space in a process observed with great concern because of its environmental implications (Perfecto et al. 1996; Tschartnke et al. 2011). In Costa Rica, from almost 100% prior to industrialization, 75% of all land and 89% of coffee plantations are now shaded. The change in the tree species composition used to provide shade coffee is also significant. In 1950, shade trees planted in Costa Rica’s coffee plantations were made up of 62% Inga and 18% Musaceae, while timber-yielding species were insignificant (DGEC 1953). By 2005, Inga had fallen to 18%, having been replaced by Poró at 25% and with a growing presence of timber-yielding species (particularly Eucalyptus and laurel), now accounting for 11% (ICAFE 2007). The reasons behind this change are related to the nature of the SET, as we will see below.
In any case, occupation data highlight the major role of the SL in both traditional and modern coffee agro-ecosystems in the country. Biomass production from all sources, on average, ranges between 2.5 and 10 Mg of dry matter per hectare, including firewood, prunings, and beans. The majority of this production is recycled on the plantation (leaves and twigs that fall spontaneously or which are pruned and then reused). In 2005, the aboveground NPP of coffee agro-ecosystems was 974.8 Tg of dry matter, of which 414.9 Tg (42.6%) corresponded to the SL, and of these, 288.2 Tg was recycled. This amount represents an average of 43.9 GJ/ha/year of recycled biomass derived from the SL in relation to total inputs consumed of 120.6 GJ/ha (Fig. 4). In other words, even under industrial management, a third of the energy incorporated into a coffee agro-ecosystem came from recycling of the shade layer. In 1935, this figure was similar: 30.5 GJ/ha out of TIC of 73.6 GJ/ha. The fact that the amount recycled in absolute terms has increased is explained by the greater extraction in traditional systems of firewood collected outside of the plantation from dominant species such as the Inga, a firewood-producing tree, and the greater relative productivity of the Poró, which has grown with industrialization. Today, EI derived from external fertilization, pesticides, labor, and machinery stands at 33.2 GJ/ha, whereas recycled biomass is over twice that amount. Consequently, to analyze energy (or nutrient or carbon) cycles without taking into account the shade layer would be to neglect a dominant element.
Functional change
Figure 4 shows the input and output flows for traditional and modern coffee agro-ecosystems in Costa Rica. The output side is separated into flows consumed directly by families, which have fallen from 68.3 to 57.1 GJ/ha, and those that go to the market, which have multiplied by 6, from 0.7 to 4.5 GJ/ha. This change describes the commercialization of economic activities over the course of the twentieth century, but also the functional change operating in farming during the SET. This change reveals the transition from an agrarian system that needed to provide multiple goods to meet most human needs to a much more simplified industrial system in which farming focuses chiefly on the production of fruits, grains, or beans destined for human and animal consumption (e.g., González de Molina and Toledo 2014). This fact is more clearly evident when the energy analysis incorporates the SL as a crucial productive component of coffee agro-ecosystems.
Four major changes characterize this transition. First, there was an increase in coffee production for commercial purposes (chiefly export), which rose from 5.7 to 15.1 GJ/ha. Second, fuel use in final production declined, from 65.6 to 50.6 GJ/ha. The energy transition made society less reliant on prunings and coffee plant by-products as fuel. Up to the 1980s, over 80% of coffee plantation owners used firewood to meet their energy needs and the majority came from coffee products (both the SL and the CL) (Lemckert and Campos 1981). Third, the weighting of products allocated to livestock has fallen, from 6.5 to 5.3 GJ/ha, in a phenomenon explained by the growing consumption of imported commercial feed in the diets of livestock. Lastly, the production of timber for industrial uses has increased from 0.7 to 4.4 GJ/ha, owing to the proliferation of timber-yielding species in the SL and a refocusing of farming orientations for greater profitability (Detlefsen and Somarriba 2012).
Efficiency and productivity
The information compiled in this paper, summarized in Table 1, establishes a number of relationships to analyze various indicators of productivity and efficiency. Annual data, as shown in Fig. 1, present coffee production, EI, and the EROIs derived therefrom. This estimation also makes it possible to offer information about the productivity of land and labor in coffee plantations, in terms of beans and also the plant and the herbaceous layer. Figure 5 sets out indicators of energy efficiency, and land and labor productivity for coffee beans and NPP (excluding the SL), in an annual series from 1935 to 2010. Only for 1935 and 2005, in addition to the above information, it is possible to estimate TIC and FP and to obtain all of the EROIs proposed.
Land productivity doubled if one only analyzes the production of marketable coffee (bean or green), but it was stable in the context of the production of total agro-ecosystem biomass. The same is true for labor productivity. In 1935, these plantations produced 0.61 kg dry matter of green coffee beans per hour worked, whereas in 2005, this figure increased to 1.04 kg/h. In other words, labor productivity increased by 71%. However, instead of measuring coffee bean production, we analyze coffee plant production (adding by-products into the equation); productivity declined 33%, from 4.17 to 2.79 kg/h. Considering the total NPP, the labor productivity declined from 13.5 to 9.6 kg/h, a decline of 29%. So, labor productivity increased if we analyze only the coffee bean production, but by including by-products, it declined. Of these by-products, some do not have a socio-economic use but rather were reused within the plantation. Consider final produce as represented in Fig. 4. For every hour worked in 1935, the traditional agro-ecosystem produced 7.8 kg/ha of goods useful within the domestic unit, including food, fuel, fodder, and wood. The modern system in 2005 produced only 3.1 kg/ha of such materials. In terms of offering subsistence to rural farming communities, labor productivity decreased in an evolution similar to that described by Boserup (1965, 1981) for processes of intensification in traditional systems.
From the perspective of energy, the relationship between labor and production (in terms of beans or NPP) reveals the relationship between production and only one input, labor. Adding the other external inputs, for which there are annual series from 1935 to 2010, or total inputs consumed, which adds reused biomass, it is possible to calculate several EROIs. Each presents a different indicator of energy efficiency in coffee agro-ecosystems.
Table 1 details six EROIs derived from the proposals of Tello et al. (2016) and described in the methodological section, as well as in other articles included in this special issue. In all cases, energy efficiency declined. In contrast with the trends in labor and land productivity, the energy efficiency measures suggest a degradation in all cases. Final EROI is the most common metric of energy return on investment, relating final produce with total inputs consumed. FEROI fell from 1.02 in 1935 to 0.51 in 2005, revealing deterioration from a barely efficient system (above 1.0) to an inefficient one. These low values are partly explained by the fact that the majority of FP biomass comes from coffee beans, which has little energetic value. (Coffee beans are not particularly valuable in terms of their energy content or their nutritional or calorific functions, but primarily for their flavor and caffeine content.). Relating NPP with TIC produces NPP EROI, which declined from 1.97 to 1.24. This decline was less marked owing to the persistence of shaded biomass and by-products from the coffee plant, combined with a notable increase in bean production. Even so, total growth of NPP was less than the increase in inputs invested.
The external final EROI highlights the dependence on inputs from outside the agro-ecosystem (EI). The deterioration in efficiency in EFEROI was much more pronounced, plunging from 18.90 to 1.86, underscoring the tremendous dependence of the agro-ecosystem today on inputs from outside the region, and its high productivity in relation to the low level of inorganic inputs in traditional systems. Internal final EROI, which links final produce with biomass reused, fell from 1.08 to 0.71. This indicator is of great agro-ecological interest: a traditional system provided 1.08 energy units for every unit recycled, regardless of EI. However, today, it is inefficient with regard to recycled energy. Current intensive models produce less final biomass more than they recycle.
Discussions and conclusions
This article reconstructs the energy flows of coffee agro-ecosystems in Costa Rica during the socio-ecological transition. It assesses energy efficiency in depth, but also distinguishes the final uses of the entire energy production of coffee agro-ecosystems, not only the marketable coffee beans. A substantial proportion of the broader energy production of the system plays a key role in the everyday subsistence of local communities. These findings point to several conclusions:
-
1.
The intensification of coffee agro-ecosystems, from a biophysical perspective, can be explained as a series of interconnected factors. New coffee varieties were more productive but also required external inputs. The increase in production raised labor demands (more energy for human work). In energy terms, the majority of energy inputs came through chemical fertilizers. This is due to the limited use—in comparison with other tropical crops—of pesticides and the low level of mechanization on coffee plantations in Costa Rica. Intensification accelerated between 1955 and 1985, generating a growing dependence on external, non-renewable inputs, highly dependent on fossil fuels for their manufacture, transportation, and use. By 1985, due to coffee’s global economic crisis, total bean production declined, partly because of a reduction in external inputs. To some extent, the economic crisis forced more efficient practices. Although direct energy (electricity or fossil fuel) was not important, embodied energy shot up, as it has done in other countries in the same period (Cleveland 1995; Guzmán et al. 2017). Coffee is thus highly dependent on fossil fuels due to embodied energy consumption. When we drink coffee, we are also drinking oil.
-
2.
Intensification, as in other countries that have undergone the Green Revolution, has led to a productive increase in harvest yield. Figure 5 presents the evolution of land productivity, labor productivity, and energy efficiency, comparing the production of beans on the one hand and that of NPP on the other.Footnote 2 Land productivity quadrupled. However, one of the main conclusions of this study is that land productivity, when analyzing total biomass, did not increase as much (it has doubled). The same is true of labor productivity, which increased with regard to harvested fruit, but not for the rest of production (with or without a socio-economic use). Including the shade layer in the estimate of NPP (only available in 1935 and 2005), this pattern is even sharper. Land productivity remained stable and the decline in labor productivity was even higher than in the annual series of Fig. 5. These results are particularly interesting in relation to Boserupian debates about the evolution of productivity in traditional systems (Boserup 1965, 1981). According to the influential theory of this Danish economist, agrarian intensification under traditional management led to an increase in land productivity, but a decrease in labor productivity. The socio-ecological transition, on the other hand, brought about an increase in both land and labor productivity, as is frequently claimed in relation to the Green Revolution. The results set out here corroborate this known pattern. However, they also bring to light new evidence about a less well-known phenomenon, which is of increasing interest. The production of biomass per land unit did not grow during the SET and this fact has had several negative effects: decreased capacity as a carbon sink in the fight against climate change Aguilera et al. (2017), less biomass available for other species and a consequent loss of biodiversity (Haberl et al. 2004), and less potential for alternative uses of biomass such as bio-energy (Haberl et al. 2010).
-
3.
Examining this process purely in terms of energy, the efficiency of coffee growing declined substantially in the process of industrialization. Coffee does not contain much energy in terms of the human diet, but it does require increasing amounts of energy in its production. Internal cycles of energy are fundamental to sustaining production, biodiversity, and soil health, and coffee plantations offer broad bio-energy potential from coffee by-products or material derived from the shade layer. Hence, it is fundamental to study plantation energy flows in order to design sustainable coffee growing systems. Intensification of production clearly brought about energy inefficiencies, although it is also true that these inefficiencies raised coffee bean production. A literature review (Infante-Amate and Picado 2016) documents many studies that provide information about energy balances in coffee systems, exposing extremes between fairly non-intensive family-run organic plantations (1.5 GJ/ha of EI) and very intensive approaches in Brazil (943 GJ/ha). The EROIs of these studies show greater efficiency in the case of conventional management, generally less than one; greater efficiency in mixed management with few external inputs, with an average EROI of 1.94; and, finally, greater efficiency in organic systems, with an average of 3.41. These data are not broadly representative and refer only to the relationship between coffee produced and external inputs. However, they highlight a trend repeated throughout the literature on agricultural energy returns of greater efficiency in organic or traditional systems (Smith et al. 2015) and also help to contextualize these results.
-
4.
The SET did not only affect agrarian systems in terms of their intensification. Coffee is a classic cash crop; only the bean is used socially, roasted to make the beverage we drink. Focusing solely on those elements, although they are undoubtedly the most lucrative part of this agrarian activity, ignores a major component of the daily subsistence of producing regions and the biophysical flows that sustain not only agriculture but also life in a broader sense. Coffee, beyond just producing a bean that ends up as a steaming hot beverage, has been a formidable supplier of goods and services for communities of growers and the surrounding ecosystems. In traditional systems, prior to the energy transition, it was the touchstone to satisfy many other needs through materials pruned from coffee plants and especially from shade layer trees that were used as fuel and as suppliers of wood and timber for domestic usage, animal feed, packaging, etc. The SET brought about the loss of multifunctionality as new products replaced traditional ones. The arrival of electricity puts a stop to the use of fuels. Imported commercial feed replaced animal feed produced on plantations. New packaging materials replaced musaceae leaves. This study of traditional systems does not propose a return to past practices, but suggests we can learn from traditional cultures in order to design sustainable futures (Toledo and Barrera-Bassols 2008). In the new context of post-carbon economies, it is becoming important to foster a multifunctional approach to farming in order to combine internal recycling with the extractions required to meet non-food requirements. We are returning to situations we have experienced in the past: a world without oil.
The intensification of tropical farming and the loss of traditional agro-forestry systems are a grave environmental problem in the world today, causing the loss of biodiversity and ecosystem services (Foley et al. 2005). In coffee production, this socio-ecological transition was extremely serious in environmental terms (Tscharntke et al. 2011). In the case of coffee growing in Costa Rica, this transition has been more moderate and concluded already in the 1980s, falling short of the hyper-intensive experiences of other countries. However, between 1950 and 1985, the traditional production system moved towards one that was dependent on external inputs, losing part of its previous multifunctionality and focusing on increased coffee yields as it transitioned to the productive system present today.
Notes
In Costa Rica, coffee covered more surface area than any other crop in 2010. In Honduras, El Salvador, and Guatemala, it was second after maize. In Nicaragua, coffee was third, after maize and beans (FAO 2016).
Note that in this case, shade production is not included since it is not available for annual series.
References
Agergaard J, Fold N, Gough KV (2009) Global–local interactions: socioeconomic and spatial dynamics in Vietnam’s coffee frontier. Geogr J 175(2):133–145. https://doi.org/10.1111/j.1475-4959.2009.00320.x
Aguilera E, Guzmán G, Infante-Amate J, Soto D, García-Ruiz R, Herrera A, Villa I, Torremocha E, Carranza G, de González MM (2015) Embodied energy in agricultural inputs. Incorporating a historical perspective. DT-SEHA 15-07. Documentos de Trabajo de la Sociedad Española de Historia Agraria
Aguilera E, Guzmán G I, Álvaro-Fuentes J, Infante-Amate J, García-Ruiz R, Carranza-Gallego G, Soto, D, Gonzalez de Molina M (2017) A historical perspective on soil organic carbon in Mediterranean cropland (Spain, 1900–2008). Science of The Total Environment 621: 634–648. https://doi.org/10.1016/j.scitotenv.2017.11.243
Beer J, Muschler R, Kass D, Somarriba E (1997) Shade management in coffee and cacao plantations. Agrofor Syst 38(1):139–164. https://doi.org/10.1023/A:1005956528316
Benavides JE (1998) Árboles y arbustos forrajeros: una alternativa agroforestal para la ganadería. In: Agroforestería para la Producción Animal en Latinoamérica. FAO, Roma, pp 367–394
Boserup E (1965) The conditions of agricultural growth: the economics of agrarian change under population pressure. Allen and Unwin, London
Boserup E (1981) Population and technological change: a study of long-term trends. Chicago University Press, Chicago
CAFETICO (1992) Cámara Nacional de Cafetaleros. Cafetico, Costa Rica
Castro F, Montes E, Raine M (2004) Centroamérica. La crisis cafetalera: efectos y estrategias para hacerle frente. FAO, Roma
Clarence-Smith WG, Topik S (2003) The global coffee economy in Africa, Asia, and Latin America. Cambridge University Press, Cambridge, pp 1500–1989. https://doi.org/10.1017/CBO9780511512193
Cleveland C (1995) The direct and indirect use of fossil fuels and electricity in USA agriculture, 1910-1990. Agric Ecosyst Environ 55(2):111–121. https://doi.org/10.1016/0167-8809(95)00615-Y
Cuadra M, Rydberg T (2006) Energy evaluation on the production, processing and export of coffee in Nicaragua. Ecol Model 196(3):421–433. https://doi.org/10.1016/j.ecolmodel.2006.02.010
Detlefsen G, Somarriba E (2012) Producción de madera en sistemas agroforestales de Centroamérica. Ministry for Foreing Affairs of Finland & CATIE
Deugd M (2003) Crisis del café: nuevas estrategias y oportunidades. Ruta, Costa Rica
DGEC (1953) Censo Agropecuario de 1950. Dirección General de Estadística y Censos
Dicum G, Luttinger N (1999) The coffee book: anatomy of an industry from the crop to the last drop. The New Press, New York.
FAO (2016) FAOSTAT. http://faostat.fao.org. Accessed 19 June 2016
Farfán-Valencia F (2005) Producción de café en un sistema intercalado con plátano dominico hartón con y sin fertilización química. Cenicafé 56(3):269–280
Flauzino A, Fonseca JJ, Jiménez R, Ribeiro A, de Castro Melo E (2014) Energy balance in the production of mountain coffee. Renew Sust Energ Rev 39:1208–1213. https://doi.org/10.1016/j.rser.2014.07.133
Foley JA, DeFries R, Asner GP, Barford C, Bonan G, Carpenter SR, Helkowski JH (2005) Global consequences of land use. Science 309(5734):570–574. https://doi.org/10.1126/science.1111772
Giannetti BF, Ogura Y, Bonilla SH, Almeida CMVB (2011a) Accounting energy flows to determine the best production model of a coffee plantation. Energy Policy 39(11):7399–7407. https://doi.org/10.1016/j.enpol.2011.09.005
Giannetti BF, Ogura Y, Bonilla SH, Almeida CMVB (2011b) Energy assessment of a coffee farm in Brazilian Cerrado considering in a broad form the environmental services, negative externalities and fair price. Agric Syst 104(9):679–688. https://doi.org/10.1016/j.agsy.2011.08.001
González de Molina M, Toledo VM (2014) The social metabolism: a socio-ecological theory of historical change. Springer, New York. https://doi.org/10.1007/978-3-319-06358-4
Grigg D (2002) The worlds of tea and coffee: patterns of consumption. Geo J 57(4):283–294. https://doi.org/10.1023/B:GEJO.0000007249.91153.c
Guzmán G, González de Molina M (2015) Energy efficiency in agrarian systems from an agroecological perspective. Agroecol Sustain Food Syst 39(8):924–952. https://doi.org/10.1080/21683565.2015.1053587
Guzmán G, Aguilera E, Soto D, Cid A, Infante-Amate J, García-Ruiz R, Herrera A, Villa I, de González MM (2014) Methodology and conversión factors to estimate the net primary productivity of historical and contemporary agroecosystems. DT-SEHA 14-07. Documentos de Trabajo de la Sociedad Española de Historia Agraria
Guzmán GI, González de Molina M, Soto Fernández D, Infante-Amate J, Aguilera E (2017) Spanish agriculture from 1900 to 2008: a long-term perspective on agroecosystem energy from an agroecological approach. Reg Environ Chang. https://doi.org/10.1007/s10113-017-1136-2
Haas G, Wetterich F, Geier U (2000) Life cycle assessment framework in agriculture on the farm level. Int J Life Cycle Assess 5(6):345–348. https://doi.org/10.1007/BF02978669
Haberl H, Schulz NB, Plutzar C, Erb KH, Krausmann F, Loibl W, Moser D, Sauberer N, Weisz H, Zechmeister HG, Zulka P (2004) Human appropriation of net primary production and species diversity in agricultural landscapes. Agric Ecosyst Environ 102(2):213–218. https://doi.org/10.1016/j.agee.2003.07.004
Haberl H, Beringer T, Bhattacharya SC, Erb KH, Hoogwijk M (2010) The global technical potential of bio-energy in 2050 considering sustainability constraints. Curr Opin Environ Sustain 2(5):394–403. https://doi.org/10.1016/j.cosust.2010.10.007
Hilje L, Castillo LE, Thrupp L, Wesseling C (1987) El Uso de los Plaguicidas en Costa Rica. Ed. Heliconia/UNED, San José
ICAFE (2007) Censo Cafetalero. 2003-2006. Principales resultados. Instituto del Café de Costa Rica. Instituto Nacional de Estadística y Censos, San José
ICAFE (various years) Instituto del Café de Costa Rica. http://www.icafe.cr. Accessed 19 June 2016
ICAITI (1967) Informe sobre el mercado de fertilizantes en Centroamérica. Anexo 1. Instituto Centroamericano de Investigación y Tecnología Industrial
IDC (1935) Censo Cafetalero de 1935. Instituto de Defensa del Café, Costa Rica
Infante-Amate J (2014) ¿Quién levantó los olivos? Historia de la especialización olivarera en el sur de España (ss. XVIII-XX). Ministerio de Agricultura, Alimentación y Medio Ambiente. Madrid
Infante-Amate J, Picado W (2016) La transición socio-ecológica en el café costarricense. Flujos de energía, materiales y uso del tiempo (1935-2010). International Conference: old and new worlds: the global challenges of rural history, Lisbon, 27–30 January, 2016
Jiménez E, Martínez P (1979) Estudios ecológicos del agroecosistema cafetalero, 2: Producción de materia orgánica en diferentes tipos de estructura [Coffea arabica, Mexico]. Biótica 4(3):109–126
Leach G (1976) Energy and food production. IPC Science and Technology, London
Lemckert A, Campos JJ (1981) Producción y consumo de leña en las fincas pequeñas de Costa Rica. Centro Agronómico Tropical de Investigación y Enseñanza. Turrialba, Costa Rica
López ML, Picado W (2012) Plantas, fertilizantes y transición energética en la caficultura contemporánea de Costa Rica. Bases para una discusión Revista de Historia 65–66:17–51
Maltby C (1980) Use of pesticides in Latin America. UNIDO/IOD.353. United Nations Industrial Development Organization, Viena
Marozzi M, Bellavita G, Varela I (2004) Análisis comparativo de dos fincas productoras de café orgánico utilizando los métodos del balance energético y agroeconómico. Econ Soc 8(24):97–118
Merlo C M (2007) Comportamiento productivo del café (Coffea arabica L variedad caturra), el poró (Erythrinapo eppigiana) el amarillon (Terminalia amazónica) y el Cashá (Choroleuco neurycydum) en sistemas agroforetales bajo manejo convencional y organicos en Turrialba. Dissertation, CATIE, Costa Rica
Montenegro GEJ (2005) Efecto de la dinámica de la materia de nutrientes de la biomasa de tres tipos de árboles de sombra en sistemas de manejo de café orgánico y convencional. Dissertation, CATIE, Costa Rica
Mora-Delgado J, Ramírez C, Quirós O (2006) Análisis beneficio-costo y cuantificación de la energía invertida en sistemas de caficultura campesina en Puriscal, Costa Rica. Agron Costarric 30(2):71–82
Muner LHD, Masera O, Fornazier MJ, Souza CVD, de Loreto MDD (2015) Energetic sustainability of three arabica coffee growing systems used by family farming units in Espírito Santo state. Engenharia Agrícola 35(3):397–405. https://doi.org/10.1590/1809-4430-Eng.Agríc.v35n3p397-405/2015
Murphy DJ, Hall CAS, Dale M, Cleveland C (2011) Order from chaos: a preliminary protocol for determining the EROI of fuels. Sustainabilty 3(12):1888–1907. https://doi.org/10.3390/su3101888
Muschler R (1999) Árboles en cafetales, Materiales de Enseñanza, 5. CATIE, Costa Rica
OAS (1970) La situación de los fertilizantes en Costa Rica. Organization of American States, Washington D.C
Perfecto I, Rice RA, Greenberg R, Van der Voort ME (1996) Shade coffee: a disappearing refuge for biodiversity. Bioscience 46(8):598–608. https://doi.org/10.2307/1312989
Picado, W (2000). La expansión del café y el cambio tecnológico desigual en la agricultura del cantón de Tarrazú, Costa Rica. Dissertation, Universidad Nacional de Costa Rica
Ramírez F (2009) Importación de plaguicidas en Costa Rica. Periodo 2007-2009. Instituto Regional de Estudios en Sustancias Tóxicas. http://cep.unep.org/repcar/informacion-de-paises/costa-rica/Impoortaciones_07-09_REPCar.pdf. Accessed 19 June 2016
Rappaport RA (1969) Pigs for the ancestors: ritual in the ecology of a New Guinea people. Oxford Univerisity Press, Oxford
Renjifo A (1992) El café en Costa Rica. Economía Cafetalera 7:29–77
Rodríguez N, Zambrano DA (2010) Los subproductos del café: fuente de energía renovable. Cenicafé 393:1–8
Romero S (2006) Aporte de biomasa y reciclaje de nutrientes en seis sistemas agroforestales de café (Coffea arabica var. Caturra), con tres niveles de manejo. Tesis de Maestaría, CATIE
Romijn M, Wilderink E (1981) Fuelwood yield from coffee prunings in the Turrialba Valley. CATIE, Turrialba
Salazar F (1985) Producción de leña y biomasa de Inga densiflora Benth en San Ramon, Costa Rica
Salazar A, Palm C (1987) Screening of leguminous tres for alley cropping on acid soils of the humid tropics. In: Khasawneh F, Sample EC, Kamprath EJ (eds) The role of phosphorus in agriculture. Madison, pp 471–514
Santos JJ, Pratt L, Pérez JM (1997) Uso de plaguicidas en la agroindustria de Costa Rica. Centro Latinoamericano para la Competitividad y el Desarrollo Sostenible, San José
Smil V (1999) Crop residues: agriculture’s largest harvest crop residues incorporate more than half of the world’s agricultural phytomass. Bioscience 49(4):299–308. https://doi.org/10.2307/1313613
Smith LG, Williams A, Pearce BD (2015) The energy efficiency of organic agriculture: a review. Renew Agric Food Syst 30(03):280–301. https://doi.org/10.1017/S1742170513000471
Tainter J (1990) The collapse of complex societies. Cambridge University Press, Cambridge
Tello E, Galán E, Sacristán V, Cunfer G, Guzmán GI, González de Molina M, Moreno-Delgado D (2016) Opening the black box of energy throughputs in farm systems: a decomposition analysis between the energy returns to external inputs, internal biomass reuses and total inputs consumed (the Vallès County, Catalonia, c. 1860 and 1999). Ecol Econ 121:160–174. https://doi.org/10.1016/j.ecolecon.2015.11.012
Toledo VM, Barrera-Bassols N (2008) La memoria biocultural: la importancia ecológica de las sabidurías tradicionales. Icaria, Barcelona
Tscharntke T, Clough Y, Bhagwat SA, Buchori D, Faust H, Hertel D, Scherber C (2011) Multifunctional shade-tree management in tropical agroforestry landscapes–a review. J Appl Ecol 48(3):619–629. https://doi.org/10.1111/j.1365-2664.2010.01939.x
Turco PHN, Esperancini MST, Bueno OC (2012) Eficiência energética da produção de café orgânico na região sul de Minas Gerais. Revista Energia na. Agricultura 27:86–95
Funding
This research has been supported by the Social Sciences and Humanities Research Council of Canada (SSHRC 895-2011-1020), Ministerio de Economía y Competitividad (HAR2012-38920-C02-01), and Becas Iberomérica del Banco Santander (2014).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Infante-Amate, J., Picado, W. Energy flows in the coffee plantations of Costa Rica: from traditional to modern systems (1935–2010). Reg Environ Change 18, 1059–1071 (2018). https://doi.org/10.1007/s10113-017-1263-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10113-017-1263-9