Abstract
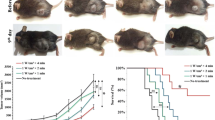
It is well established that laser phototherapy (LP) is contraindicated directly over cancer cells, due to its bio modulatory effects in cell and blood vessel proliferation. The aim of the present study was to analyze the influence of typical low-level laser therapy (LLLT) and high intensity laser therapy (HILT) and an in-between dose of 9 J on collagen fibers and blood vessels content in melanoma tumors (B16F10) implanted in mice. Melanoma tumor cells were injected in male Balb C mice which were distributed in four groups: control (no irradiated) or irradiated by 3, 9, or 21 J (150; 450, or 1050 J/cm2). LP was performed in daily sessions for 3 days with a InGaAlP—660 nm (mean output: 50 mW, spot size: 2 mm2). Tumor volume was analyzed using (1) picrosirius staining to quantify collagen fibers content and (2) Verhoeff’s method to quantify blood vessels content. Tumor growth outcome measured in the 3-J group was not significantly different from controls. Nine and 21-J groups, presented significant and dose-dependent increases in tumor volume. Quantitative analysis of the intensity of collagen fibers and their organization in stroma and peri-tumoral microenvironment showed significant differences between irradiated and control group. Blood vessels count of 21-J group outnumbered the other groups. High doses (≥ 9 J) of LP showed a dose-dependent tumor growth, different collagen fibers characteristics, and eventually blood vessel growth, while a typical LLLT dose (3 J) appeared harmless on melanoma cell activity.






Similar content being viewed by others
References
Chan HHL, Xiang L, Leung JCK, Tsang KWT, Lai K (2003) In vitro study examining the effect of sub-lethal QS 755 nm lasers on the expression of p16INK4a on melanoma cell lines. Lasers Surg Med 32:88–93
Kujawa J, Zavodnik IB, Lapshina A, Labieniec M, Bryszewska M (2004) Cell survival, DNA, and protein damage in B14 cells under low-intensity near-infrared (810 nm) laser irradiation. Photomed Laser Surg 22(6):504–508
Mognato M, Squizzato F, Facchin F, Zaghetto L, Corti L (2004) Cell growth modulation of human cells irradiated in vitro low-level laser therapy. Photomed Laser Surg 22(6):523–526
de Castro JL, Pinheiro AL, Werneck CE, Soares CP (2005) The effect of laser therapy on the proliferation of oral KB carcinoma cells: an in vitro study. Photomed Laser Surg 23(6):586–589
Sroka R, Schaffer M, Fuchs C, Pongratz T, Schrader-Reichard U, Busch M, Schaffer PM, Duhmke E, Baumgartner R (1999) Effects on the mitosis of normal and tumor cells induced by light treatment of different wavelengths. Lasers Surg Med 25(3):263–271
van Leeuwen RL, Dekker SK, Byers HR, Vermeer BJ, Grevelink JM (1996) Modulation of alpha 4 beta 1 and alpha 5 beta 1 integrin expression: heterogeneous effects of Q-switched Ruby, Nd:YAG, and Alexandrite lasers on melanoma cells in vitro. Lasers Surg Med 18(1):63–71
Zhu NW, Perks CM, Burd AR, Holly JM (1999) Changes in the levels of integrin and focal adhesion kinase (FAK) in human melanoma cells following 532 nm laser treatment. Int J Cancer 82(3):353–358
Marchesini R, Dasdia T, Melloni E, Rocca E (1989) Effect of low-energy laser irradiation on colony formation capability in different human tumor cells in vitro. Lasers Surg Med 9(1):59–62
Ocanã-Quero JM, Perez de la Lastra J, Gomez-Villamandos R, Moreno-Millan M (1998) Biological effect of helium-neon (He-Ne) laser irradiation on mouse myeloma (Sp2-Ag14) cell line in vitro. Lasers Med Sci 13:214–218
Jamieson CW, Litwin MS, Longo SE, Krementz ET (1969) Enhancement of melanoma cell culture growth rate by ruby laser radiation. Life Sci 8(2):101–106
Abe M, Fujisawa K, Suzuki H, Sugimoto T, Kanno T (1993) Role of 830 nm low reactive level laser on the growth of an implanted glioma in mice. Keio J Med 42(4):177–179
Mester E, Lapis K, Tota JG (1971) Ultrastructural changes in Ehrlich ascites tumor cells following laser irradiation. Arch Geschwulstforsch 38(3):210–220
Frigo L, Luppi JSS, Favero GM et al (2009) The effect of low-level laser irradiation (In-Ga-Al-AsP—660 nm) on melanoma in vitro and in vivo. BMC Cancer 9, article 404
Lukashev ME, Werb ME (1998) ECM signalling: orchestrating cell behaviour and misbehaviour. Trends Cell Biol 8:437–441
Mettouchi A, Klein S, Guo W, Lopez-Lago M, Lemichez E, Westwick JK, Giancotti FG (2001) Integrin-specific activation of Rac controls progression through the G(1) phase of the cell cycle. Mol Cell 8:115–127
Zhao J, Pestell R, Guan JL (2001) Transcriptional activation of cyclin D1 promoter by FAK contributes to cell cycle progression. Mol Biol Cell 12:4066–4077
Baron-Van Evercooren A, Kleinman HK, Seppä HE, Rentier B, Dubois-Dalcq M (1982) Fibronectin promotes rat Schwann cell growth and motility. J Cell Biol 93(1):211–216
Vitale M, Illario M, Di Matola T, Casamassima A, Fenzi G, Rossi G (1997) Integrin binding to immobilized collagen and fibronectin stimulates the proliferation of human thyroid cells in culture. Endocrinology 138(4):1642–1648
Mester E (1966) The use of the laser beam in therapy. Orv Hetil 107(22):1012–1016
Medrado AR, Pugliese LS, Reis SR, Andrade ZA (2003) Influence of low level laser therapy on wound healing and its biological action upon myofibroblasts. Lasers Surg Med 32(3):239–244
Gavish L, Perez L, Gertz SD (2006) Low-level laser irradiation modulates matrix metalloproteinase activity and gene expression in porcine aortic smooth muscle cells. Lasers Surg Med 38(8):779–786
Choi SK, Kim JH, Lee D, Lee JB, Kim HM, Tchah HW, Hahn TW, Joo M, Ha CI (2008) Different epithelial cleavage planes produced by various epikeratomes in epithelial laser in situ keratomileusis. J Cataract Refract Surg 34(12):2079–2084
Chen CZ, Raghunath M (2009) Focus on collagen: in vitro systems to study fibrogenesis and antifibrosis state of the art. Fibrogenesis Tissue Repair 2:7
Yamamoto Y, Kono T, Kotani H, Kasai S, Mito M (1996) Effect of low-power laser irradiation on procollagen synthesis in human fibroblasts. J Clin Laser Med Surg 14(3):129–132
Ferreira AN, Silveira L, Genovese WJ, de Araújo VC, Frigo L, de Mesquita RA, Guedes E (2006) Effect of GaAIAs laser on reactional dentinogenesis induction in human teeth. Photomed Laser Surg 24(3):358–365
Kikuchi T, Maemondo M, Narumi K, Matsumoto K, Nakamura T, Nukiwa T (2002) Tumor suppression induced by intratumor administration of adenovirus vector expressing NK4, a 4-kringle antagonist of hepatocyte growth factor, and naive dendritic cells. Blood 100(12):3950–3959
Kuwano H, Miyazaki T, Tsutsumi S, Hirayama I, Shimura T, Mochiki E, Nomoto K, Fukuchi M, Kato H, Asao T (2004) Cell density modulates the metastatic aggressiveness of a mouse colon cancer cell line, colon 26. Oncology 67(5–6):441–449
Rich L, Whittaker P (2005) Collagen and picrosirius red staining: a polarized light assessment of fibrillar hue and spatial distribution. Braz J Morphol Sci 22(2):97–104
Barbosa-Júnior AA (2001) Morphological computer-assisted quantitative estimation of stained fibrous tissue in liver sections: applications in diagnosis and experimental research. J Bras Patol 37(3):197–200
Coutinho EM et al (1997) Pathogenesis of schistosomal “pipestem” fibrosis (a low-protein diet inhibits the development of “pipestem” fibrosis in mice). Int J Exp Pathol 78:337–342
Karu TI (1990) Effects of visible radiation on cultured cells. Photochem Photobiol 52:1089–1098
Liotta LA, Kohn EC (2001) The microenvironment of the tumour–host interface. Nature 411(6835):375–379
Mueller MM, Fusenig NE (2004) Friends or foes—bipolar effects of the tumour stroma in cancer. Nat Rev Cancer 4(11):839–849
Tlsty TD, Coussens LM (2006) Tumor stroma and regulation of cancer development. Annu Rev Pathol 1:119–150
Pereira AN, Eduardo Cde P, Matson E, Marques MM (2002) Effect of low-power laser irradiation on cell growth and procollagen synthesis of cultured fibroblasts. Lasers Surg Med 31(4):263–267
Ignatieva N, Zakharkina O, Andreeva I, Sobol E, Kamensky V, Lunin V (2008) Effects of laser irradiation on collagen organization in chemically induced degenerative annulus fibrosus of lumbar intervertebral disc. Lasers Surg Med 40(6):422–432
Prockop D, Kivirikko KI (1995) Collagens: molecular biology, diseases, and potentials for therapy. Annu Rev Biochem 64:403–434
Yu HS, Chang KL, Yu CL, Chen JW, Chen GS (1996) Low-energy helium-neon laser irradiation stimulates interleukin-1 alpha and interleukin-8 release from cultured human keratinocytes. J Investig Dermatol 107(4):593–596
Aimbire F, Albertini R, Pacheco MT et al (2006) Low-level laser therapy induces dose-dependent reduction of TNF-alpha levels in acute inflammation. Photomed Laser Surg 24:33–37
Herz DB, Aitken K, Bagli D (2003) Collagen directly stimulates bladder smooth muscle cell growth in vitro: regulation by extracellular regulated mitogen activated protein kinase. J Urol 170:2072–2076
Noël A, Jost M, Maquoi E (2008) Matrix metalloproteinases at cancer tumor–host interface. Semin Cell Dev Biol 19(1):52–60 Review
Koyama H, Raines EW, Bornfeldt KE, Roberts JM, Ross R (1996) Fibrillar collagen inhibits arterial smooth muscle proliferation through regulation of Cdk2 inhibitors. Cell 87:1069–1078
Bacakova L, Wilhel J, Herget J, Novotna J, Eckhart A (1997) Oxidized collagen stimulates proliferation of vascular smooth muscle cells. Exp Mol Pathol 64:185–194
Henriet P, Zhong ZD, Brooks PC, Weinberg KI, DeClerck YA (2000) Contact with fibrillar collagen inhibits melanoma cell proliferation by up-regulating p27KIP1. Proc Natl Acad Sci U S A 97:10026–10031
Hagendoorn J, Tong R, Fukumura D, Lin Q, Lobo J, Padera TP, Xu L, Kucherlapati R, Jain RK (2006) Onset of abnormal blood and lymphatic vessel function and interstitial hypertension in early stages of carcinogenesis. Cancer Res 66(7):3360–3364
Zhang W, Wu C, Pan W, Tian L, Xia J (2004) Low-power Helium-Neon laser irradiation enhances the expression of VEGF in murine myocardium. Chin Med J 117(10):1476–1480
Ihsan FR (2005) Low-level laser therapy accelerates collateral circulation and enhances microcirculation. Photomed Laser Surg 23(3):289–294
Funding
Professor Lucio Frigo is thankful for the financial support of FAPESP (2007/59124-0).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical commit
All experiments were carried out in accordance with the guidelines from Cruzeiro do Sul University Bioethical Council for human and animal care, PROTOCOL 011/07.
Conflict of interest
The authors declare that they have no conflict of interest.
Disclaimer
Professor Ernesto Cesar Pinto Leal-Junior received research support from Multi Radiance Medical (Solon, OH), a laser device manufacturer.
Informed consent
All authors agree to the submission of this manuscript.
Rights and permissions
About this article
Cite this article
Frigo, L., Cordeiro, J.M., Favero, G.M. et al. High doses of laser phototherapy can increase proliferation in melanoma stromal connective tissue. Lasers Med Sci 33, 1215–1223 (2018). https://doi.org/10.1007/s10103-018-2461-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10103-018-2461-5