Abstract
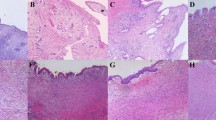
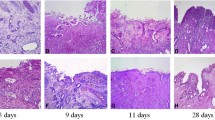
This research aims to observe and compare the wound healing process of urethral bladder after transurethral holmium laser resection of bladder tumor (HoLRBT) and transurethral resection of bladder tumor (TURBT) and explore the possible mechanism of wound healing and bladder re-epithelialization after HoLRBT. An animal model of canine achieving HoLRBT and TURBT was established. Cystoscopy was performed at different time points (3 days and 1, 2, 3, and 4 weeks) after operation to observe the wound healing and re-epithelialization of bladder epithelium. Bladder mucosa specimens were obtained and histopathological changes of the bladder epithelium were observed under light microscope after HE staining. Immunochemistry was used to determine the cell expression ofCK5, CK14, EGF, EGFR; microRNA expressions of CK5, CK14, EGF, and EGFR were measured by qRT-PCR. The changes of urinary EGF concentration were detected by ELISA. The bladder epithelial wound was repaired and re-epithelialized at 1 week after HoLRBT. At the 4th week, the bladder wound was basically completed and re-epithelialized; repair of bladder epithelial wounds recapitulates the wounds with the proliferation and migration of residual epithelial cells under the wound and the bladder epithelium that proliferates alongside the wound surface to complete re-epithelialization. The process begins at 1 week after surgery and basically completes at 4 weeks after surgery. CK5 and CK14 positive cells were detected in the basal cells of the bladder epithelium after HoLRBT, and the expression of CK5 and CK14 mRNA in the basal cells of the bladder epithelium under hyperplasia was significantly higher than that of the normal bladder epithelial basal cells. Bladder epithelial wound repair of TURBT group was performed by the proliferative differentiation of the peri-bladder epithelium adjacent to the wound edge and crawled to the wound surface to complete the re-epithelialization process. The wound repair and re-epithelialization were significantly slower than HoLRBT group. The CK5 and CK14 positive cells can also be detected in the basal cells of marginal hyperplasia of basal margin, and the expression of CK5 and CK14 mRNA in the basal cells of the peri-bladder hyperplasia is obviously higher than that of the normal bladder epithelial basal cells. The expression of EGF in bladder regenerating epithelium gradually increased with time after HoLRBT. Bladder basal cells and bladder regenerating epithelium express high levels of EGFR after HoLRBT. The concentration of EGF in urine after HoLRBT and TURBT increased significantly after surgery, and peaked at 3 days after operation. The urinary EGF concentration in HoLRBT group was higher than that in TURBT group at 3 and 4 weeks after operation. The re-epithelialization process can be seen 1 week after the cystectomy with holmium laser cystectomy, and the epithelialization rate is faster than the traditional transection surgery. This is because the residual bladder epithelial stem cells and wound marginal epithelial cells are involved in the process of wound repair and re-epithelialization following HoLRBT. But only the marginal epithelial tissue participates in the re-epithelialization process after TURBT, so the repair rate of TURBT is slower. The repair of bladder epithelium after HoLRBT is related to the stimulation of tissue factor EGF. The regenerated bladder epithelium also participates in the wound repair process by means of autocrine of EGF.







Similar content being viewed by others
References
Wong MCS et al (2018a) The global epidemiology of bladder cancer: a joinpoint regression analysis of its incidence and mortality trends and projection. Sci Rep 8(1):1129. https://doi.org/10.1038/s41598–018-19199-z
Malats N, Real FX (2015) Epidemiology of bladder cancer. Hematol Oncol Clin North Am 29(2):177–189, vii. https://doi.org/10.1016/j.hoc.2014.10.001
Parsons RL et al (1966) The effect of the laser of dog bladders: a preliminary report. J Urol 95(5):716–717
Fried NM (2006) Therapeutic applications of lasers in urology: an update. Expert Rev Med Devices 3(1):81–94. https://doi.org/10.1586/17434440.3.1.81
Soler-Martinez J et al (2007) Holmium laser treatment for low grade, low stage, noninvasive bladder cancer with local anesthesia and early instillation of mitomycin C. J Urol 178(6):2337–2339. https://doi.org/10.1016/j.juro.2007.08.034
Teng JF et al (2013) Holmium laser versus conventional transurethral resection of the bladder tumor. Chin Med J 126(9):1761–1765
Cano-Garcia MC et al (2016) Outpatient holmium laser treatment for recurrent low-grade superficial bladder cancer under local anesthesia. Minerva Urol Nefrol 68(2):204–208
Muraro GB, Grifoni R, Spazzafumo L (2005) Endoscopic therapy of superficial bladder cancer in high-risk patients: holmium laser versus transurethral resection. Surg Technol Int 14:222–226
Roupret M et al (2018b) European Association of Urology guidelines on upper urinary tract urothelial carcinoma: 2017 update. Eur Urol 73(1):111–122. https://doi.org/10.1016/j.eururo.2017.07.036
Patel SG et al (2015a) Intravesical therapy for bladder cancer. Expert Opin Pharmacother 16(6):889–901. https://doi.org/10.1517/14656566.2015.1024656
Fuchs E, Horsley V (2008) More than one way to skin... Genes Dev. 22(8):976–985. https://doi.org/10.1101/gad.1645908
Hsu YC, Li LS, Fuchs E (2014) Emerging interactions between skin stem cells and their niches. Nat Med 20(8):847–856. https://doi.org/10.1038/nm.3643
Cao Y et al (2015b) Re-epithelialization resulted from prostate basal cells in canine prostatic urethra may represent the ideal healing method after two-micron laser resection of the prostate. Asian J Androl 17(5):831–838. https://doi.org/10.4103/1008-682X.146972
Wang XJ et al (2017) Androgen deprivation accelerates the prostatic urethra wound healing after thulium laser resection of the prostate by promoting re-epithelialization and regulating the macrophage polarization. Prostate 77(7):708–717. https://doi.org/10.1002/pros.23301
Xia SJ (2009) Two-micron (thulium) laser resection of the prostate-tangerine technique: a new method for BPH treatment. Asian J Androl 11(3):277–281. https://doi.org/10.1038/aja.2009.17
Cao Y et al (2010) Changed clathrin regulatory proteins in the brains of Alzheimer’s disease patients and animal models. J Alzheimers Dis 22(1):329–342. https://doi.org/10.3233/JAD-2010-100162
Yang H et al (2017) Comparison of the efficacy and feasibility of laser enucleation of bladder tumor versus transurethral resection of bladder tumor: a meta-analysis. Lasers Med Sci 32(9):2005–2012. https://doi.org/10.1007/s10103–017–2308-5
Raja et al (2007) Wound re-epithelialization: modulating keratinocyte migration in wound healing. Front Biosci 12:2849–2868
Potten CS, Morris RJ (1988) Epithelial stem cells in vivo. J Cell Sci Suppl 10:45–62
Jones PH, Harper S, Watt FM (1995) Stem cell patterning and fate in human epidermis. Cell 80(1):83–93
Prajapati A et al (2013a) Prostate stem cells in the development of benign prostate hyperplasia and prostate cancer: emerging role and concepts. Biomed Res Int 2013:107954. https://doi.org/10.1155/2013/107954
Van Leenders GJLH, Schalken JA (2001) Stem cell differentiation within the human prostate epithelium: implications for prostate carcinogenesis. BJU Int 88:35–42. https://doi.org/10.1046/j.1464-410X.2001.00117.x
Peng LH et al (2013b) Transplantation of bone-marrow-derived mesenchymal and epidermal stem cells contribute to wound healing with different regenerative features. Cell Tissue Res 352(3):573–583. https://doi.org/10.1007/s00441–013–1609-7
McQuibban GA et al (2002) Matrix metalloproteinase processing of monocyte chemoattractant proteins generates CC chemokine receptor antagonists with anti-inflammatory properties in vivo. Blood 100(4):1160–1167
O’Leary R, Arrowsmith M, Wood EJ (2002) Characterization of the living skin equivalent as a model of cutaneous re-epithelialization. Cell Biochem Funct 20(2):129–141. https://doi.org/10.1002/cbf.965
Barrientos S et al (2008) Growth factors and cytokines in wound healing. Wound Repair Regen 16(5):585–601. https://doi.org/10.1111/j.1524-475X.2008.00410.x
Zeng F, Harris RC (2014) Epidermal growth factor from gene organization to bedside. Semin Cell Dev Biol 28:2–11. https://doi.org/10.1016/j.semcdb.2014.01.011
Varley C et al (2005) Autocrine regulation of human urothelial cell proliferation and migration during regenerative responses in vitro. Exp Cell Res 306(1):216–229. https://doi.org/10.1016/j.yexcr.2005.02.004
Freeman MR et al (1997) Heparin-binding EGF-like growth factor is an autocrine growth factor for human urothelial cells and is synthesized by epithelial and smooth muscle cells in the human bladder. J Clin Investig 99(5):1028–1036. https://doi.org/10.1172/Jci119230
Wang C, Ross WT, Mysorekar IU (2017) Urothelial generation and regeneration in development, injury, and cancer. Dev Dyn 246(4):336–343. https://doi.org/10.1002/dvdy.24487
Funding
The present study was supported financially by grants from Shanghai Science and Technology Committee (13DZ1940602), Shanghai Hospital Development Center (SHDC12014215), and Foundation of Shanghai Municipal Commission of Health and Family Planning (20134423).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
Approval for the animal studies was obtained from the Medical Science Ethics Committee of Shanghai First People’s Hospital.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Han, S., Dong, K., Shen, M. et al. Observation and mechanism study of bladder wound healing after transurethral holmium laser resection of bladder tumor. Lasers Med Sci 34, 1217–1227 (2019). https://doi.org/10.1007/s10103-018-02713-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10103-018-02713-0