Abstract
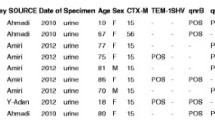
Our aim was to evaluate the prevalence of fosfomycin-resistant strains among ESBL-producing Escherichia coli isolates recovered from community patients in Switzerland. A total of 1225 ESBL-producing E. coli isolates were collected between 2012 and 2013 from a private and community laboratory. Fosfomycin resistance was assessed by using the novel rapid fosfomycin/E. coli NP test and agar dilution method. Resistant isolates were further investigated for acquired resistance genes fosA1–7 by PCR and sequencing. Pulsed-field gel electrophoresis (PFGE) and multilocus sequence typing (MLST) were performed to evaluate the clonal relationship among fosA3-carrying isolates. Out of the 1225 ESBL-producing E. coli isolates analyzed in this study, 1208 were fosfomycin susceptible while 17 were fosfomycin resistant. No discrepancy was observed between the rapid fosfomycin/E. coli NP test and the agar dilution method taken as the gold standard. Five out of the 17 resistant isolates carried a fosA-like gene. No clonal relationship was observed among those isolates. Here, the prevalence of fosfomycin resistance among ESBL-producing E. coli isolates in the community is reported for the first time in Switzerland, being ca. 1.4%. Among the five isolates carrying a fosA gene, four encoded the FosA3 enzyme, being the most prevalent fosfomycin-resistant determinant. An excellent correlation was observed between minimum inhibitory concentration–based susceptibility categorization and results of the rapid fosfomycin/E. coli NP test, further indicating the excellent sensitivity and specificity of this recently developed rapid test whose results are obtained in less than 2 h.
Similar content being viewed by others
References
Flores-Mireles AL, Walker JN, Caparon M, Hultgren SJ (2015) Urinary tract infections: epidemiology, mechanisms of infection and treatment options. Nat Rev Microbiol 13(5):269–284
Pitout JDD, Laupland KB (2008) Extended-spectrum β-lactamase-producing Enterobacteriaceae: an emerging public-health concern. Lancet Infect Dis 8(3):159–166
Ayad A, Drissi M, de Curraize C, Dupont C, Hartmann A, Solanas S et al (2016) Occurence of ArmA and RmtB aminoglycoside resistance 16S rRNA methylases in extended-spectrum ß-lactamases producing Escherichia coli in Algerian hospitals. Front Microbiol 7:1409
Lupo A, Saras E, Madec JY, Haenni M (2018) Emergence of bla CTX-M-55 associated with fosA, rmtB and mcr gene variants in Escherichia coli from various animal species in France. J Antimicrob Chemother 73(4):867–872
Falagas ME, Kastoris AC, Kapaskelis AM, Karageorgopoulos DE (2010) Fosfomycin for the treatment of multidrug-resistant, including extended-spectrum β-lactamase producing, Enterobacteriaceae infections: a systematic review. Lancet Infect Dis 10:43–50
Falagas ME, Vouloumanou EK, Samonis G, Vardakas KZ (2016) Fosfomycin. Clin Microbiol Rev 29(2):321–347
Castaneda-Garcia A, Blazquez J, Rodriguez-Rojas A (2013) Molecular mechanisms and clinical impact of acquired and intrinsic fosfomycin resistance. Antibiotics. 2(2):217–236
Silver LL (2017) Fosfomycin: mechanism and resistance. Cold Spring Harb Perspect Med 7(2):a025262
Nakamura G, Wachino J, Sato N, Kimura K, Yamada K, Jin W et al (2014) Practical agar-based disk potentiation test for detection of fosfomycin-nonsusceptible Escherichia coli clinical isolates producing glutathione S-transferases. J Clin Microbiol 52(9):3175–3179
Ma Y, Xu X, Guo Q, Wang P, Wang W, Wang M (2015) Characterization of fosA5, a new plasmid-mediated fosfomycin resistance gene in Escherichia coli. Lett Appl Microbiol 60(3):259–264
Guo Q, Tomich AD, McElheny CL, Cooper VS, Stoesser N, Wang M et al (2016) Glutathione-S-transferase FosA6 of Klebsiella pneumoniae origin conferring fosfomycin resistance in ESBL-producing Escherichia coli. J Antimicrob Chemother 71(9):2460–2465
Wachino J, Yamane K, Suzuki S, Kimura K, Arakawa Y (2010) Prevalence of fosfomycin resistance among CTX-M-producing Escherichia coli clinical isolates in Japan and identification of novel plasmid-mediated fosfomycin-modifying enzymes. Antimicrob Agents Chemother 54(7):3061–3064
Alrowais H, McElheny CL, Spychala CN, Sastry S, Guo Q, Butt AA et al (2015) Fosfomycin resistance in Escherichia coli, Pennsylvania, USA. Emerg Infect Dis 21(11):2045–2047
Yao H, Wu D, Lei L, Shen Z, Wang Y, Liao K (2016) The detection of fosfomycin resistance genes in Enterobacteriaceae from pets and their owners. Vet Microbiol 193:67–71
Benzerara Y, Gallah S, Hommeril B, Genel N, Decre D, Rottman M et al (2017) Emergence of plasmid-mediated fosfomycin-resistance genes among Escherichia coli isolates, France. Emerg Infect Dis 23(9):1564–1567
Oteo J, Orden B, Bautista V, Cuevas O, Arroyo M, Martinez-Ruiz R et al (2009) CTX-M-15-producing urinary Escherichia coli O25b-ST131-phylogroup B2 has acquired resistance to fosfomycin. J Antimicrob Chemother 64(4):712–717
Peretz A, Naamneh B, Tkhawkho L, Nitzan O (2019) High rates of fosfomycin resistance in Gram-negative urinary isolates from Israel. Microb Drug Resist In press 0(0):1-5
Institute CaLS (2018) Performance standards for antimicrobial susceptibility testing; 28th informational supplement. CLSI document M100-S28. Clinical and Laboratory Standards Institute, Wayne, PA
European Committee on Antimicrobial Susceptibility Testing (2019) Breakpoint tables for interpretation of MICs and zone diameters. Version 9.0. http://www.eucast.org/fileadmin/src/media/PDFs/EUCAST_files/Breakpoint_tables/v_9.0_Breakpoint_Tables.pdf Accessed 10 jan 2019
Nordmann P, Poirel L, Mueller L (2019) Rapid detection of Fosfomycin resistance in Escherichia coli. J Clin Microbiol 57(1):e01531–e01518
Erb S, Frei R, Tschudin Sutter S, Egli A, Dangel M, Bonkat G et al (2018) Basic patient characteristics predict antimicrobial resistance in E. coli from urinary tract specimens: a retrospective cohort analysis of 5246 urine samples. Swiss Med Wkly 148:w14660
Lee SY, Park YJ, Yu JK, Jung S, Kim Y, Jeong SH et al (2012) Prevalence of acquired fosfomycin resistance among extended-spectrum ß-lactamase-producing Escherichia coli and Klebsiella pneumoniae clinical isolates in Korea and IS26-composite transposon surrounding fosA3. J Antimicrob Chemother 67(12):2843–2847
Cattoir V, Guérin F (2018) How is fosfomycin resistance developed in Escherichia coli? Future Microbiol 13(16):1693–1696
Funding
This work was funded by the Swiss National Science Foundation (project FNS-407240_177382).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mueller, L., Cimen, C., Poirel, L. et al. Prevalence of fosfomycin resistance among ESBL-producing Escherichia coli isolates in the community, Switzerland. Eur J Clin Microbiol Infect Dis 38, 945–949 (2019). https://doi.org/10.1007/s10096-019-03531-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-019-03531-0