Abstract
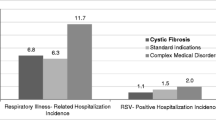
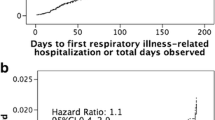
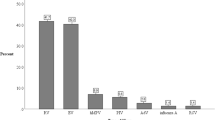
Respiratory syncytial virus (RSV) infection in cystic fibrosis (CF) infants is associated with significant morbidities. This study’s objective is to evaluate the effectiveness and adverse events related to palivizumab (PVZ) in CF infants. Data on respiratory-related illness (RIH) and RSV hospitalizations (RSVH) were collected retrospectively in CF infants aged < 2 years in Alberta, Canada, from 2000 to 2017. Logistic regression models were used to compare the odds of RSVH or RIH in PVZ infants from the Canadian registry of palivizumab (CARESS) versus untreated (UPVZ) infants from Alberta, after adjusting for potential confounders. Illness severity was compared between cohorts using χ2 and t tests. A total of 267 CF infants were included: 183 (PVZ) and 84 (UPVZ). A total of 53.3% were tested for RSV. Fifty-five infants experienced a RIH and 10 had a RSVH. The PVZ cohort experienced similar odds of RSVH but decreased odds of RIH versus UPVZ, adjusting for gestational age, birth weight, birth during RSV peak months, and presence of siblings (Exp(B) = 0.23 [0.11–0.49], p < 0.0005). In RSVH-related subjects, PVZ subjects experienced shorter length of overall stay (LOS; t = 2.39 [df = 7], p = 0.048). In those with a RIH, the PVZ group had shorter overall intensive care unit (t = 3.52 [df = 15], p = 0.003) and hospital LOS (t = 2.11 [df = 52], p = 0.04). No serious adverse events were related to PVZ. The odds of RSVH were similar between groups, but PVZ subjects had decreased odds of RIH. The low number of RSV tests performed may explain the similarity in RSVH rates. Significant differences in LOS may indicate decreased RSVH and RIH illness severity in the PVZ versus UPVZ groups.
Similar content being viewed by others
References
Website:.http://www.cysticfibrosis.ca/wp-content/uploads/2013/10/The-Facts-About-Cystic-Fibrosis.pdf. Accessed 18 Jan 2018
Stick SM, Brennan S, Murray C, Douglas T, von Ungern-Sternberg BS, Garratt LW, Gangell CL, De Klerk N, Linnane B, Ranganathan S, Robinson P, Robertson C, Sly PD (2009) Bronchiectasis in infants and preschool children diagnosed with cystic fibrosis after newborn screening. J Pediatr 155(5):623–628
Sly PD, Brennan S, Gangell C, De Klerk N, Murray C, Mott L, Stick SM, Robinson PJ, Robertson CF, Ranganathan SC (2009) Lung disease at diagnosis in infants with cystic fibrosis detected by newborn screening. Am J Respir Crit Care Med 180(2):146–152
Collins CL, Pollard AJ (2002) Respiratory syncytial virus infections in children and adults. J Inf Secur 45(1):10–17
Figueras-Aloy J, Manzoni P, Paes B, Simões EAF, Bont L, Checchia PA, Faroux B, Carbonell-Estrany X (2016) Defining the risk and associated morbidity and mortality of severe respiratory syncytial virus infection among preterm infants without chronic lung disease or congenital heart disease. Infect Dis Ther 5(4):417–452
Paes B, Fauroux B, Figueras-Aloy J, Bont L, Checchia PA, Simões EAF, Manzoni P, Carbonell-Estrany X (2016) Defining the risk and associated morbidity and mortality of severe respiratory syncytial virus infection among infants with chronic lung disease. Infect Dis Ther 5(4):453–471
Checchia PA, Paes B, Bont L, Manzoni P, Simões EAF, Fauroux B, Figueras-Aloy J, Carbonell-Estrany X (2017) Defining the risk and associated morbidity and mortality of severe respiratory syncytial virus infection among infants with congenital heart disease. Infect Dis Ther 6(1):37–56
Manzoni P, Figueras-Aloy J, Simões EAF, Checchia PA, Fauroux B, Bont L, Paes B, Carbonell-Estrany X (2017) Defining the incidence and associated morbidity and mortality of severe respiratory syncytial virus infection among children with chronic diseases. Infect Dis Ther 6(3):383–411
Arnold SR, Wang EE, Law BJ, Boucher FD, Stephens D, Robinson JL, Dobson S, Langley JM, McDonald J, MacDonald NE, Mitchell I (1999) Variable morbidity of respiratory syncytial virus infection in patients with underlying lung disease: a review of the PICNIC RSV database. Pediatr Infect Dis J 18(10):866–869
Abman SH, Ogle JW, Butler-Simon N, Rumack CM, Accurso FJ (1988) Role of respiratory syncytial virus in early hospitalizations for respiratory distress of young infants with cystic fibrosis. J Pediatr 113(5):826–830
Hiatt PW, Grace SC, Kozinetz CA, Raboudi SH, Treece DG, Taber LH, Piedra PA (1999) Effects of viral lower respiratory tract infection on lung function in infants with cystic fibrosis. Pediatrics 103(3):619–626
Murray J, Bottle A, Sharland M, Modi N, Aylin P, Majeed A, Saxena S (2014) Risk factors for hospital admission with RSV bronchiolitis in England: a population-based birth cohort study. PLoS One 9(2):e89186
Kristensen K, Hjuler T, Ravn H, Simoẽs EAF, Stensballe LG (2012) Chronic diseases, chromosomal abnormalities, and congenital malformations as risk factors for respiratory syncytial virus hospitalization: a population-based cohort study. Clin Infect Dis 54(6):810–817
Resch B (2017) Product review on the monoclonal antibody palivizumab for prevention of respiratory syncytial virus infection. Hum Vaccin Immunother 13(9):2138–2149
The IMpact-RSV Study Group (1998) Palivizumab, a humanized respiratory syncytial virus monoclonal antibody, reduces hospitalization from respiratory syncytial virus infection in high-risk infants. Pediatrics 102(3 Pt 1):531–537
Feltes TF, Cabalka AK, Meissner HC, Piazza FM, Carlin DA, Top FH, Connor EM, Sondheimer HM (2003) Palivizumab prophylaxis reduces hospitalization due to respiratory syncytial virus in young children with hemodynamically significant congenital heart disease. J Pediatr 143(4):532–540
Feltes TF, Sondheimer HM, Tulloh RMR, Harris BS, Jensen KM, Losonsky GA, Griffin MP (2011) A randomized controlled trial of motavizumab versus palivizumab for the prophylaxis of serious respiratory syncytial virus disease in children with hemodynamically significant congenital heart disease. Pediatr Res 70(2):186–191
Paes B, Mitchell I, Li A, Harimoto T, Lanctôt KL (2013) Respiratory-related hospitalizations following prophylaxis in the Canadian registry for palivizumab (2005-2012) compared to other international registries. Clin Dev Immunol 2013:917068
Chan P, Li A, Paes B, Abraha H, Mitchell I, Lanctôt KL (2015) Adherence to palivizumab for respiratory syncytial virus prevention in the Canadian registry of Palivizumab. Pediatr Infect Dis J 34(12):e290–e297
Gaboli M, de la Cruz ÒA, de Agüero MI, Moreno-Galdó A, Pérez GP, de Querol MS (2014) Use of palivizumab in infants and young children with severe respiratory disease: a Delphi study. Pediatr Pulmonol 49(5):490–502
Sánchez-Solis M, Gartner S, Bosch-Gimenez V, Garcia-Marcos L (2013) Is palivizumab effective as a prophylaxis of respiratory syncytial virus infections in cystic fibrosis patients? A meta-analysis. Allergol Immunopathol (Madr) 43(3):298–303
Groves HE, Jenkins L, Macfarlane M, Reid A, Lynn F, Shields MD (2016) Efficacy and long-term outcomes of palivizumab prophylaxis to prevent respiratory syncytial virus infection in infants with cystic fibrosis in Northern Ireland. Pediatr Pulmonol 51(4):379–385
Manzoni P, Paes B, Lanctot KL, Dall’Agnola A, Mitchell I, Calabrese S, Maule M, Girardi E, Harimoto T, Li A (2017) Outcomes of infants receiving Palivizumab prophylaxis for respiratory syncytial virus in Canada and Italy: an international, prospective cohort study. Pediatr Infect Dis J 36(1):2–8
Metz J, Eber E, Resch B (2017) Respiratory syncytial virus infection-associated hospitalization rates in infants and children with cystic fibrosis. Pediatr Infect Dis J 36(6):545–548
Linnane B, Kiernan MG, O’Connell NH, Kearse L, Dunne CP (2015) Anti-RSV prophylaxis efficacy for infants and young children with cystic fibrosis in Ireland. Multidiscip Respir Med 10:32
Winterstein AG, Eworuke E, Xu D, Schuler P (2013) Palivizumab immunoprophylaxis effectiveness in children with cystic fibrosis. Pediatr Pulmonol 48(9):874–884
Giebels K, Marcotte JE, Podoba J, Rousseau C, Denis MH, Fauvel V, Laberge S (2008) Prophylaxis against respiratory syncytial virus in young children with cystic fibrosis. Pediatr Pulmonol 43(2):169–174
Augustine EF, Adams HR, Mink JW (2013) Clinical trials in rare disease: challenges and opportunities. J Child Neurol 28(9):1142–1150
The needs of the few (2010) Nature 466(7303):160
Robinson KA, Odelola OA, Saldanha IJ (2016) Palivizumab for prophylaxis against respiratory syncytial virus infection in children with cystic fibrosis. Cochrane Database Syst Rev 7:CD007743
Website: http://www.cysticfibrosis.ca/de/action/download?downloads=16&file=dl_2016+Registry+Annual+Data+Report.pdf. Accessed 18 Jan 2018
Acknowledgements
The authors wish to acknowledge the valuable contributions of Jenna Cunningham and Dorothy Ens (research assistants) and Monica Ruff (research coordinator).
Funding
This study was funded by an investigator-initiated grant from AbbVie Corporation, Montreal, Canada. The CARESS registry is registered under: ClinicalTrials.gov Identifier: NCT00420966.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
CB received financial support from AbbVie International for travel to an international conference to present the findings of this study. A.L, B.P, I.M, and K.L. have received investigator initiated research funding or received compensation as advisors or lecturers from AbbVie Corporation and MedImmune. PC has no conflicts of interest to declare. No honorarium, grant, or other form of payment was given to anyone to produce the manuscript.
Ethical approval
The study was approved by the research ethics board of the Alberta Children’s Hospital. The CARESS study was reviewed and approved by each site’s respective research ethics board for compliance with the Declaration of Helsinki principles and the standards of Good Clinical Practice.
Informed consent
Informed consent was obtained for the PVZ subjects by each of the 32 recruitment centers across Canada who participated in the CARESS project.
Rights and permissions
About this article
Cite this article
Bjornson, C., Chan, P., Li, A. et al. Palivizumab prophylaxis for respiratory syncytial virus in infants with cystic fibrosis: is there a need?. Eur J Clin Microbiol Infect Dis 37, 1113–1118 (2018). https://doi.org/10.1007/s10096-018-3225-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-018-3225-7