Abstract
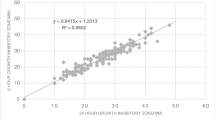
The disk diffusion (DD) method remains the most popular manual technique for antibiotic susceptibility testing (AST) in clinical microbiology laboratories. This is because of its simplicity, reproducibility, and limited cost compared to (automated) microdilution systems, which are usually less sensitive at detecting certain important mechanisms of resistance. Here, we evaluate the PREVI® Isola automated seeder system using a new protocol for spreading bacterial suspensions (eight deposits of calibrated inocula of bacteria, followed by two rounds of rotation) in comparison with manual DD reference testing on a large series of clinical and reference strains. The average time required for seeding one agar plate for DD with this new protocol was 51 s per plate, i.e., 70 agar plates/h. Reproducibility and repeatability was assessed on three reference and three randomly chosen clinical strains, as usually requested by the European Committee on Antimicrobial Susceptibility Testing (EUCAST), and was excellent compared to the manual method. The standard deviations of zones of growth inhibition showed no statistical discrimination. The correlation between the two methods, assessed using 294 clinical isolates and a panel of six antibiotics (n = 3,528 zones of growth inhibition measured), was excellent, with a correlation coefficient of 0.977. The new PREVI® Isola protocol adapted for DD had a sensitivity of 99 % and a specificity of 100 % compared to the manual technique for interpreting DD as recommended by the EUCAST.


Similar content being viewed by others
References
Greub G, Prod’hom G (2011) Automation in clinical bacteriology: what system to choose? Clin Microbiol Infect 17(5):655–660
van Belkum A, Durand G, Peyret M, Chatellier S, Zambardi G, Schrenzel J, Shortridge D, Engelhardt A, Dunne WM Jr (2013) Rapid clinical bacteriology and its future impact. Ann Lab Med 33(1):14–27
Seng P, Drancourt M, Gouriet F, La Scola B, Fournier PE, Rolain JM, Raoult D (2009) Ongoing revolution in bacteriology: routine identification of bacteria by matrix-assisted laser desorption ionization time-of-flight mass spectrometry. Clin Infect Dis 49(4):543–551
Mischnik A, Mieth M, Busch CJ, Hofer S, Zimmermann S (2012) First evaluation of automated specimen inoculation for wound swab samples by use of the Previ Isola system compared to manual inoculation in a routine laboratory: finding a cost-effective and accurate approach. J Clin Microbiol 50(8):2732–2736
Jorgensen JH, Ferraro MJ (2009) Antimicrobial susceptibility testing: a review of general principles and contemporary practices. Clin Infect Dis 49(11):1749–1755
Winstanley T, Courvalin P (2011) Expert systems in clinical microbiology. Clin Microbiol Rev 24(3):515–556
Renaud FN, Bergeron E, Tigaud S, Fuhrmann C, Gravagna B, Freney J (2005) Evaluation of the new Vitek 2 GN card for the identification of gram-negative bacilli frequently encountered in clinical laboratories. Eur J Clin Microbiol Infect Dis 24(10):671–676
Glasson JH, Guthrie LH, Nielsen DJ, Bethell FA (2008) Evaluation of an automated instrument for inoculating and spreading samples onto agar plates. J Clin Microbiol 46(4):1281–1284
Leclercq R, Cantón R, Brown DF, Giske CG, Heisig P, MacGowan AP, Mouton JW, Nordmann P, Rodloff AC, Rossolini GM, Soussy C-J, Steinbakk M, Winstanley TG, Kahlmeter G (2013) EUCAST expert rules in antimicrobial susceptibility testing. Clin Microbiol Infect 19(2):141–160
Galar A, Yuste JR, Espinosa M, Guillén-Grima F, Hernáez-Crespo S, Leiva J (2012) Clinical and economic impact of rapid reporting of bacterial identification and antimicrobial susceptibility results of the most frequently processed specimen types. Eur J Clin Microbiol Infect Dis 31(9):2445–2452
Galar A, Leiva J, Espinosa M, Guillén-Grima F, Hernáez S, Yuste JR (2012) Clinical and economic evaluation of the impact of rapid microbiological diagnostic testing. J Infect 65(4):302–309
Acknowledgments
We are very grateful to Fanny Languasco and Linda Hadjadj for their technical assistance. This work was partly funded by IHU Méditerranée Infection.
Conflict of interest
Alex van Belkum, Corine Fulchiron, and Rachel Huguet are employees of bioMérieux, the company which manufactures and sells the PREVI® Isola instrument.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below are the links to the electronic supplementary material.
Fig. S1
(PPTX 1048 kb)
Table S1
Susceptible, intermediate, or resistant isolates, as determined by the manual technique and PREVI® Isola readings of zone diameters (DOCX 24 kb)
Table S2
Analysis of the discrepant strains (DOCX 39 kb)
Table S3
Comparison of MICs obtained using the manual technique versus the PREVI® Isola protocol (DOCX 49 kb)
Rights and permissions
About this article
Cite this article
Le Page, S., van Belkum, A., Fulchiron, C. et al. Evaluation of the PREVI® Isola automated seeder system compared to reference manual inoculation for antibiotic susceptibility testing by the disk diffusion method. Eur J Clin Microbiol Infect Dis 34, 1859–1869 (2015). https://doi.org/10.1007/s10096-015-2424-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-015-2424-8