Abstract
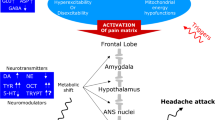
The pathogenesis of migraine as well as cluster headache (CH) is yet a debated question. In this review, we discuss the possible role of the of tyrosine and tryptophan metabolism in the pathogenesis of these primary headaches. These include the abnormalities in the synthesis of neurotransmitters: high level of DA, low level of NE and very elevated levels of octopamine and synephrine (neuromodulators) in plasma of episodic migraine without aura and CH patients. We hypothesize that the imbalance between the levels of neurotransmitters and elusive amines synthesis is due to a metabolic shift directing tyrosine toward an increased decarboxylase and reduced hydroxylase enzyme activities. The metabolic shift of the tyrosine is favored by a state of neuronal hyperexcitability and a reduced mitochondrial activity present in migraine. In addition we present biochemical studies performed in chronic migraine and chronic tension-type headache patients to verify if the same anomalies of the tyrosine and tryptophan metabolism are present in these primary headaches and, if so, their possible role in the chronicity process of CM and CTTH. The results show that important abnormalities of tyrosine metabolism are present only in CM patients (very high plasma levels of DA, NE and tryptamine). Tryptamine plasma levels were found significantly lower in both CM and CTTH patients. In view of this, we propose that migraine and, possibly, CH attacks derive from neurotransmitter and neuromodulator metabolic abnormalities in a hyperexcitable and hypoenergetic brain that spread from the frontal lobe, downstream, resulting in abnormally activated nuclei of the pain matrix. The low tryptamine plasma levels found in CM and CTTH patients suggest that these two primary chronic headaches are characterized by a common insufficient serotoninergic control of the pain threshold.
Similar content being viewed by others
References
Headache Classification Committee of the International Headache Society (IHS) (2013) The international classification of headache disorders, 3 edition (beta version). Cephalagia 33(9):627–808
Welch KMA (1987) Migraine a biobehavioral disorder. Arch Neurol 44:323–327
D’Andrea G, Leon A (2010) Pathogenesis of migraine: from neurotransmitters to neuromodulators and beyond. Neurol Sci Suppl 1:S1–S7
D’Andrea G, Cananzi AR, Ferro Milone F, Joseph R, Grunfeld S, Welch KMA (1989) Platelet levels of glutamate and aspartate in normal subjects. Stroke 20(2):299–300
Millan MJ (2002) Descending control of pain. Prog Neurobiol 66:355–474
D’Andrea G, Nordera GP, Perini F, Allais G, Granella F (2007) Biochemistry of neuro-modulation in primary headaches: focus on tyrosine metabolism. Neurol Sci 28 (Suppl) 2:S94–S96
D’Andrea G, Terrazzino S, Fortin D, Cocco P, Balbi T (2003) Elusive amines and primary headaches: historical background and prospectives. Neurol Sci 24:S65–S67
Axelrod J, Saavedra JM (1977) Octopamine. Nature 265:501–504
Cananzi AR, D’Andrea G, Perini F, Zamberaln F, Welch KMA (1995) Platelet plasma levels of glutamate and glutamine in migraine with and without aura. Cephalalgia 15(2):132–135
Welch KMA, D’Andrea G, Tepley N, Barkley G, Ramadan NM (1990) The concept of migraine as a state of central neuronal hyperexcitability. Neurol Clin 8:817–828
Cao Y, Welch KMA, Aurora S, Vikinsgtad EM (1999) Functional MRI-BOLD of visual triggered headache in patients with migraine. Arch Neurol 56:548–554
Van Gelder N (1987) Calcium mobility and release associated with EEG abnormalities, migraine and epilepsy. Butterworths, Boston, pp 367–378
Prescot A, Becerra L, Pendse G, Shannon T et al (2009) Excitatory neurotransmitters in brain regions in interictal migraine patients. Mol Pain 5–34:1–11
Dela Aleja G, Ramos A, Mato-Abado A, Martinez-Salio A et al (2012) Higher glutamate to glutamine ratios in occipital regions in women with migraine during interictal state. Headache 53(2):365–375
Aurora SK, Ahmad BK, Welch KM, Bhardhwaj P, Ramadan NM (1998) Transcranial magnetic stimulation confirms hyperexcitability of occipital cortex in migraine. Neurology 50(4):1111–1114
Khedr EM, Ahmed MA, Mohamed KA (2006) Motor and visual cortical excitability in migraineurs patients with and without aura: trnscranial magnetic stimulation. Neurophysiol Clin 36(1):13–18
Ramadan NM, Halvorson H, Vande-Linde A, Levine SR, Helpern JA, Welch KM (1989) Low brain magnesium and migraine. Headache 29(7):416–419
Van Harreveld A (1984) The nature of chick’s magnesium sensitive retinal spreading depression. J Neurobiol 15:333–334
Lees G, Leach MJ (1993) Studies on the mechanism of action of novel anticonvulsant lamotrigine (lamictal) using primary neuroglia cultures from rat cortex. Brain Res 612:190–199
D’Andrea G, Granella F, Cadaldini M, Manzoni GC (1999) Effectiveness of lamotrigine in the prophylaxis. Cephalalgia 19:64–66
Lampl C, Buzath A, Klinger D, Neumann K (1999) Lamotrigine in the prophylactic treatment of migraine with aura: a pilot study. Cephalalgia 19:58–63
Chen WT, Fuh JL, Wang SJ (2001) Persistent migrainous visual phenomena might be responsive to lamotrigine. Headache 41(8):823–825
Lampl C, Katsarava Z, Diener HC, Limmroth V (2005) Lamotrigine reduces migraine aura e migraine attacks in patients with migraine with aura. J Neurol Neurosurg Psychiatry 76(12):1730–1732
D’Andrea G, Nordera GP, Allais G (2006) Treatment of the aura: solving the puzzle. Neurol Sci 27 (Suppl) 2:S96–S99
Griffin NJ, Ruggiero L, Lipton RB, Silberstein SD et al (2003) Premonitory symptoms in migraine: an electronic diary study. Neurology 60(6):935–940
Quintela E, Castillo J, Mnoz P, Pascual J (2006) Premonitory and resolution symptoms in migraine: a prospective study in 100 unselected patients. Cephalalgia 26(9):1051–1060
Demarquay G, Royet JP, Giroud P, Chazot G, Valade D, Ryvlin P (2006) Rating of olfactory judgement in migraine patients. Cephalalgia 26:1123–1130
Denuelle M, Fabre N, Payoux P, Chollet F, Geroud G (2007) Hypothalamic activation in spontaneous migraine attacks. Headache 47(10):1418–1426
Cologno D, Ceccarelli G, Petretta V, D’Onofrio F, Bussone G (2008) High prevalence of dopaminergic premonitory symptoms in migraine patients with restless legs syndrome: a pathogenetic link? Neurol Sci 29 Suppl 1:S166–S168
D’Andrea G, Terrazzino S, Leon A, Fortin D, Perini F, Granella F, Bussone G (2004) Elevated levels of circulating trace amines in primary headaches. Neurology 62:1701–1705
D’Andrea G, Granella F, Leone M, Perini F, Farruggio A, Bussone G (2006) Abnormal platelet trace amine profiles in migraine with and without aura. Cephalalgia 26(8):968–972
Moulton EA, Burstein R, Tully S, Hargreaves R, Becerra L, Borsook D (2008) Inter-ictal dysfunction descending modulatory centers in migraine patients. PLoS ONE 3(11):e3799
Fumal A, Laureys S, Di Clemente L, Boly M, Bohotin V, VandenheedeM Coppola GL, Salmon E, Kupers R, Shoenen J (2006) Orbitofrontal cortex involvement in chronic analgesic—overuse headache evolving from episodic migraine. Brain 129:543–550
Welch KMA, Cao Y, Aurora SK, Wiggins G, Vikingstad EM (1998) MRI of the occipital cortex, red nucleus and substantia nigra during visual aura of migraine. Neurology 51:1465–1469
Willer C, May A, Limmroth V, Juptner M, Kaube H, Schayck RV, Coenen HH, Diener HC (1995) Brain stem activation in spontaneous human migraine attacks. Nat Med 1(7):658–660
Altier N, Stewart J (1999) The tachinin NK-1 receptor antagonist RP-67580, infused into ventral tegmental area prevents stress-induced analgesia in the formalin test. Physiol Behav 66:717–721
D’Andrea G, Terrazzino S, Fortin D, Farruggio A, Rinaldi L, Leon A (2003) HPLC electrochemical detection of trace amines in human plasma and platelets and expression of mRNA transcripts of trace amines receptors in circulating leukocytes. Neurosci Lett 346(1–2):89–92
Danielson TJ, Boulton AA, Robertson HS (1977) m-octopamine, p-octopamine and phenylethanolamine in mammalian brain: a sensitive specific assay and effect of drugs. J Neurochem 29:1131–1135
D’Andrea G, Granella F, Perini F, Farruggio A, Leone M, Bussone G (2006) Platelet levels of dopamine are increased in migraine and cluster headache. Headache 46(4):585–591
Fernandez F, Lea RA, Colson NJ, Bellis C, Quinlan S, Griffits LR (2006) Association between a 19 bp deletion polymorphism at dopamine beta-hydroxylase (DBH) locus and migraine with aura. J Neurol Sci 251(1–2):118–123
Magos A, Brincat M, Zilkha KJ, Studd JW (1985) Serum dopamine β-hydroxylase activity in menstrual migraine. J Neurol Neurosurg Psychiatry 48(4):328–331
Gallai V, Gaiti A, Sarchielli P, Coata G, Trequattrini A, Paciaroni M (1992) Evidence for an altered dopamine β-hydroxylase activity in migraine and tension type headache. Acta Neurol Scand 86:403–446
D’Andrea G, Cananzi AR, Morra M, Fornasiero S, Zamberlan F, Welch KMA et al (1989) Platelet as model to test autonomic function in migraine. Funct Neurol 4(1):79–83
Martignoni F, Blandini F, D’Andrea G et al (1990) Platelet and plasma catecholamines in migraine patients. Evidences of menstrual-related variability of the noradrenergic tone. Biog Amines 10:227–237
Nagel-Leiby S, D’Andrea G, Grunfeld S, Welch KMA (1990) Ovarian steroid levels in migraine with and without aura. Cephalalgia 10:147–152
Peres MFP, Sanchez del Rio M, Seabra MLV, Tufik S, Abucham J, Cipolla-Neto J, Siberstein SD, Zuckermann E (2007) Hypothalamic involvement in chronic migraine. J Neurol Neurosurg Psychiatry 71:747–751
D’Andrea G, Hasselmark L, Cananzi AR, Alecci M, Perini F, Zamberlan F, Welch KMA (1995) Metabolism and menstrual cycle rhythmicity of serotonin in primary headaches. Headache 35(4):216–221
Fioroni L, D’Andrea G, Alecci M, Cananzi AR, Facchinetti F (1996) Platelet serotonin pathway in menstrual migraine. Cephalalgia 16(6):427–430
Casucci G, Villani V, Frediani F (2008) Central mechanism of action of antimigraine prophylactic drugs. Neurol Sci 29(Suppl 1):S123–S126
Bigal ME, Hetherington H, Pan J, Tsang A et al (2008) Occipital levels of GABA are related to severe headaches in migraine. Neurology 70(22):2078–2080
Welch KMA, Chabi E, Nell JH, Bartosh K, Achar VS, Meyer JS (1975) Cerebrospinal fluid gamma aminobutyric acid levels and migraine. Br Med J 3(5882):516–517
Yang SP, Pau KY, Spies HG (1997) Gonadectomy alters tyrosine hydroxylase and norepinephrine transporter mRNA levels in the locus coeruleus of the rabbit. J Neuroendocrinol 9(10):763–768
Virmani A, Gaetani F, Imam S, Binienda Z, Ali S (2002) The protective role of l-carnitine against neurotoxicity evoked by drug abuse, methamphetamine, could be related to mitochondrial dysfunction. Ann N Y Acad Sci 965:225–232
Balbi T, Fusco M, Vasapollo D, Boschetto R, Cocco P, Leon A, Farruggio A (2005) The presence of trace amines in postmortem cerebrospinal fluid in humans. J Forensic Sci 50(3):630–632
Welch KMA, Nagesh V, Aurora SK, Gelman N (2001) Periacqueductal gray matter dysfunction in migraine: cause or burden of illness? Headache 41:629–637
Welch KMA (2009) Iron in the migraine brain: a resilient hypothesis. Cephalalgia 29(3):283–285
May A, Barha A, Buchel C, Frakowiak SR, Goasby JP (1998) Hypothalamic activation in cluster headache attacks. Lancet 352:275–278
May A, Barha A, Buchel C, Frakowiak SR, Goasby JP (2000) PET and MRA findings in cluster headache and MRA in experimental pain. Neurology 55:1328–1335
Piacentino M, D’Andrea G, Perini F, Volpin L (2014) Drug-resistant cluster headache: long-term evaluation of pain control by posterior hypothalamic deep-brain stimulation. World Neurosurg 81(2):442e11–15
D’Andrea G, Granella F, Perini F, Farruggio A, Leone M, Bussone G (2006) Platelet levels of dopamine are increased in migraine and cluster headache. Headache 46:585–591
Scher AI, Stewart WF, Ricci JA, Lipton RB (2003) Factors associated with the onset and remission of chronic daily headache in a population-based study. Pain 106:81–89
Katsarava Z, Schneeweiss S, Kurth T et al (2004) Incidence and predictors for chronicity of headache in patients with episodic migraine. Neurology 62:788–790
D’Andrea G, D’Amico D, Bussone G, Bolner A et al (2013) The role of tyrosine metabolism in the pathogenesis of chronic migraine. Cephalalgia 33(11):932–937
Berry DB (2004) Mammalian central nervous system trace amines. Pharmacologic amphetamines, physiologic neuromodulators. J Neurochem 90:257–271
Anwar MA, Ford WR, Herbert AA et al (2013) Signal transduction and modulating pathways in tryptamine-evoked vasopressor responses of the rat isolated perfused mesenteric bed. Vasc Pharmacol 58:140–149
D’Andrea G, D’Amico D, Bussone G, Bolner A et al (2014) Tryptamine levels are low in plasma of chronic migraine and chronic tension type headache. Neurol Sci 35:1941–1945
De Boer SF, Koolhass JM (2005) 5-HT1A and 5-HT1B receptor agonists and aggression: a pharmacological challenge on the serotonin deficiency hypothesis. Eur J Pharmacol 526(1–3):125–139
Huo FQ, Huang FS, Lv BC et al (2010) Activation of serotonin in ventrolateral orbital cortex depresses persistent nociception : a presynaptic inhibition mechanism. Neurochem Int 57:749–755
Pockros AL, Pentkowski NS, Swinford SE et al (2011) Blockade of 5-HT2A in medial prefrontal cortex attenuates reinstatement of cue-elicited cocaine-seeking behavior in rats. Psycopharmacology (Berl) 213(2–3):307–320
QuGL Huo FQ, Huang FS et al (2008) The role of 5-HT receptors subtypes in the ventrolateral orbital cortex of 5-HT-induced antinociception in the rat. Neuroscience 152:487–494
Conflict of interest
The authors declare that they have no conflict of interest related to the publication of this article.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
D’Andrea, G., Cevoli, S., Colavito, D. et al. Biochemistry of primary headaches: role of tyrosine and tryptophan metabolism. Neurol Sci 36 (Suppl 1), 17–22 (2015). https://doi.org/10.1007/s10072-015-2131-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-015-2131-3